As filed with the Securities and Exchange Commission
September 15, 2023
Registration Statement No. 333-
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM S-1
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
ADITXT, INC.
(Exact name of registrant as specified in its charter)
| Delaware |
|
23219 |
|
82-3204328 |
(State or other jurisdiction of
incorporation or organization) |
|
(Primary Standard Industrial
Classification Code Number) |
|
(I.R.S. Employer
Identification Number) |
737 N. Fifth Street, Suite 200
Richmond, VA 23219
(650) 870-1200
(Address and telephone number of registrant’s principal executive offices)
Amro Albanna
Aditxt, Inc.
Chief Executive Officer
737 N. Fifth Street, Suite 200
Richmond, VA 23219
(650) 870-1200
(Name, address, including zip code, and telephone number, including area code, of agent for service)
Copies to:
| Richard A. Friedman, Esq. |
| Sean F. Reid, Esq. |
| Emily A. Mastoloni, Esq. |
| Sheppard, Mullin, Richter & Hampton LLP |
| 30 Rockefeller Plaza |
| New York, NY 10112 |
| Tel: (212) 653-8700 |
| Fax: (212) 653-8701 |
Approximate date of commencement of proposed
sale to the public:
As soon as practicable after the effective date of this registration statement becomes effective.
If any of the securities being
registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933 check
the following box: ☒
If this Form is filed to register
additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities
Act registration statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective
amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement
number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective
amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement
number of the earlier effective registration statement for the same offering. ☐
Indicate by check mark whether
the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company or an emerging
growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting
company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer ☐ |
|
Accelerated filer ☐ |
|
Non-accelerated filer ☒ |
|
Smaller reporting company ☒ |
| |
|
|
|
|
|
Emerging growth company ☒ |
If an emerging growth company,
indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial
accounting standards provided to Section 7(a)(2)(B) of the Securities Act. ☐
The Registrant hereby amends this Registration
Statement on such date or dates as may be necessary to delay its effective date until the Registrant shall file a further amendment which
specifically states that this Registration Statement shall thereafter become effective in accordance with Section 8(a) of the Securities
Act of 1933, or until the Registration Statement shall become effective on such date as the Commission, acting pursuant to said Section
8(a), may determine.
The information in
this preliminary prospectus is not complete and may be changed. The selling stockholders may not sell these securities until the registration
statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these securities
and it is not soliciting an offer to buy these securities in any state or jurisdiction where the offer or sale is not permitted.
| PRELIMINARY PROSPECTUS |
SUBJECT
TO COMPLETION |
DATED
September 15, 2023 |

Aditxt, Inc.
Up to 2,060,000 shares of Common Stock
Pursuant to this prospectus,
the selling stockholders identified herein (the “Selling Stockholders”) are offering on a resale basis an aggregate of up
to 2,060,000 shares of common stock of Aditxt, Inc. (the “Company,” “we,” “us” or our”), par
value $0.001 per share (the “Common Stock”), consisting of (a) up to an aggregate of 1,000,000 shares of Common Stock that
are issuable upon exercise of pre-funded warrants (the “Pre-Funded Warrants”), and (b) up to an aggregate of 1,000,000 shares
of Common Stock that are issuable upon exercise of warrants (the “Common Warrants”), in each cases of (a) and (b) purchased
pursuant to securities purchase agreements by and between us and the Selling Stockholders, each dated, August 31, 2023 (the “Purchase
Agreement”), and (c) up to 60,000 shares of Common Stock that are issuable upon exercise of certain warrants (the “Placement
Agent Warrants”, together with the Pre-Funded Warrants and the Common Warrants, the “Warrants”) issued to designees
of H.C. Wainwright & Co., LLC, our placement agent (the “Placement Agent”) pursuant to an engagement letter in connection
with the Purchase Agreement and the offering contemplated thereunder.
We will not receive any of
the proceeds from the sale by the Selling Stockholders of the Common Stock. Upon any exercise of the Warrants by payment of cash, however,
we will receive the exercise price of the Warrants, which, if exercised in cash with respect to the 2,060,000 shares of Common Stock offered
hereby, would result in gross proceeds to us of approximately $10.75 million. However, we cannot predict when and in what amounts or if
the Warrants will be exercised by payments of cash and it is possible that the Warrants may expire and never be exercised, in which case
we would not receive any cash proceeds. In addition, upon exercise of any of the Common Warrants,
we will pay the Placement Agent a cash fee equal to 7.75% of the gross proceeds received from the exercise of the Common Warrants and
will also issue to the Placement Agent (or its designees) additional Placement Agent Warrants to purchase a number of shares of Common
Stock equal to 6.0% of the aggregate number of shares of Common Stock issued upon such exercise of the Common Warrants.
The Selling Stockholders may
sell or otherwise dispose of the Common Stock covered by this prospectus in a number of different ways and at varying prices. We provide
more information about how the Selling Stockholders may sell or otherwise dispose of the Common Stock covered by this prospectus in the
section entitled “Plan of Distribution” on page 47. Discounts, concessions, commissions and similar selling expenses attributable
to the sale of Common Stock covered by this prospectus will be borne by the Selling Stockholders. We will pay all expenses (other than
discounts, concessions, commissions and similar selling expenses) relating to the registration of the Common Stock with the Securities
and Exchange Commission (the “SEC”).
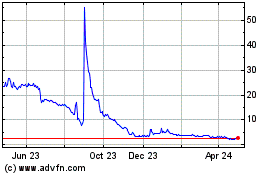
Our common stock is listed
on The Nasdaq Capital Market under the symbol “ADTX”. On September 14, 2023, the closing price as reported on The Nasdaq Capital
Market was $20.46 per share.
We are an “emerging
growth company” under the federal securities laws and, as such, are subject to reduced public company reporting requirements.
Investing in our common
stock involves a high degree of risk. See “Risk Factors” beginning on page 15 of this prospectus.
Neither the Securities
and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus
is truthful or complete. Any representation to the contrary is a criminal offense.
The date of this prospectus is
, 2023
TABLE OF CONTENTS
ABOUT THIS PROSPECTUS
This prospectus relates to
the resale by the Selling Stockholders identified in this prospectus under the caption “Selling Stockholders,” from time to
time, of up to an aggregate of 2,060,000 shares of Common Stock. We are not selling any shares of Common Stock under this prospectus,
and we will not receive any proceeds from the sale of shares of Common Stock offered hereby by the Selling Stockholders, although we may
receive cash from the exercise of the Warrants.
You should rely only on the
information provided in this prospectus, including any information incorporated by reference. We have not authorized anyone to provide
you with any other information and we take no responsibility for, and can provide no assurances as to the reliability of, any other information
that others may give you. The information contained in this prospectus speaks only as of the date set forth on the cover page and may
not reflect subsequent changes in our business, financial condition, results of operations and prospects.
We are not, and the Selling
Stockholders are not, making offers to sell these securities in any jurisdiction in which an offer or solicitation is not authorized or
permitted or in which the person making such offer or solicitation is not qualified to do so or to any person to whom it is unlawful to
make such an offer or solicitation. You should read this prospectus, including any information incorporated by reference, in its entirety
before making an investment decision. You should also read and consider the information in the documents to which we have referred you
in the sections entitled “Where You Can Find More Information” and “Incorporation of Certain Information by Reference.”
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING
STATEMENTS
This prospectus contains forward-looking
statements, which reflect the views of our management with respect to future events and financial performance. These forward-looking statements
are subject to a number of uncertainties and other factors that could cause actual results to differ materially from such statements.
Forward-looking statements are identified by words such as “anticipates,” “believes,” “estimates,”
“expects,” “intends,” “plans,” “projects,” “targets,” and similar expressions.
Such forward-looking statements may be contained in the sections “Risk Factors,” and “Business,” among other places
in this prospectus. Readers are cautioned not to place undue reliance on these forward-looking statements, which are based on the information
available to management at this time and which speak only as of this date. We undertake no obligation to update or revise any forward-looking
statements, whether as a result of new information, future events or otherwise. For a discussion of some of the factors that may cause
actual results to differ materially from those suggested by the forward-looking statements, please read carefully the information under
“Risk Factors.”
The identification in this
document of factors that may affect future performance and the accuracy of forward-looking statements is meant to be illustrative and
by no means exhaustive. All forward-looking statements should be evaluated with the understanding of their inherent uncertainty. You may
rely only on the information contained in this prospectus.
We have not authorized anyone
to provide information different from that contained in this prospectus. Neither the delivery of this prospectus nor the sale of our common
stock means that information contained in this prospectus is correct after the date of this prospectus. This prospectus is not an offer
to sell or solicitation of an offer to buy these securities in any circumstances under which the offer or solicitation is unlawful.
PROSPECTUS SUMMARY
This summary highlights
certain information about us, this offering and selected information contained elsewhere in this prospectus and in the documents incorporated
by reference. This summary is not complete and does not contain all the information that you should consider before deciding whether to
invest in our securities. For a more complete understanding of our company and this offering, we encourage you to read and consider carefully
the more detailed information contained in or incorporated by reference in this prospectus, including the information contained under
the heading “Risk Factors” beginning on page 15 of this prospectus.
Overview and Mission
We believe the world needs—and
deserves—a new approach to innovating that harnesses the power of large groups of stakeholders who work together to ensure that
the most promising innovations make it into the hands of people who need them most.
We were incorporated in the
State of Delaware on September 28, 2017, and our headquarters are in Richmond, Virginia. The company was founded with a mission of bringing
stakeholders together, to transform promising innovations into products and services that could address some of the most challenging needs.
The socialization of innovation through engaging stakeholders in every aspect of it, is key to transforming more innovations, more rapidly,
and more efficiently.
