DALLAS, July 20, 2021 /PRNewswire/ -- Lantern
Pharma (NASDAQ: LTRN), a clinical stage biopharmaceutical company
using its proprietary RADR® artificial intelligence
("A.I.") platform to transform the cost, pace, and timeline of
oncology drug discovery and development, announced
today positive new data from its ongoing pancreatic cancer
collaboration with the Pancreatic Cancer Institute at Fox Chase
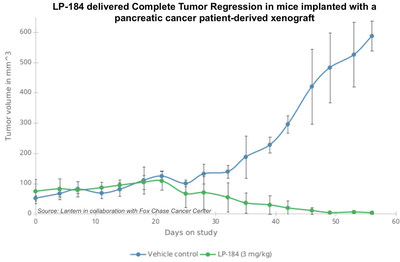
Cancer Center. Preclinical data demonstrated that the drug
candidate, LP-184, demonstrated significant and rapid pancreatic
tumor shrinkage, by over 90%, in in-vivo mouse models in 8
weeks. In comparison, the tumors in the untreated mice grew by over
eleven-fold in volume during the same 8 week period.
Additional positive data on the efficacy and potency of LP-184
was gathered from 6 pancreatic cancer cell lines, and an additional
5 patient-derived xenograft (PDX) ex-vivo tumor models.
Significant reduction of cancer cells and cancer cell growth was
observed across all pancreatic cancer cell lines and PDX models
that were tested in the study with IC50 values being in the
nanomolar range (45-270 nM). These data and observations are being
prepared for peer-reviewed publications, manuscripts and scientific
conferences. Data from this study will be used to power future
insights and analysis provided by Lantern's A.I. platform,
RADR® , in addition to further enhancing the signature
of response for LP-184 in pancreatic cancers. Lantern believes this
to be a significant positive advancement for LP-184 in targeted
pancreatic cancers and plans to advance the collaboration with Fox
Chase Cancer Center into the next phase.
CRISPR knockout data confirmed the importance of PTGR1 in LP-184
cytotoxicity in pancreatic cancer, and validates the RADR-generated
hypothesis that PTGR1 plays the leading role in orchestrating
responsiveness of tumors to LP-184. The research conducted at
Fox Chase Cancer Center leveraged CRISPR editing to silence the
gene PTGR1 in pancreatic cancer cells — this resulted in virtually
no-response by the pancreatic cancer cells to the drug. Those
pancreatic cancer cells with PTGR1 expression (untouched by
gene-editing) had heightened response to LP-184 causing cell death
and IC50 values in the sub-100nM range. Lantern believes that using
a defined genetic signature for patient selection can enhance the
likelihood of clinical trial success and focus future clinical
trials on those patients that will benefit most from the
therapy.
The research has been conducted in collaboration with Dr.
Igor Astsaturov, an established, NCI
funded, physician scientist and co-leader of the Marvin &
Conchetta Greenberg Pancreatic Cancer Institute at Fox Chase Cancer
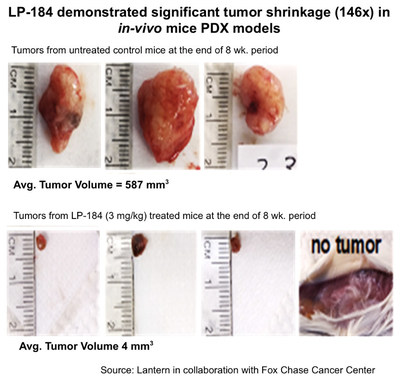
Center. Results demonstrated that LP-184 significantly and rapidly
shrunk pancreatic cancer xenografts in mice, and after treatment
with once weekly dosing at 3 mg/kg for 8 weeks, no tumors were
present in 1 of 4 treated mice and in 3 of 4 mice the average size
of remaining tumors was approximately 7% of the original tumor and
146-fold smaller than the untreated tumors.
Additional research was conducted with Dr. Astsaturov to further
confirm the increased sensitivity to LP-184 in tumors that had
damage to DNA repair pathways. It was observed that pancreatic
tumors that harbored genetic alternations in the following DNA
repair pathways — NER (nucleotide excision repair) and HR
(homologous recombination) — had 2-fold increased sensitivity to
LP-184. This observed data has implications in increasing the
potential number of pancreatic cancer patients that can benefit
from LP-184, and also in confirming prior Lantern research focused
on aiming this drug-candidate at other cancers that have DNA repair
pathway mutations. These could be mutations or deficiencies in
genes such as: BRCA1, BRCA2, ATM, ATR, ERCC2, ERCC3, ERCC4, ERCC5,
ERCC6, FANCD2, RAD51 and PALB2.
"These data mechanistically validate LP-184's potential as a
synthetic lethal agent in many HRD (homologous recombination
deficient) and NERD (nucleotide excision repair deficient)
cancers." stated Dr. Astsaturov. "As a result, these data may
be highly supportive of a future role for LP-184 in a
genetically-defined subset of pancreatic cancer."
Pancreatic cancer is an orphan disease and has a five-year
survival rate of 7.9%. This means that only
approximately 8 in 100 people will have survived for five years and
beyond. The 10-year survival rate of the disease is 1%, meaning
only approximately 1 in 100 people survive 10 years and beyond.
Pancreatic cancer has among the lowest 5-year survival rate of any
of the 22 common cancers. GLOBOCAN estimates that for pancreatic
cancer there are approximately 490,000 thousand new cases of
pancreatic cancer globally, with over 62,000 occurring in
North America annually. Targeting
a specific subset of pancreatic cancer patients that are
genetically defined has the potential to increase beneficial
therapeutic options for patients and may ultimately improve
survival for those with this cancer.
