Illumina and Next Generation Genomic Partner to Launch VeriSeq™ NIPT Solution in Thailand
May 31 2021 - 4:00PM
Business Wire
Next-generation Sequencing-based CE-IVD Solution Provides
Accurate, Reliable, Fast and Scalable End-to-end Genome-wide
Noninvasive Prenatal Testing
Illumina, Inc., the global leader in DNA sequencing and
array-based technologies and Next Generation Genomic Co., Ltd. (NGG
Thailand), the Association of Southeast Asian Nations (ASEAN)
leaders in laboratory services and reproductive science, today
announced the launch of VeriSeq™ NIPT Solution v2 in Thailand, a
CE-IVD, next-generation sequencing (NGS)-based approach to
noninvasive prenatal testing (NIPT).
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20210531005030/en/
Illumina's VeriSeq NIPT Solution v2
(Photo: Business Wire)
The automated in-lab IVD solution will allow NGG Thailand to
launch the Qualifi Prenatal Test and be the first laboratory in
South East Asia to detect anomalies that targeted assays miss and
deliver more insights into the health of a pregnancy compared to
standard NIPT offerings. Using Illumina’s VeriSeq NIPT Solution v2,
the test delivers a comprehensive view of the fetal genome compared
to other CE-IVD NIPT products, enabling healthcare providers to
support expectant parents with informed, timely and personalized
pregnancy management options better than ever before.
“With expanded NIPT, healthcare providers can obtain a
comprehensive view of all 23 chromosome pairs while limiting risk
to their patient,” said Assoc. Prof. Boonsri Chanrachakul, M.D.,
Ph.D., OB/GYN specializing in Maternal Fetal Medicine, Bangkok,
Thailand. “This technique will offer the accurate, sensitive, and
specific screening for chromosomal changes and provide more
information to healthcare providers and expectant parents to
empower their journey through pregnancy.”
While conventional prenatal screening has been available for
over 30 years, these tests were limited in their ability to screen
beyond aneuploidies of chromosomes 21, 18, and 13. NGG Thailand’s
Qualifi Prenatal Test uses VeriSeq NIPT Solution v2 to provide
accurate information about fetal chromosomal status as early as 10
weeks of gestation using a single maternal blood draw. This
noninvasive test provides a whole-genome sequencing (WGS) approach
to NIPT, expanding prenatal screening beyond the three most common
aneuploidies of chromosomes 21, 18 and 13, to all rare autosomal
aneuploidies (RAAs), sex chromosome aneuploidies (SCAs), and large
partial duplications and deletions.
Illumina Vice President and General Manager of Asia Pacific and
Japan, Ms Gretchen Weightman, explained that the availability of
VeriSeq NIPT Solution v2 is a key milestone in the partnership
between Illumina and NGG Thailand.
“Illumina’s comprehensive technologies, coupled with NGG
Thailand’s expertise, will enable healthcare providers and
expectant parents to unlock the most critical information possible
today,” Ms Weightman said.
The CE-IVD VeriSeq NIPT Solution v2 is now registered for use in
Thailand, Vietnam, Singapore, South Korea, Australia, New Zealand,
Israel, South Africa and across most countries in Europe.
Clinical Accuracy
The clinical accuracy of the CE-IVD VeriSeq NIPT Solution v2,
with respect to outcomes determined by a clinical reference
standard assessment, was demonstrated by evaluating more than 2,300
plasma samples from pregnant women with singleton and twin
pregnancies undergoing prenatal screening for fetal chromosome
aneuploidies and partial deletions and duplications of 7 Mb or
greater. The study determined that VeriSeq NIPT Solution v2
provided highly sensitive and specific results – 98.8 percent
passed assay quality control on the first pass.
Click here for more information on VeriSeq NIPT Solution v2.
ABOUT NEXT GENERATION GENOMIC THAILAND
Next Generation Genomic (NGG) is an excellent center for
reproductive and prenatal genetic testing. Our mission is to
provide high quality laboratory services with continuous quality
improvement and leadership in education. Our services cover all
steps from preconception to postnatal genetic testing. With our
extensive experience and ISO15189/15190 certifications, we provide
reliable, fast and high quality services for the ultimate goal of
having healthy babies and families. To learn more, please visit our
website: www.nggthailand.com and connect with us on Facebook.
ABOUT ILLUMINA
Illumina is improving human health by unlocking the power of the
genome. Our focus on innovation has established us as the global
leader in DNA sequencing and array-based technologies, serving
customers in the research, clinical, and applied markets. Our
products are used for applications in the life sciences, oncology,
reproductive health, agriculture, and other emerging segments. To
learn more, visit www.illumina.com and connect with us on Twitter,
Facebook, LinkedIn, Instagram, and YouTube.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210531005030/en/
Media: Samantha Beal US: +1 858.882.6843
ilmn-pr@illumina.com
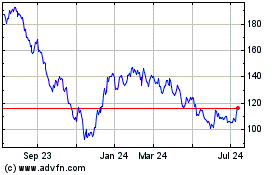
Illumina (NASDAQ:ILMN)
Historical Stock Chart
From Aug 2024 to Sep 2024
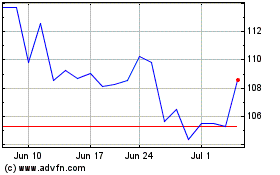
Illumina (NASDAQ:ILMN)
Historical Stock Chart
From Sep 2023 to Sep 2024