At inception, the first innovation
we took on was an immune modulation technology titled ADI/Adimune with a focus on prolonging life and enhancing life quality of patients
that have undergone organ transplants. Since then, we expanded our portfolio of innovations, and we continue to evaluate a variety of
promising health innovations.
Our Model
Aditxt is not about a single
idea or a single molecule. It is about making sure the right innovation is made possible. Our business model has three main components
as follows:
| |
(1) |
Securing an Innovation: Our process begins with identifying and securing innovations through licensing or acquisition of an innovation asset. Assets come from a variety of sources including research institutions, government agencies, and private organizations. |
| |
(2) |
Growing an Innovation: Once an innovation is secured, we surround it with activation resources that take a systemized approach to bringing that idea to life. Our activation resources include innovation, operations, commercialization, finance, content and engagement, personnel, and administration. |
| |
(3) |
Monetizing an Innovation: Our goal is for each innovation to become commercial-stage and financially and operationally self-sustainable, to create shareholder value. |
We engage various stakeholders
for each of our programs on every level. This includes identifying researchers and research institution partners, such as Stanford University;
leading health institutions to get critical trials underway, such as Mayo Clinic; manufacturing partners who enable us to take innovations
from preclinical to clinical; municipalities and governments, such as the city of Richmond and the state of Virginia and public health
agencies who work with us to launch our program, Pearsanta’s laboratory; and thousands of shareholders around the globe. We seek
to enable promising innovation to become purposeful products that have the power to change lives.
Our Value Proposition
We believe that far too often,
promising treatment or technology does not reach commercialization due to lack of expertise, key resources, or efficiency. As a result,
potentially life-changing and lifesaving treatments are not available to the individuals who so desperately need them.
Aditxt seeks to bring the
holistic concept of an efficient, socialized ecosystem for advancing and accelerating innovations. Our process: We seek to license or
acquire promising innovations. We will then form and build out a subsidiary around each innovation and support the subsidiaries through
innovation, operation, commercialization, content and engagement, finance, personnel, and administration to thrive and grow as a successful,
monetizable business.
Since our inception, we have
built infrastructure consisting of innovation, operation, commercialization, content and engagement, finance, personnel, and administration,
to support the rapid transformation of untapped innovations. Each of the main components of our infrastructure has established global
access to partnerships with industry leaders, top-rated research and medical institutions, universities, manufacturing and distribution
companies, and critical infrastructure such as CLIA-certified state-of-the art labs and GMP manufacturing.
The Shifting Landscape of Innovation
Innovation
in general, and health innovations specifically, require significant resources. The convergence of biotech, high-tech, and media offers
new possibilities of accelerating breakthrough innovations faster and more efficiently. This approach reflects our mission of “Making
Promising Innovations Possible, Together”.
People deserve innovative
solutions, which have never been more within reach. We believe the best idea, best product and the best solution will come from creating
an ecosystem where all stakeholders, such as vendors, customers, municipalities, and shareholders contribute. When we disrupt the way
we’re innovating, through our collaborative model, we believe we can move faster and more efficiently to activate viable solutions
that have the potential to make a measurable impact.
Our Growth Strategy
We believe that the era of
precision and personalized medicine is here and that people around the globe would benefit from health diagnostics and treatments that
more accurately pinpoint the problems and more precisely treat the condition. In addition to our current programs, Adimune and Pearsanta,
we look to bring in future health innovations in the areas of software and AI, medical devices, therapeutics, and other technologies that
take a fundamentally different approach to health because they prioritize personalized precision medicine, timely disease root cause analysis,
and targeted treatments.
Year over year, we plan to
continue building our infrastructure and securing more personalized and precision health innovations that align with our mission. These
opportunities may come in different forms such as IP, an early-stage company, or a late-stage company. We will continue to scale our systemized
approach to the innovation process, making large-scale automation and enterprise systems available to our portfolio companies at every
stage of their growth. Specifically, certain subsidiaries will need to grow through further M&A activities, operational infrastructure
implementation, and development or acquisition of critical technologies.
Our Team
Aditxt is led by an entrepreneurial
team with passion for transforming promising innovations into successful businesses. Our leadership come from a variety of different industries,
with collective expertise in founding startup innovation companies, developing and marketing biopharmaceutical and diagnostic products,
designing clinical trials, manufacturing, and management of private and public companies. We have deep experience in identifying and accessing
promising health innovations and developing them into products and services with the ability to scale. We understand the capital markets,
both public and private, as well as M&A and facilitating complex IPOs.
The following are profiles
of three subsidiaries we have formed, including the terms of the intellectual property licenses that have been sublicensed from Aditxt
to help build each of the businesses.
THE ADITXT PROGRAMS
ADIMUNE, INC.
Formed in January 2023, Adimune™,
Inc. (“Adimune”) is focused on leading our immune modulation therapeutic programs. Adimune’s proprietary immune modulation
product Apoptotic DNA Immunotherapy™, or ADI-100™, utilizes a novel approach that mimics the way our bodies naturally induce
tolerance to our own tissues. It includes two DNA molecules designed to deliver signals to induce tolerance. ADI-100 has been successfully
tested in several preclinical models (e.g., skin grafting, psoriasis, type 1 diabetes, multiple sclerosis).
In May 2023, Adimune entered
into a clinical trial agreement with Mayo Clinic to advance clinical studies targeting autoimmune diseases of the central nervous system
(“CNS”) with the initial focus on the rare, but debilitating, autoimmune disease Stiff Person Syndrome (“SPS”).
According to the National Organization of Rare Diseases, the exact incidence and prevalence of SPS is unknown; however, one estimate places
the incidence at approximately one in one million individuals in the general population.
Pending approval by the International
Review Board, a human trial for SPS is expected get underway in the second half of 2023 or the first half of 2024 with enrollment of 10-15
patients, some of whom may also have type 1 diabetes. ADI-100 will initially be tested for safety and efficacy. ADI-100 is designed to
tolerize against an antigen known as glutamic acid decarboxylase (“GAD”), which is implicated in type-1 diabetes, psoriasis,
and in many autoimmune diseases of the CNS.
Background
The
discovery of immunosuppressive (anti-rejection and monoclonal) drugs over 40 years ago has made possible life-saving organ transplantation
procedures and blocking of unwanted immune responses in autoimmune diseases. However, immune suppression leads to significant undesirable
side effects, such as increased susceptibility to life-threatening infections and cancers, because it indiscriminately and broadly suppresses
immune function throughout the body. While the use of these drugs has been justifiable because they prevent or delay organ rejection,
their use for treatment of autoimmune diseases and allergies may not be acceptable because of the aforementioned side effects. Furthermore,
often transplanted organs ultimately fail despite the use of immune suppression, and about 40% of transplanted organs survive no more
than five years.
Through
Aditxt, Adimune has the right of use to the exclusive worldwide license for commercializing ADI nucleic acid-based technology (which is
currently at the pre-clinical stage) from Loma Linda University. ADI uses a novel approach that mimics the way the body naturally induces
tolerance to our own tissues (“therapeutically induced immune tolerance”). While immune suppression requires continuous administration
to prevent rejection of a transplanted organ, induction of tolerance has the potential to retrain the immune system to accept the organ
for longer periods of time. ADI may allow patients to live with transplanted organs with significantly reduced immune suppression. ADI
is a technology platform which we believe can be engineered to address a wide variety of indications.
Advantages
ADI™
is a nucleic acid-based technology (e.g., DNA-based), which we believe selectively suppresses only those immune cells involved
in attacking or rejecting self and transplanted tissues and organs. It does so by tapping into the body’s natural process of cell
turnover (i.e., apoptosis) to retrain the immune system to stop unwanted attacks on self or transplanted tissues. Apoptosis is a natural
process used by the body to clear dying cells and to allow recognition and tolerance to self-tissues. ADI triggers this process by enabling
the cells of the immune system to recognize the targeted tissues as “self.” Conceptually, it is designed to retrain the immune
system to accept the tissues, similar to how natural apoptosis reminds our immune system to be tolerant to our own “self”
tissues.
While
various groups have promoted tolerance through cell therapies and ex vivo manipulation of patient cells (i.e., takes
place outside the body), to our knowledge, we will be unique in our approach of using in-body induction of apoptosis to promote tolerance
to specific tissues. In addition, ADI treatment itself will not require additional hospitalization but only an injection of minute
amounts of the therapeutic drug into the skin.
Moreover,
preclinical studies have demonstrated that ADI treatment significantly and substantially prolongs graft survival, in addition to successfully
“reversing” other established immune-mediated inflammatory processes.
License Agreement with Loma Linda University (“LLU”)
On March 15, 2018, we entered
into a License Agreement with LLU, which was subsequently amended on July 1, 2020. Pursuant to the LLU License Agreement, we obtained
the exclusive royalty-bearing worldwide license to all intellectual property, including patents, technical information, trade secrets,
proprietary rights, technology, know-how, data, formulas, drawings, and specifications, owned or controlled by LLU and/or any of its affiliates
(the “LLU Patent and Technology Rights”) and related to therapy for immune-mediated inflammatory diseases (the Adi™
technology). In consideration for the LLU License Agreement, we issued 625 shares of common stock to LLU.
PEARSANTA, INC.
Formed in January 2023, our
subsidiary Pearsanta™, Inc. (“Pearsanta”) seeks to take personalized medicine to a whole new level by delivering “Health
by the Numbers.” Since its founding, Pearsanta has been building the platform for enabling our vision of lab quality testing, anytime,
anywhere. Our plan for Pearsanta’s platform is for it to be the transactional backbone for sample collection, sample processing
(on- and off-site), and reporting. This will require the development and convergence of multiple components developed by Pearsanta, or
through transactions with third parties, including collection devices, “lab-on-a-chip” technologies, Lab Developed Test (LDT)
assays, a data-driven analysis engine, and telemedicine. According to a comprehensive research report by Market Research Future, the clinical
and consumer diagnostic market is estimated to hit $429.3 billion by 2030.