"We are highly encouraged by the results of this preclinical
research and look forward to reporting the full results at future
scientific conferences and in publications," noted Dr. Kishor Bhatia, Lantern's Chief Scientific
Officer. "The study observed the significant and targeted
anti-tumor effects of LP-184, even in pancreatic cancers that were
resistant to standard-of-care drugs. Moreover, we also validated
through the elegant work done with Dr. Astsaturov's lab, by use of
CRISPR-editing, that PTGR1 does directly link to the anti-tumor
activity of LP-184. We expect that we will be able to exploit this
biomarker mechanism in various tumors beyond pancreatic in the
future."
Lantern believes that LP-184 acts by damaging DNA selectively in
tumors that express high levels of the enzyme PTGR1 – which occurs
in several solid tumors. Analysis with Lantern's data platform,
RADR®, indicated that 35-40% of pancreatic cancer
transcriptomes in clinical databases have elevated PTGR1
expression. Lantern has also begun discussions on the design of
first-in-human clinical studies for LP-184 in collaboration with
Dr. Igor Astsaturov and other key
opinion leaders in the pancreatic cancer treatment landscape.
Lantern plans on initiating IND (Investigational New Drug)
application enabling animal studies later this year, and Phase 1
human trials following the filing of a future IND application.
Contacts:
Investor Relations
Panna Sharma, CEO
IR@lanternpharma.com
628-777-3339
Public Relations
Nicholas Koulermos, Vice
President
lantern@5wpr.com
646-843-1812
About Lantern Pharma
Lantern Pharma (LTRN) is a
clinical-stage oncology-focused biopharmaceutical company
leveraging its
proprietary RADR® A.I. platform and
machine learning to discover biomarker signatures that identify
patients most likely to respond to its pipeline
of genomically-targeted therapeutics. Lantern is currently
developing four drug candidates and an ADC program across seven
disclosed tumor targets, including two phase 2 programs. By
targeting drugs to patients whose genomic profile identifies them
as having the highest probability of benefiting from the drug,
Lantern's approach represents the potential to deliver
best-in-class outcomes. More information is available
at: www.lanternpharma.com and Twitter @lanternpharma.
Forward-looking Statements
This press release contains
forward-looking statements within the meaning of Section 27A of the
Securities Act of 1933, as amended, and Section 21E of the
Securities Exchange Act of 1934, as amended. These forward-looking
statements include, among other things, statements relating to:
future events or our future financial performance; the potential
advantages of our RADR® platform in identifying
drug candidates and patient populations that are likely to respond
to a drug candidate; our strategic plans to advance the development
of our drug candidates and antibody drug conjugate (ADC)
development program; estimates regarding the development timing for
our drug candidates and ADC development program; our research and
development efforts of our internal drug discovery programs and the
utilization of our RADR® platform to streamline the
drug development process; our intention to leverage artificial
intelligence, machine learning and genomic data to streamline and
transform the pace, risk and cost of oncology drug discovery and
development and to identify patient populations that would likely
respond to a drug candidate; estimates regarding potential markets
and potential market sizes; sales estimates for our drug candidates
and our plans to discover and develop drug candidates and to
maximize their commercial potential by advancing such drug
candidates ourselves or in collaboration with others. Any
statements that are not statements of historical fact (including,
without limitation, statements that use words such
as "anticipate," "believe," "contemplate," "could,"
"estimate," "expect," "intend," "seek," "may," "might," "plan,"
"potential," "predict," "project," "target," "aim," "should,"
"will," "would," or the negative of these words or other similar
expressions) should be considered forward-looking statements.
There are a number of important factors that could cause our actual
results to differ materially from those indicated by the
forward-looking statements, such as (i) the impact of the COVID-19
pandemic, (ii) the risk that our research and the research of
our collaborators in the area of pancreatic cancer may
not be successful; (iii) the risk that none of our product
candidates has received FDA marketing approval, and we may not be
able to successfully initiate, conduct, or conclude clinical
testing for or obtain marketing approval for our product
candidates; (iv) the risk that no drug product based on our
proprietary RADR A.I. platform has received FDA marketing approval
or otherwise been incorporated into a commercial product, and (v)
those other factors set forth in the Risk Factors section in our
Annual Report on Form 10-K for the year ended December 31, 2020, filed with the Securities and
Exchange Commission on March 10,
2021. You may access our Annual Report on Form 10-K for the
year ended December 31, 2020 under
the investor SEC filings tab of our website at
www.lanternpharma.com or on the SEC's website
at www.sec.gov. Given these risks and uncertainties, we can
give no assurances that our forward-looking statements will prove
to be accurate, or that any other results or events projected or
contemplated by our forward-looking statements will in fact occur,
and we caution investors not to place undue reliance on these
statements. All forward-looking statements in this press release
represent our judgment as of the date hereof, and, except as
otherwise required by law, we disclaim any obligation to update any
forward-looking statements to conform the statement to actual
results or changes in our expectations.

 View original content to download
multimedia:https://www.prnewswire.com/news-releases/lantern-pharma-announces-significant-positive-preclinical-data-in-pancreatic-cancer-with-drug-candidate-lp-184-301337294.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/lantern-pharma-announces-significant-positive-preclinical-data-in-pancreatic-cancer-with-drug-candidate-lp-184-301337294.html
SOURCE Lantern Pharma