We believe that timely and
personalized testing enables far more informed treatment decisions. Pearsanta’s platform is being developed as a seamless digital
healthcare solution. This platform will integrate at-location sample collection, Point-of-Care (“POC”) and LDT assays, and
an analytical reporting engine, with telemedicine-enabled visits with licensed physicians to review test results and, if necessary, order
a prescription. Pearsanta’s goal of extending its platform to enable consumers to monitor their health more proactively as the goal
is to provide a more complete picture about someone’s dynamic health status, factoring in genetic makeup and their response to medication.
The POC component of Pearsanta would enable diagnostic testing at-home, at work, in pharmacies, and more to generate results quickly so
that an individual can access necessary treatment faster. With certain infections, prescribing the most effective treatment according
to one’s numbers can prevent hospital emergency room admissions and potentially life-threatening consequences.
Examples of indication-focused
tests for the Test2Treat platform will include the evaluation for advanced urinary tract infections (“UTIs”), COVID-19/flu/respiratory
syncytial virus, sexually transmitted infections, gut health, pharmacogenomics (i.e., how your genes affect the way your body responds
to certain therapeutics), and sepsis. We believe that these offerings are novel and needed as the current standard of care using broad
spectrum antibiotic treatment can be ineffective and potentially life-threatening. For example, improperly prescribed antibiotics may
approach 50% of outpatient cases. Further, according to an article published in Physician’s Weekly, only 1% of board-certified critical
care medicine physicians are trained in infectious disease.
Licensed Technologies – AditxtScoreTM
We
intend to sublicense to Pearsanta an exclusive worldwide sub-license for commercializing the AditxtScore™ technology which provides
a personalized comprehensive profile of the immune system. AditxtScore is intended to detect individual immune responses to viruses, bacteria,
peptides, drugs, supplements, bone marrow and solid organ transplants, and cancer. It has broad applicability to many other agents of
clinical interest impacting the immune system, including those not yet identified such as emerging infectious agents.
AditxtScore
is being designed to enable individuals and their healthcare providers to understand, manage and monitor their immune profiles and to
stay informed about attacks on or by their immune system. We believe AditxtScore can also assist the medical community and individuals
by being able to anticipate the immune system’s potential response to viruses, bacteria, allergens, and foreign tissues such as
transplanted organs. This technology may be able to serve as a warning signal, thereby allowing for more time to respond appropriately.
Its advantages include the ability to provide simple, rapid, accurate, high throughput assays that can be multiplexed to determine the
immune status with respect to several factors simultaneously, in approximately 3-16 hours. In addition, it can determine and differentiate
between distinct types of cellular and humoral immune responses (e.g., T and B cells and other cell types). It also provides for simultaneous
monitoring of cell activation and levels of cytokine release (i.e., cytokine storms).
We
are actively involved in the regulatory approval process for AditxtScore assays for clinical use and securing manufacturing, marketing,
and distribution partnerships for application in the various markets. To obtain regulatory approval to use AditxtScore as a clinical assay,
we have conducted validation studies to evaluate its performance in detection of antibodies and plan to continue conducting additional
validation studies for new applications in autoimmune diseases.
Advantages
The
sophistication of the AditxtScore technology includes the following:
| |
● |
greater sensitivity/specificity. |
| |
● |
20-fold higher dynamic range, greatly reducing signal to noise compared to conventional assays. |
| |
● |
ability to customize assays and multiplex a large number of analytes with speed and efficiency. |
| |
● |
ability to test for cellular immune responses (i.e., T and B cells and cytokines). |
| |
● |
proprietary reporting algorithm. |
License Agreement with Leland Stanford Junior University (“Stanford”)
On February 3, 2020, we entered
into an exclusive license agreement (the “February 2020 License Agreement”) with Stanford with regard to a patent concerning
a method for detection and measurement of specific cellular responses. Pursuant to the February 2020 License Agreement, we received an
exclusive worldwide license to Stanford’s patent with regard to use, import, offer, and sale of Licensed Products (as defined in
the agreement). The license to the patented technology is exclusive, including the right to sublicense, beginning on the effective date
of the agreement, and ending when the patent expires. Under the exclusivity agreement, we acknowledged that Stanford had already granted
a non-exclusive license in the Nonexclusive Field of Use, under the Licensed Patents in the Licensed Field of Use in the Licensed Territory
(as those terms are defined in the “February 2020 License Agreement”). However, Stanford agreed not to grant further licenses
under the Licensed Patents in the Licensed Field of Use in the Licensed Territory. On December 29, 2021, we entered into an amendment
to the February 2020 License Agreement which extended our exclusive right to license the technology deployed in AditxtScoreTM and
securing worldwide exclusivity in all fields of use of the licensed technology.
ADIVIR, INC.
Formed in April of 2023, Adivir™,
Inc., is Aditxt’s most recently formed wholly owned subsidiary, dedicated to the clinical and commercial development efforts of
innovative antiviral products, starting with Favipiravir-based monotreatment or combination therapies. These products have the potential
to address a wide range of infectious diseases, including those that currently lack viable treatment options.
Background
On
April 18, 2023, we entered into an Asset Purchase Agreement (the “Asset Purchase Agreement”) with Cellvera Global Holdings
LLC (“Cellvera Global”), Cellvera Holdings Ltd. (“BVI Holdco”), Cellvera, Ltd. (“Cellvera Ltd.”),
Cellvera Development LLC (“Cellvera Development” and together with Cellvera Global, BVI Holdco, Cellvera Ltd. and Cellvera
Development (the “Sellers”), AiPharma Group Ltd. (“Seller Owner” and collectively with the Sellers, “Cellvera”),
and the legal representative of Cellvera, pursuant to which, the Company will purchase Cellvera’s 50% ownership interest in G Response
Aid FZE (“GRA”), certain other intellectual property and all goodwill related thereto (the “Acquired Assets”). Unless
expressly stated otherwise herein, capitalized terms used but not defined herein have the meanings ascribed to them in the Asset Purchase
Agreement. Pursuant to the Asset Purchase Agreement, the consideration for the Acquired Assets consists of (A) $24.5 million,
comprised of: (i) the forgiveness of the Company’s $14.5 million loan to Cellvera Global, and (ii) approximately $10 million in
cash, and (B) future revenue sharing payments for a term of seven years. GRA holds an exclusive, worldwide license for the antiviral medication,
Avigan® 200mg, excluding Japan, China and Russia. The other 50% interest in GRA is held by Agility, Inc. (“Agility”).
Additionally,
upon the closing, the Share Exchange Agreement previously entered into as of December 28, 2021, between Cellvera Global Holdings, LLC
f/k/a AiPharma Global Holdings, LLC (together with other affiliates and subsidiaries) and the Company, and all other related agreements
will be terminated.
The
obligations of the Company to consummate the Closing are subject to the satisfaction or waiver, at or prior to the Closing of certain
conditions, including but not limited to, the following:
| |
(i) |
Satisfactory completion of due diligence; |
| |
(ii) |
Completion by the Company of financing sufficient to consummate the transactions contemplated by the Asset Purchase Agreement; |
| |
(iii) |
Receipt by the Company of all required Consents from Governmental Bodies for the Acquisition, including but not limited to, any consents required to complete the transfer and assignment of Cellvera’s membership interests in GRA; |
| |
(iv) |
Receipt of executed payoff letters reflecting the amount required to be fully pay all of each of Seller’s and Seller Owner’s Debt to be paid at Closing; |
| |
(v) |
Receipt by the Company of a release from Agility; |
| |
(vi) |
Execution of an agreement acceptable to the Company with respect to the acquisition by the Company of certain intellectual property presently held by a third party; |
| |
(vii) |
Execution of an amendment to an asset purchase agreement previously entered into by Cellvera with a third party that effectively grants the Company the rights to acquire the intellectual property from the third party under such agreement; |
| |
(viii) |
Receipt of a fairness opinion by the Company with respect to the transactions contemplated by the Asset Purchase Agreement; and |
| |
(ix) |
Receipt by the Company from the Seller Owner of written consent, whether through its official liquidator or the Board of Directors of Seller Owner, to the sale and purchase of the Acquired Assets and Assumed Liabilities pursuant to the Assert Purchase Agreement. |
There
can be no assurance that the conditions to closing will be satisfied or that the proposed acquisition will be completed as proposed or
at all.
Our commitment to building our antiviral portfolio
is strategic and timely. We believe that there has never has there been a more important time to address the growing global need to uncover
new treatments or commercialize existing ones that treat life-threatening global viral infections.
Recent Developments
Nasdaq Hearing
As
previously disclosed on a Current Report on Form 8-K filed in May 26, 2023, on May 23, 2023, we received written notice (the “May
Notification Letter”) from Nasdaq that, based upon the stockholders equity reported by the Company in its Form 10-Q for the
period ended March 31, 2023, and as of March 31, 2023, the Company was no longer in compliance with Nasdaq Listing Rule 5550(b)(1), which
requires a company to maintain a minimum of $2,500,000 in stockholders’ equity, a market value of listed securities of at least
$35 million, or net income from continuing operations of $500,000 in the most recently completed fiscal year or in two of the three most
recently completed fiscal years. The May Notification Letter further provided that the Company had 45 calendar days, or until July 7,
2023, to submit a plan to regain compliance and if the plan is accepted by Nasdaq, an extension of up to 180 calendar days, or until November
19, 2023 to evidence compliance. On June 22, 2023, we received a letter from Nasdaq notifying the Company that it has failed to
maintain compliance with the minimum bid price rule in Nasdaq Listing Rule 5550(a)(2) (the “Minimum Bid Price Rule”) as the
closing price of Company’s common stock has remained below $1.00 for over 30 consecutive trading days. On June 29, 2023, we submitted
an appeal to Nasdaq, which stayed the delisting and suspension of our securities pending the decision of the Nasdaq Hearings Panel (the
“Panel”) no later than 4:00 p.m. Eastern Time on June 29, 2023. The hearing was held on August 31, 2023, which represented
the tenth trading day that the closing price of our Common Stock was above $1.00 per share. At the hearing, we also presented our views
and our plans to regain compliance with the stockholders’ equity requirement to the Panel. It is our understanding that the Panel
typically issues its decision within 30 days after the hearing.
Reverse Stock Split
As previously disclosed on
a Current Report on Form 8-K filed on August 17, 2023, on August 17, 2023, we filed a Certificate of Amendment to our Amended and Restated
Certificate of Amendment with the Secretary of State of the State of Delaware to effect a 1-for-40 reverse stock split of our shares of
Common Stock.
August 2023 Private Placement
As
previously disclosed in a Current Report on Form 8-K filed on September 6, 2023, on August 31, 2023, we entered into a securities purchase
agreement (the “Purchase Agreement”) with an institutional investor for the issuance
and sale in a private placement (the “Private Placement”) of (i) pre-funded warrants (the “Pre-Funded Warrants”)
to purchase up to 1,000,000 shares of the Company’s common stock, par value $0.001 (the “Common Stock”), at an exercise
price of $0.001 per share, and (ii) warrants (the “Common Warrants”) to purchase up to 1,000,000 shares of the Company’s
Common Stock at an exercise price of $10.00 per share.
The
Common Warrants are exercisable immediately upon issuance and have a term of exercise equal to five and one-half years from the date of
issuance. The Pre-Funded Warrants are exercisable immediately and may be exercised at any time until the Pre-Funded Warrants are exercised
in full. A holder of Pre-Funded Warrants or Warrants (together with its affiliates) may not exercise any portion of a warrant to the extent
that the holder would own more than 4.99% (or, at the election of the holder 9.99%) of the Company’s outstanding common stock immediately
after exercise.
In
connection with the Private Placement, we entered into a registration rights agreement (the “Registration Rights Agreement”),
dated as of August 31, 2023, with the investor, pursuant to which the Company agreed to prepare and file a registration statement with
the Securities and Exchange Commission (the “SEC”) registering the resale of the shares of Common Stock underlying the Pre-Funded
Warrants and the Common Warrants no later than 15 days after the date of the Registration Rights Agreement, and to use best efforts to
have the registration statement declared effective as promptly as practical thereafter, and in any event no later than 45 days following
the date of the Registration Rights Agreement (or 75 days following the date of the Registration Rights Agreement in the event of a “full
review” by the SEC).
The
Private Placement closed on September 6, 2023. We received net proceeds from the Private Placement of approximately $9 million, after
deducting placement agent fees and expenses and estimated offering expenses payable by us.
H.C.
Wainwright & Co., LLC (“Wainwright”) served as our exclusive placement agent in connection with the Private Placement,
pursuant to those certain engagement letters, dated as of March 27, 2023 and April 25, 2023, as amended, between the Company and Wainwright
(the “Engagement Letter”). Pursuant to the Engagement Letter, we paid Wainwright (i) a total cash fee equal to 7.75% of the
aggregate gross proceeds of the Private Placement, (ii) a management fee of 1.0% of the aggregate gross proceeds of the Private Placement,
(iii) a non-accountable expense allowance of $50,000, and (iv) $100,000 for legal fees and other out-of-pocket expenses. In addition,
we issued to Wainwright or its designees warrants (the “Placement Agent Warrants”) to purchase up to an aggregate of 60,000
shares of Common Stock at an exercise price equal to $12.50 per share. The Placement Agent Warrants are exercisable immediately upon issuance
and have a term of exercise equal to five and one-half years from the date of issuance.
July Private Placement
On
July 3, 2023, we entered into a Securities Purchase Agreement (the “First Tranche Securities Purchase Agreement”) with an
accredited investor (the “First Tranche Investor”) pursuant to which we issued and sold a secured promissory note in the principal
amount of $375,000 (the “First Tranche Note”) resulting in gross proceeds of $250,000. In connection with the issuance of
the Note, we issued 3,906 shares of common stock (the “First Tranche Commitment Shares”) as a commitment fee to the investor.
Pursuant to the Securities Purchase Agreement, we are obligated to obtain approval of our shareholders (“Shareholder Approval”)
with respect to the issuance of any securities in connection with the Securities Purchase Agreement and the Note in excess of 19.99% of
our issued and outstanding shares on the closing date, which is equal to 33,791 shares of our common stock. Pursuant to the First Tranche
Securities Purchase Agreement, we also granted piggy-back registration rights to the investor with respect to First Tranche Commitment
Shares and any shares of our common stock issuable upon conversion of the First Tranche Note. In addition, we agreed that we would not
enter into any public or private offering of securities that has rights or benefits superior to the First Tranche Investor without providing
such rights and benefits to the First Tranche investor. Under the First Tranche Note, the First Tranche Investor has the right to require
us to immediately apply up to 25% of the cash proceeds from any source, to repay all or any
portion of the outstanding balance of the First Tranche Note. The First Tranche Note has a maturity date of December 31, 2023 and is convertible
following Shareholder Approval and the occurrence of an Event of Default (as defined in the Note) at a conversion price of $18.00 per
share. As of the date of this prospectus, the First Tranche Note has been fully repaid.
In
connection with the First Tranche Securities Purchase Agreement and the issuance of the First Tranche Note, we and certain of our subsidiaries
also entered into a Security Agreement with the investor (the “First Tranche Security Agreement”) pursuant to which we granted
the investor a security interest in certain Collateral (as defined in the First Tranche Security Agreement) to secure our obligations
under the First Tranche Note. In addition, we entered into a Registration Rights Agreement with the investor (the “First Tranche
Registration Rights Agreement”) pursuant to which we agreed to prepare and file with the U.S. Securities and Exchange Commission
a registration statement covering the resale of the First Tranche Commitment Shares and any shares of our common stock issuable upon conversion
of the First Tranche Note within 120 days of the closing date and to have such registration statement declared effective within 150 days
of the closing date.
On
July 24, 2023, we entered into a Securities Purchase Agreement (the “Second Tranche Securities Purchase Agreement”) with an
accredited investor (the “Second Tranche Investor”) pursuant to which the Company issued and sold a secured promissory note
in the principal amount of $2,625,000 (the “Second Tranche Note”) resulting in gross proceeds to the Company of $1,750,000.
In connection with the issuance of the Note, we agreed to issue a total of 27,343 shares of common stock (the “Second Tranche Commitment
Shares”) as a commitment fee to the investor. At the request of the investor, we issued 17,277 Second Tranche Commitment Shares
and will issue the remaining 10,066 Second Tranche Commitment Shares within 120 days, subject to the investor’s discretion. Pursuant
to the Second Tranche Securities Purchase Agreement, we are obligated to obtain approval of our shareholders with respect to the issuance
of any securities in connection with the Second Tranche Securities Purchase Agreement and the Second Tranche Note in excess of 19.99%
of our issued and outstanding shares on the closing date, which is equal to 38,026 shares of the Company’s common stock. Pursuant
to the Second Tranche Securities Purchase Agreement, we also granted piggy-back registration rights to the investor with respect to Second
Tranche Commitment Shares and any shares of our common stock issuable upon conversion of the Second Tranche Note. In addition, we agreed
that we would not enter into any public or private offering of securities that has rights or benefits superior to the Second Tranche Investor
without providing such rights and benefits to the Second Tranche Investor. Under the Second Tranche Note, the Second Tranche Investor
has the right to require us to immediately apply up to 25% of the cash proceeds from any source, to repay all or any portion of the outstanding balance of the Second Tranche Note. The Second Tranche Note has a maturity date of December
31, 2023 and is convertible following shareholder approval and the occurrence of an Event of Default (as defined in the Note) at a conversion
price of $15.60 per share.
In
connection with the Second Tranche Securities Purchase Agreement and the issuance of the Second Tranche Note, we and certain of our subsidiaries
also entered into a Security Agreement with the investor (the “Second Tranche Security Agreement”) pursuant to which we granted
the investor a security interest in certain Collateral (as defined in the Second Tranche Security Agreement) to secure its obligations
under the Second Tranche Note. In addition, we entered into a Registration Rights Agreement with the investor (the “Second Tranche
Registration Rights Agreement”) pursuant to which we agreed to prepare and file with the U.S. Securities and Exchange Commission
a registration statement covering the resale of the Second Tranche Commitment Shares and any shares of our common stock issuable upon
conversion of the Second Tranche Note within 90 days of the closing date and to have such registration statement declared effective within
120 days of the closing date.
July
2023 Loan Transactions
On
July 3, 2023, we entered into a Business Loan and Security Agreement (the “July Loan Agreement”) with a commercial funding
source (the “July Lender”), pursuant to which we obtained a loan from the July Lender in the principal amount of $215,000,
which includes origination fees of $10,750 (the “July Loan”). Pursuant to the July Loan Agreement, we granted the Lender a
continuing secondary security interest in certain collateral (as defined in the July Loan Agreement). The total amount of interest and
fees payable by us to the July Lender under the July Loan will be $322,285, which will be repaid in 13 weekly installments of $24,500
with a final payment of $3,785 in the fourteenth week. As of August 18, 2023, the outstanding balance on the July Loan is $311,076. On
August 23, 2023, we entered into a new Business Loan and Security Agreement with the July Lender, pursuant to which we consolidated the
amounts outstanding under the April Loan and the July Loan and obtained a new loan in the principal amount of $1.4 million.
July 2023 Private
Placement of Series C Preferred Stock
On
July 11, 2023, we entered into a Subscription and Investment Representation Agreement (the “Subscription Agreement”) with
Amro Albanna, our Chief Executive Officer, who is an accredited investor (the “Purchaser”), pursuant to which we agreed to
issue and sell one (1) share of the Company’s Series C Preferred Stock, par value $0.001 per share (the “Preferred Stock”),
to the Purchaser for $1,000.00 in cash. The sale closed on July 11, 2023.
On
July 11, 2023, we filed a certificate of designation (the “Certificate of Designation”) with the Secretary of State of Delaware,
effective as of the time of filing, designating the rights, preferences, privileges and restrictions of the share of Preferred Stock.
The Certificate of Designation provides that the share of Preferred Stock will have 250,000,000 votes and will vote together with the
outstanding shares of our common stock as a single class exclusively with respect to any proposal to amend the Company’s Amended
and Restated Certificate of Incorporation to effect a reverse stock split of our common stock. The Preferred Stock will be voted, without
action by the holder, on any such proposal in the same proportion as shares of common stock are voted. The Preferred Stock otherwise has
no voting rights except as otherwise required by the General Corporation Law of the State of Delaware.
The
Preferred Stock is not convertible into, or exchangeable for, shares of any other class or series of stock or other securities of the
Company. The Preferred Stock has no rights with respect to any distribution of assets of the Company, including upon a liquidation, bankruptcy,
reorganization, merger, acquisition, sale, dissolution or winding up of the Company, whether voluntarily or involuntarily. The holder
of the Preferred Stock will not be entitled to receive dividends of any kind.
The
outstanding share of Preferred Stock shall be redeemed in whole, but not in part, at any time (i) if such redemption is ordered by the
Board of Directors in its sole discretion or (ii) automatically upon the effectiveness of the amendment to the Certificate of Incorporation
implementing a reverse stock split. Upon such redemption, the holder of the Preferred Stock will receive consideration of $1,000.00 in
cash.
Resignation of Executive
Officer
On
July 21, 2023, Matthew Shatzkes tendered his resignation as Chief Legal Officer, General Counsel and Corporate Secretary of the Company.
In connection with his resignation, we entered into a Separation Agreement and General Release with Mr. Shatzkes (the “Separation
Agreement”). Pursuant to the Separation Agreement, Mr. Shatzkes employment with the Company terminated on August 4, 2023 (the “Termination
Date”). In addition, we agreed to pay Mr. Shatzkes within seven days after the Termination Date: (i) $122,292.32, representing all
accrued salary and wages (inclusive of Base Compensation and earned Subsequent Quarterly Bonus amounts, as those terms are defined in
Mr. Shatzkes employment agreement) (the “Accrued Salary and Wages”), and (ii) $32,575.84, representing Mr. Shatzkes accrued,
but unused paid time off (the “Accrued PTO”). On August 11, 2023, we paid Mr. Shatzkes $64,808. Pursuant to the Separation
Agreement, we also agreed to pay Mr. Shatzkes: (i) $385,000, representing 12 months of Mr. Shatzkes Base Compensation (as that term is
defined in Mr. Shatzkes employment agreement) (the “Severance Base Compensation”), and (ii) $290,000, representing Mr. Shatzkes
Subsequent Year Minimum Bonus (as such term is defined in Mr. Shatzkes employment agreement) (the “Severance Bonus”), on the
60th day following the Termination Date. In addition, we are required to reimburse Mr. Shatzkes COBRA premium for a period of 12 months
and shall cause any restricted stock units granted to Mr. Shatzkes to immediately vest as of the Termination Date.
On August 15, 2023, we entered
into an Amendment to Separation Agreement and General Release with Mr. Shatzkes (the “Separation Agreement Amendment”). Pursuant
to the Separation Agreement Amendment, we are required to pay Mr. Shatzkes, upon the earlier of (i) September 1, 2023 or (ii) two business
days following the closing of a capital raise by the Company, an amount equal to $91,060.16, which amount represents the balance of Mr.
Shatzkes’ Accrued Salary and Wages and Accrued PTO plus an additional $1,000 to serve as consideration for entering into the Separation
Agreement Amendment. In addition, under the Separation Agreement Amendment, we are required to pay Mr. Shatzkes the Severance Base Compensation
and the Severance Bonus upon the earlier of (i) the 60th day following the Termination Date or (ii) two business days following
the closing of a capital raise by the Company. We expect to pay the amounts due to Mr. Shatzkes from the proceeds of our recently completed Private Placement.
Termination of Letter
of Intent
On August 1, 2023, the Company
and Natural State Genomics and Natural State Laboratories mutually agreed to terminate the Amended and Restated Non-Binding Letter of
Intent dated June 12, 2023.
August 2023 Secured Loan Transaction
On
August 23, 2023, we entered into a Business Loan and Security Agreement (the “August Loan Agreement”) with a commercial funding
source (the “August Lender”) pursuant to which we obtained a loan from the August Lender in the principal amount of $1,400,000,
which will satisfy the outstanding balances on loans that we originally obtained From the August Lender in April 2023 and July 2023, and
includes origination fees of $70,000 (the “August Loan”). Pursuant to the August Loan Agreement, we granted the August Lender
a continuing secondary security interest in certain collateral (as defined in the August Loan Agreement). The total amount of interest
and fees payable by us to the August Lender under the August Loan will be $2,079,000, which will be repaid in 21 weekly installments of
$99,000.
Corporate Information
We were incorporated as a
Delaware corporation on September 28, 2017. Our principal executive offices are located at 737 N. Fifth Street, Suite 200 Richmond, VA
23219, and our telephone number is (650) 870-1200.
Our common stock trades on
The Nasdaq Capital Market under the symbol “ADTX.”
THE OFFERING
Common Stock to be
offered by the Selling
Stockholders |
|
Up to 2,060,000 shares of Common Stock |
| |
|
|
Common Stock
outstanding prior to this
offering |
|
238,853 shares of Common Stock |
| |
|
|
Common Stock to be
outstanding after this
offering |
|
2,298,853 shares of Common Stock, assuming the exercise of all of the Warrants |
| Use of proceeds |
|
We will not receive any proceeds from the sale of the shares of Common Stock by the Selling Stockholders, except for the Warrant exercise price paid for the Common Stock offered hereby and issuable upon the exercise of the Warrants. See “Use of Proceeds” on page 45 of this prospectus. |
| |
|
|
| Risk factors |
|
See “Risk Factors” beginning on page 15 of this prospectus, as well as other information included in this prospectus, for a discussion of factors you should read and consider carefully before investing in our securities. |
| |
|
|
| Nasdaq Capital Markets symbol |
|
Our common stock is listed on The Nasdaq Capital Markets under the symbol “ADTX”. There is no established trading market for the warrants or the pre-funded warrants, and we do not expect a trading market to develop. We do not intend to list the warrants or the pre-funded warrants on any securities exchange or other trading market. Without a trading market, the liquidity of the warrants and pre-funded warrants will be extremely limited. |
The
number of shares of our common stock to be outstanding after this offering as shown above is based on 238,853 shares outstanding as of
September 14, 2023 and excludes as of that date:
| |
● |
2,258,934 shares of our common stock issuable upon exercise of warrants, subject to vesting having a weighted average exercise price of $34.51 per share; |
| |
● |
1,118 shares of our common stock issuable upon exercise of outstanding options under our 2017 Equity Incentive Plan or the 2017 Plan, subject to vesting; and |
| |
● |
64 shares of our common stock subject to outstanding Restricted Stock Units under our 2021 Omnibus Equity Incentive Plan, or the 2021 Plan, are subject to vesting. |
Except as otherwise indicated
herein, all information in this prospectus assumes no sale of pre-funded warrants, which, if sold, would reduce the number of shares of
common stock that we are offering on a one-for-one basis, no exercise of the warrants or placement agent warrants issued in this offering,
and no exercise of options issued under our Plan or of warrants described above, including the Placement Agent Warrants.
RISK FACTORS
An investment in our securities
involves a high degree of risk. This prospectus contains a discussion of the risks applicable to an investment in our securities. Prior
to deciding about investing in our securities, you should carefully consider the specific factors discussed within this prospectus. The
risks and uncertainties we have described are not the only ones we face. Additional risks and uncertainties not presently known to us
or that we currently deem immaterial may also affect our operations. The occurrence of any of these known or unknown risks might cause
you to lose all or part of your investment in the offered securities.
Risks Related to Our Financial Position and
Need for Capital
Our financial situation creates doubt whether
we will continue as a going concern.
We were incorporated in September
2017 and have a limited operating history and our business is subject to all the risks inherent in the establishment of a new business
enterprise. Our likelihood of success must be considered in light of the problems, expenses, difficulties, complications and delays frequently
encountered in connection with development and expansion of a new business enterprise. Since inception, we have incurred losses and expect
to continue to operate at a net loss for at least the next several years as we commence our research and development efforts, conduct
clinical trials, and develop manufacturing, sales, marketing, and distribution capabilities. Our net loss for the years ended December
31, 2022 and 2021 was $27,649,876 and $46,371,364, respectively, and our accumulated deficit as of December 31, 2022 was $95,040,362.
Our net loss for the six months ended June 30, 2023 and 2022 was $11,666,724 and $11,909,147, respectively, and our accumulated deficit
as of June 30, 2023 was $106,707,086. There can be no assurance that the products under development by us will be approved for sale in
the U.S. or elsewhere. Furthermore, there can be no assurance that if such products are approved, they will be successfully commercialized,
and the extent of our future losses and the timing of our profitability are highly uncertain. If we are unable to achieve profitability,
we may be unable to continue our operations. There can be no assurances that we will be able to achieve a level of revenues adequate to
generate sufficient cash flow from operations or additional financing through private placements, public offerings and/or bank financing
necessary to support our working capital requirements. To the extent that funds generated from any private placements, public offerings
and/or bank financing are insufficient, we will have to raise additional working capital. No assurance can be given that additional financing
will be available, or if available, will be on acceptable terms. These conditions raise substantial doubt about our ability to continue
as a going concern. If adequate working capital is not available, we may be forced to discontinue operations, which would cause investors
to lose their entire investment.
Our cash and cash equivalents
were approximately $6,364,409 as of September 12, 2023. We will not receive any of the proceeds from the sale by the Selling Stockholders
of the Common Stock. Upon any exercise of the Warrants by payment of cash, however, we will receive the exercise price of the Warrants,
which, if exercised in cash with respect to the 2,060,000 shares of Common Stock offered hereby, would result in gross proceeds to us
of approximately $10.75 million. However, we cannot predict when and in what amounts or if the Warrants will be exercised by payments
of cash and it is possible that the Warrants may expire and never be exercised, in which case we would not receive any cash proceeds.
In addition, upon exercise of any of the Common Warrants, we will pay the Placement Agent a
cash fee equal to 7.75% of the gross proceeds received from the exercise of the Common Warrants and will also issue to the Placement Agent
(or its designees) additional Placement Agent Warrants to purchase a number of shares of Common Stock equal to 6.0% of the aggregate number
of shares of Common Stock issued upon such exercise of the Common Warrants. There can be no assurance that the Warrants will be exercised
for cash, and if they are not, that it will not have a material adverse effect on our business.
If we fail to obtain the capital necessary
to fund our operations, we will be unable to continue or complete our product development and you will likely lose your entire investment.
We will need to continue to
seek capital from time to time to continue development of our lead drug candidate beyond our initial combined Phase I/Iia clinical trial
and to acquire and develop other product candidates. Once approved for commercialization, we cannot provide any assurances that any revenues
it may generate in the future will be sufficient to fund our ongoing operations.
Our business or operations
may change in a manner that would consume available funds more rapidly than anticipated and substantial additional funding may be required
to maintain operations, fund expansion, develop new or enhance products, acquire complementary products, business or technologies, or
otherwise respond to competitive pressures and opportunities, such as a change in the regulatory environment or a change in preferred
treatment modalities. In addition, we may need to accelerate the growth of our sales capabilities and distribution beyond what is currently
envisioned, and this would require additional capital. However, we may not be able to secure funding when we need it or on favorable terms.
We may not be able to raise sufficient funds to commercialize the product candidates we intend to develop.
If we cannot raise adequate
funds to satisfy our capital requirements, we will have to delay, scale back or eliminate our research and development activities, clinical
studies, or future operations. We may also be required to obtain funds through arrangements with collaborators, which arrangements may
require us to relinquish rights to certain technologies or products that we otherwise would not consider relinquishing, including rights
to future product candidates or certain major geographic markets. This could result in sharing revenues which we might otherwise retain
for ourselves. Any of these actions may harm our business, financial condition, and results of operations.
The amount of capital we may
need depends on many factors, including the progress, timing and scope of our product development programs; the progress, timing and scope
of our preclinical studies and clinical trials; the time and cost necessary to obtain regulatory approvals; the time and cost necessary
to further develop manufacturing processes and arrange for contract manufacturing; our ability to enter into and maintain collaborative,
licensing and other commercial relationships; and our partners’ commitment of time and resources to the development and commercialization
of our products.
Our obligations
to certain of our creditors are secured by security interests in our assets, so if we default on those obligations, our creditors could
foreclose on some or all of our assets.
Our
obligations to certain of our creditors are secured by security interests in our assets. As of September 14, 2023, approximately 3.4
million was owed to such secured creditors. Under such agreements, we are required to pay $201,857 on a weekly basis to such creditors.
As of the date of this prospectus, certain of our secured creditors have agreed to a temporary reduction in the required weekly payments.
If we default on our obligations under these agreements, our secured creditors could foreclose on its security interests and liquidate
some or all of these assets, which would harm our financial condition and results of operations and would require us to reduce or cease
operations and possibly seek Bankruptcy Protection.
In the event we
pursue Bankruptcy Protection, we will be subject to the risks and uncertainties associated with such proceedings.
In the event we file for relief
under the United States Bankruptcy Code, our operations, our ability to develop and execute our business plan and our continuation as
a going concern will be subject to the risks and uncertainties associated with bankruptcy proceedings, including, among others: our ability
to execute, confirm and consummate a plan of reorganization; the additional, significant costs of bankruptcy proceedings and related fees;
our ability to obtain sufficient financing to allow us to emerge from bankruptcy and execute our business plan post-emergence, and our
ability to comply with terms and conditions of that financing; our ability to continue our operations in the ordinary course; our ability
to maintain our relationships with our consumers, business partners, counterparties, employees and other third parties; our ability to
obtain, maintain or renew contracts that are critical to our operations on reasonably acceptable terms and conditions; our ability to
attract, motivate and retain key employees; the ability of third parties to use certain limited safe harbor provisions of the United States
Bankruptcy Code to terminate contracts without first seeking Bankruptcy Court approval; the ability of third parties to force us to into
Chapter 7 proceedings rather than Chapter 11 proceedings and the actions and decisions of our stakeholders and other third parties who
have interests in our bankruptcy proceedings that may be inconsistent with our operational and strategic plans. Any delays in our bankruptcy
proceedings would increase the risks of our being unable to reorganize our business and emerge from bankruptcy proceedings and may increase
our costs associated with the bankruptcy process or result in prolonged operational disruption for us. Also, we would need the prior approval
of the bankruptcy court for transactions outside the ordinary course of business during the course of any bankruptcy proceedings, which
may limit our ability to respond timely to certain events or take advantage of certain opportunities. Because of the risks and uncertainties
associated with any bankruptcy proceedings, we cannot accurately predict or quantify the ultimate impact of events that could occur during
any such proceedings. There can be no guarantees that if we seek Bankruptcy Protection we will emerge from Bankruptcy Protection as a
going concern or that holders of our common stock will receive any recovery from any bankruptcy proceedings.
In the event we
are unable to pursue Bankruptcy Protection under Chapter 11 of the United States Bankruptcy Code, or, if pursued, successfully emerge
from such proceedings, it may be necessary to pursue Bankruptcy Protection under Chapter 7 of the United States Bankruptcy Code for all
or a part of our businesses.
In the event we are unable
to pursue Bankruptcy Protection under Chapter 11 of the United States Bankruptcy Code, or, if pursued, successfully emerge from such proceedings,
it may be necessary for us to pursue Bankruptcy Protection under Chapter 7 of the United States Bankruptcy Code for all or a part of our
businesses. In such event, a Chapter 7 trustee would be appointed or elected to liquidate our assets for distribution in accordance with
the priorities established by the United States Bankruptcy Code. We believe that liquidation under Chapter 7 would result in significantly
smaller distributions being made to our stakeholders than those we might obtain under Chapter 11 primarily because of the likelihood that
the assets would have to be sold or otherwise disposed of in a distressed fashion over a short period of time rather than in a controlled
manner and as a going concern.
We will need to raise substantial additional
capital, which may not be available on acceptable terms, or at all. Failure to obtain this necessary capital when needed may force us
to delay, limit or terminate our product development efforts or cease operations.
We do not expect that our
current cash position will be sufficient to fund our current operations for the next 12 months. Our operating plan may change because
of many factors currently unknown to us, and we may need to seek additional funds sooner than planned, through public or private equity
or debt financings, government or other third-party funding, marketing and distribution arrangements and other collaborations, strategic
alliances and licensing arrangements or a combination of these approaches. In any event, we will require additional capital to obtain
regulatory approval for, and to commercialize, our product candidates. Raising funds in the current economic environment may present additional
challenges. Even if we believe we have sufficient funds for our current or future operating plans, we may seek additional capital if market
conditions are favorable or if we have specific strategic considerations.
Any additional fundraising
efforts may divert our management from their day-to-day activities, which may adversely affect our ability to develop and commercialize
our product candidates. In addition, we cannot guarantee that future financing will be available in sufficient amounts or on terms acceptable
to us, if at all. Moreover, the terms of any financing may adversely affect the holdings or the rights of our stockholders and the issuance
of additional securities, whether equity or debt, by us, or the possibility of such issuance, may cause the market price of our shares
to decline. The sale of additional equity or convertible securities may dilute our existing stockholders. The incurrence of indebtedness
would result in increased fixed payment obligations, and we may be required to agree to certain restrictive covenants, such as limitations
on our ability to incur additional debt, limitations on our ability to acquire, sell or license intellectual property rights and other
operating restrictions that could adversely impact our ability to conduct our business. We could also be required to seek funds through
arrangements with collaborative partners or otherwise at an earlier stage than otherwise would be desirable and we may be required to
relinquish rights to some of our technologies or product candidates or otherwise agree to terms unfavorable to us, any of which may have
a material adverse effect on our business, operating results and prospects.
If we are unable to obtain
funding on a timely basis, we may be required to significantly curtail, delay, or discontinue one or more of our research or development
programs or the commercialization of any product candidate or be unable to expand our operations or otherwise capitalize on our business
opportunities, as desired, which could materially affect our business, financial condition and results of operations.
Even if we can raise additional funding,
we may be required to do so on terms that are dilutive to you.
The capital markets have been
unpredictable in the past for unprofitable companies such as ours. In addition, it is generally difficult for development stage companies
to raise capital under current market conditions. The amount of capital that a company such as ours is able to raise often depends on
variables that are beyond our control. As a result, we may not be able to secure financing on terms attractive to us, or at all. If we
can consummate a financing arrangement, the amount raised may not be sufficient to meet our future needs. If adequate funds are not available
on acceptable terms, or at all, our business, including our results of operations, financial condition and our continued viability will
be materially adversely affected.
Risks Related to Product Development, Regulatory
Approval, Manufacturing and Commercialization
The regulatory approval process is expensive,
time-consuming, and uncertain and may prevent us from obtaining approvals for the commercialization of our future product candidates,
if any.
We will not be permitted to
market our product candidates in the United States until we receive approval from the FDA, or in any foreign countries until we receive
the requisite approval from corresponding agencies in such countries. The testing, manufacturing, labeling, approval, selling, marketing
and distribution of health and life science-related products are subject to extensive regulation, which regulations differ from country
to country.
Successfully completing our
clinical program and obtaining approval of a Biologics License Application (“BLA”) is a complex, lengthy, expensive and uncertain
process, and the FDA or other applicable foreign regulator may delay, limit or deny approval of our product candidates for many reasons,
including, among others, because:
| |
● |
we may not be able to demonstrate that our product candidates are safe and effective in treating patients to the satisfaction of the FDA or foreign regulator; |
| |
● |
the results of our clinical trials may not meet the level of statistical or clinical significance required by the FDA or foreign regulator for marketing approval; |
| |
● |
the FDA or foreign regulator may disagree with the number, design, size, conduct or implementation of our clinical trials; |
| |
● |
the FDA or foreign regulator may require that we conduct additional clinical trials; |
| |
● |
the FDA or foreign regulator may not approve the formulation, labeling or specifications of our product candidates; |
| |
● |
the contract research organizations (CROs) and other contractors that we may retain to conduct our clinical trials may take actions outside of our control that materially adversely impact our clinical trials; |
| |
● |
the FDA or foreign regulator may find the data from preclinical studies and clinical trials insufficient to demonstrate that our product candidate(s) are safe and effective for their proposed indications; |
| |
● |
the FDA or foreign regulator may disagree with our interpretation of data from our preclinical studies and clinical trials; |
| |
● |
the FDA or foreign regulator may not accept data generated at our clinical trial sites or may disagree with us over whether to accept efficacy results from clinical trial sites outside the United States or outside the EU, as applicable, where the standard of care is potentially different from that in the United States or in the EU, as applicable; |
| |
● |
if and when our BLAs or foreign equivalents are submitted to the applicable regulatory authorities, such agencies may have difficulties scheduling the necessary review meetings in a timely manner, may recommend against approval of our application or may recommend or require, as a condition of approval, additional preclinical studies or clinical trials, limitations on approved labeling or distribution and use restrictions; |
| |
● |
the FDA or foreign regulator may require development of a Risk Evaluation and Mitigation Strategy (REMS), which would use risk minimization strategies to ensure that the benefits of certain prescription drugs outweigh their risks, as a condition of approval or post-approval; |
| |
● |
the FDA or other applicable foreign regulatory agencies may not approve the manufacturing processes or facilities of third-party manufacturers with which we contract; or |
| |
● |
the FDA or the other applicable foreign regulatory agencies may change their approval policies or adopt new regulations. |
We may encounter substantial delays in completing
our clinical studies which in turn will require additional costs, or we may fail to demonstrate adequate safety and efficacy to the satisfaction
of applicable regulatory authorities.
It is difficult to predict
if or when any of our product candidates, will prove safe or effective in humans or will receive regulatory approval. Before
obtaining marketing approval from regulatory authorities for the sale of our product candidates, we must conduct extensive clinical studies
to demonstrate the safety and efficacy of the product candidates in humans. Clinical testing is expensive, time-consuming, and uncertain
as to outcome. We cannot guarantee that any clinical studies will be conducted as planned or completed on schedule, if at all. A failure
of one or more clinical studies can occur at any stage of testing. Events that may prevent successful or timely completion of clinical
development include:
| |
● |
delays in reaching, or failing to reach, a consensus with regulatory agencies on study design; |
| |
● |
delays in reaching, or failing to reach, agreement on acceptable terms with a sufficient number of prospective contract research organizations (“CROs”) and clinical study sites, the terms of which can be subject to extensive negotiation and may vary significantly among different CROs and trial sites; |
| |
|
|
| |
● |
delays in obtaining required Institutional Review Board (“IRB”) or Ethics Committee (“EC”) approval at each clinical study site; |
| |
|
|
| |
● |
delays in recruiting a sufficient number of suitable patients to participate in our clinical studies including, but not limited to, recruitment challenges due to COVID-19; |
| |
|
|
| |
● |
imposition of a clinical hold by regulatory agencies, after an inspection of our clinical study operations or study sites; |
| |
|
|
| |
● |
failure by our CROs, other third parties or us to adhere to the clinical study, regulatory or legal requirements; |
| |
|
|
| |
● |
failure to perform in accordance with the FDA’s good clinical practices (“GCP”) or applicable regulatory guidelines in other countries; |
| |
|
|
| |
● |
delays in the testing, validation, manufacturing, and delivery of sufficient quantities of our product candidates to the clinical sites; |
| |
|
|
| |
● |
delays in having patients’ complete participation in a study or return for post-treatment follow-up; |
| |
|
|
| |
● |
clinical study sites or patients dropping out of a study; |
| |
|
|
| |
● |
delay or failure to address any patient safety concerns that arise during the course of a trial; |
| |
|
|
| |
● |
unanticipated costs or increases in costs of clinical trials of our product candidates; |
| |
|
|
| |
● |
occurrence of serious adverse events associated with the product candidates that are viewed to outweigh their potential benefits; or |
| |
|
|
| |
● |
changes in regulatory requirements and guidance that require amending or submitting new clinical protocols. |
We could also encounter delays
if a clinical trial is suspended or terminated by us, by the IRBs or Ecs of the institutions in which such trials are being conducted,
by an independent Safety Review Board (“SRB”) for such trial or by the FDA, European Medicines Agency (“EMA”),
or other regulatory authorities. Such authorities may suspend or terminate a clinical trial due to a number of factors, including
failure to conduct the clinical trial in accordance with regulatory requirements or our clinical protocols, inspection of the clinical
trial operations or trial site by the FDA, EMA, or other regulatory authorities resulting in the imposition of a clinical hold, unforeseen
safety issues or adverse side effects, failure to demonstrate a benefit from using a drug, changes in governmental regulations or administrative
actions or lack of adequate funding to continue the clinical trial.
Any inability to successfully
complete preclinical and clinical development could result in additional costs to us or impair our ability to generate revenues from product
sales, regulatory and commercialization milestones, and royalties. In addition, if we make manufacturing or formulation changes to our
product candidates, we may need to conduct additional studies to bridge our modified product candidates to earlier versions.
Clinical study delays could
also shorten any periods during which we may have the exclusive right to commercialize our product candidates or allow our competitors
to bring products to market before we do, which could impair our ability to successfully commercialize our product candidates. In
addition, any delays in completing our clinical trials will increase our costs, slow down our product candidate development
and approval process and jeopardize our ability to commence product sales and generate revenues. Any of these occurrences may significantly
harm our business, financial condition, and prospects. In addition, many of the factors that cause, or lead to, a delay in the commencement
or completion of clinical trials may also ultimately lead to the denial of regulatory approval of our product candidates.
The outcome of preclinical
studies and early clinical trials may not be predictive of the success of later clinical trials, and interim results of a clinical trial
do not necessarily predict final results. Further, preclinical and clinical data are often susceptible to various interpretations and
analyses, and many companies that have believed their product candidates performed satisfactorily in preclinical studies and clinical
trials have, nonetheless, failed to obtain marketing approval. If the results of our clinical studies are inconclusive or if there
are safety concerns or adverse events associated with our other product candidates, we may:
| |
● |
be delayed in obtaining marketing approval for our product candidates, if approved at all; |
| |
|
|
| |
● |
obtain approval for indications or patient populations that are not as broad as intended or desired; |
| |
|
|
| |
● |
obtain approval with labeling that includes significant use or distribution restrictions or safety warnings; |
| |
|
|
| |
● |
be required to change the way the product is administered; |
| |
|
|
| |
● |
be required to perform additional clinical studies to support approval or be subject to additional post-marketing testing requirements; |
| |
|
|
| |
● |
have regulatory authorities withdraw their approval of a product or impose restrictions on its distribution in the form of a modified risk evaluation and mitigation strategy; |
| |
|
|
| |
● |
be sued; or |
| |
|
|
| |
● |
experience damage to our reputation. |
If we, ours collaborators,
or our contract manufacturing organizations (“CMOs”) fail to comply with applicable regulatory requirements at any stage during
the regulatory process, such noncompliance could result in, among other things delays in the approval of applications or supplements to
approved applications; refusal of a regulatory authority, including the FDA, to review pending market approval applications or supplements
to approved applications; warning letters; fines; import and/or export restrictions; product recalls or seizures; injunctions; total or
partial suspension of production; civil penalties; withdrawals of previously approved marketing applications or licenses; recommendations
by the FDA or other regulatory authorities against governmental contracts; and/or criminal prosecutions.
Additionally, our product
candidates could potentially cause other adverse events that have not yet been predicted. The inclusion of ill patients in our clinical
studies may result in deaths or other adverse medical events due to other therapies or medications that such patients may be using. As
described above, any of these events could prevent us from achieving or maintaining market acceptance of our product candidates and impair
our ability to commercialize our products.
We may not be able to meet requirements
for the chemistry, manufacturing, and control of our drug product candidates.
To receive approval of our
products by the FDA and comparable foreign regulatory authorities, we must show that we and our contract manufacturing partners are able
to characterize, control and manufacture our drug products safely and in accordance with regulatory requirements. This includes synthesizing
the active ingredient, developing an acceptable formulation, performing tests to adequately characterize the formulated product, documenting
a repeatable manufacturing process, and demonstrating that our drug products meet stability requirements. Meeting these chemistry, manufacturing
and control (“CMC”) requirements is a complex task that requires specialized expertise. If we are not able to meet the CMC
requirements, we may not be successful in getting our products approved.
Enrollment and
retention of patients in clinical trials is an expensive and time-consuming process and could be made more difficult or rendered impossible
by multiple factors outside our control.
We may encounter delays or
difficulties in enrolling, or be unable to enroll, a sufficient number of patients to complete any of our clinical trials on its current
timelines, or at all, and even once enrolled we may be unable to retain a sufficient number of patients to complete any of our trials.
Enrollment in our clinical trials may be slower than we anticipate, leading to delays in our development timelines. For example, we may
face difficulty enrolling or maintaining a sufficient number of patients in our clinical trials due to the existing alternative treatments
approved for any of our targeted indications as patients may decline to enroll or decide to withdraw from our clinical trials due to the
risk of receiving placebo. Patient enrollment and retention in clinical trials depends on many factors, including the size of the patient
population, the nature of the trial protocol, our ability to recruit clinical trial investigators with the appropriate competencies and
experience, the existing body of safety and efficacy data with respect to the study drug, the number and nature of competing treatments
and ongoing clinical trials of competing drugs for the same indication, the proximity of patients to clinical sites, the eligibility criteria
for the trial and the proportion of patients screened that meets those criteria, our ability to obtain and maintain patient consents,
and our ability to successfully complete prerequisite studies before enrolling certain patient populations.
Furthermore, any negative
results or new safety signals we may report in clinical trials of our product candidates may make it difficult or impossible to recruit
and retain patients in other clinical trials. Similarly, negative results reported by our competitors about their drug candidates may
negatively affect patient recruitment in our clinical trials. Also, marketing authorization of competitors in this same class of drugs
may impair our ability to enroll patients into our clinical trials, delaying or potentially preventing it from completing recruitment
of one or more of our trials.
Delays or failures in planned
patient enrollment or retention may result in increased costs, program delays or both, which could have a harmful effect on our ability
to develop our product candidates or could render further development impossible. In addition, we expect to rely on CROs and clinical
trial sites to ensure proper and timely conduct of our future clinical trials, and, while we intend to enter into agreements governing
their services, we will be limited in our ability to compel their actual performance.
If our future pre-clinical development or
future clinical Phase I/II studies are unsuccessful, we may be unable to obtain regulatory approval of, or commercialize, our product
candidates on a timely basis or at all.
The successful completion
of pre-clinical development and multiple clinical trials is critical to the success of our future products. If the pre-clinical development
and clinical trials are unsuccessful or produce inconsistent results or unanticipated adverse side effects, or if we are unable to collect
reliable data, regulatory approval of our products could be delayed or not given and as a result we may be unable to commercialize our
products. Generally, we expect to engage third parties such as consultants, universities or other collaboration partners to conduct clinical
trials on our behalf. Incompatible practices or misapplication of our products by these third parties could impair the success of our
clinical trials.
Even if we receive regulatory approval for
any of our product candidates, we may not be able to successfully commercialize the product and the revenue that we generate from their
sales, if any, may be limited.
If approved for marketing,
the commercial success of our product candidates will depend upon each product’s acceptance by the medical community, including
physicians, patients, and health care payors. The degree of market acceptance for any of our product candidates will depend on a number
of factors, including:
| |
● |
demonstration of clinical safety and efficacy; |
| |
|
|
| |
● |
relative convenience, dosing burden and ease of administration; |
| |
|
|
| |
● |
the prevalence and severity of any adverse effects; |
| |
|
|
| |
● |
the willingness of physicians to prescribe our product candidates, and the target patient population to try new therapies; |
| |
|
|
| |
● |
efficacy of our product candidates compared to competing products; |
| |
|
|
| |
● |
the introduction of any new products that may in the future become available targeting indications for which our product candidates may be approved; |
| |
|
|
| |
● |
new procedures or therapies that may reduce the incidences of any of the indications in which our product candidates may show utility; |
| |
● |
pricing and cost-effectiveness; |
| |
|
|
| |
● |
the inclusion or omission of our product candidates in applicable therapeutic and vaccine guidelines; |
| |
|
|
| |
● |
the effectiveness of our own or any future collaborators’ sales and marketing strategies; |
| |
|
|
| |
● |
limitations or warnings contained in approved labeling from regulatory authorities; |
| |
|
|
| |
● |
our ability to obtain and maintain sufficient third-party coverage or reimbursement from government health care programs, including Medicare and Medicaid, private health insurers and other third-party payors or to receive the necessary pricing approvals from government bodies regulating the pricing and usage of therapeutics; and |
| |
|
|
| |
● |
the willingness of patients to pay out-of-pocket in the absence of third-party coverage or reimbursement or government pricing approvals. |
If any of our product candidates
are approved, but do not achieve an adequate level of acceptance by physicians, health care payors, and patients, we may not generate
sufficient revenues and we may not be able to achieve or sustain profitability. Our efforts to educate the medical community and third-party
payors on the benefits of our product candidates may require significant resources and may never be successful.
In addition, even if we obtain
regulatory approvals, the timing or scope of any approvals may prohibit or reduce our ability to commercialize our product candidates
successfully. For example, if the approval process takes too long, we may miss market opportunities and give other companies the ability
to develop competing products or establish market dominance. Any regulatory approval we ultimately obtain may be limited or subject to
restrictions or post-approval commitments that render our product candidates not commercially viable. For example, regulatory authorities
may approve any of our product candidates for fewer or more limited indications than we request, may grant approval contingent on the
performance of costly post-marketing clinical trials, or may approve any of our product candidates with a label that does not include
the labeling claims necessary or desirable for the successful commercialization for that indication. Further, the FDA or comparable foreign
regulatory authorities may place conditions on approvals or require risk management plans or a Risk Evaluation and Mitigation Strategy
(“REMS”) to assure the safe use of the drug. If the FDA or applicable foreign regulatory agency concludes a REMS is needed,
the sponsor of the BLA must submit a proposed REMS; the regulatory agencies will not approve the BLA without an approved REMS, if required.
A REMS could include medication guides, physician communication plans, or elements to assure safe use, such as restricted distribution
methods, patient registries and other risk minimization tools. The regulatory agencies may also require a REMS for approved product when
new safety information emerges. Any of these limitations on approval or marketing could restrict the commercial promotion, distribution,
prescription or dispensing of our product candidates. Moreover, product approvals may be withdrawn for non-compliance with regulatory
standards or if problems occur following the initial marketing of the product. Any of the foregoing scenarios could materially harm the
commercial success of our product candidates.
Adverse events involving our products may
lead the FDA or applicable foreign regulatory agency to delay or deny clearance for our products or result in product recalls that could
harm our reputation, business and financial results.
Once a product receives regulatory
clearance or approval, the agency has the authority to require the recall of commercialized products in the event of adverse side effects,
material deficiencies or defects in design or manufacture. The authority to require a recall must be based on a regulatory finding that
there is a reasonable probability that the product would cause serious injury or death. Manufacturers may, under their own initiative,
recall a product if any material deficiency in a product is found. A government-mandated or voluntary recall by us or one of our distributors
could occur because of adverse side effects, impurities or other product contamination, manufacturing errors, design or labeling defects
or other deficiencies and issues. Recalls of any of our products would divert managerial and financial resources and have an adverse effect
on our financial condition and results of operations. The regulatory agencies require that certain classifications of recalls be reported
to them within ten (10) working days after the recall is initiated. Companies are required to maintain certain records of recalls, even
if they are not reportable to the regulatory agency. We may initiate voluntary recalls involving our products in the future that we determine
do not require notification of the regulatory agencies. If the regulatory agency disagrees with our determinations, they could require
us to report those actions as recalls. A future recall announcement could harm our reputation with customers and negatively affect our
sales. In addition, the regulatory agency could take enforcement action for failing to report the recalls when they were conducted.
The in-licensing of technologies and the
successful testing and early development of technologies in the laboratory may not be indicative of future results and may not result
in commercially viable technologies or products. Further, our future products may have to be modified from their originally conceived
versions in order to reach or be successful in the market.
Positive results from laboratory
testing and early developmental successes, may not be predictive of future successful development, commercialization and sales results
and should not be relied upon as evidence that products developed from our technologies will become commercially viable and successful.
Further, the products we plan to develop in the future may have to be significantly modified from their originally conceived versions
in order for us to control costs, compete with similar products, receive market acceptance, meet specific development and commercialization
timeframes, avoid potential infringement of the proprietary rights of others, or otherwise succeed in developing our business and earning
ongoing revenues. This can be a costly and resource draining activity. What appear to be promising technologies when we license them may
not lead to viable technologies or products, or to commercial success.
Complying with numerous regulations pertaining
to our business is an expensive and time-consuming process, and any failure to comply could result in substantial penalties.
We are subject to the Clinical
Laboratory Improvement Amendment of 1988, or CLIA, which is a federal law regulating clinical laboratories that perform testing on specimens
derived from humans for the purpose of providing information for the diagnosis, prevention, or treatment of disease. Our clinical laboratory
is in Richmond, Virginia and must be certified under CLIA for us to perform testing on human specimens. CLIA is intended to ensure the
quality and reliability of clinical laboratories in the United States by mandating specific standards in the areas of personnel qualifications,
administration, and participation in proficiency testing, patient test management, quality control, quality assurance and inspections.
We currently hold a CLIA certificate to perform high-complexity testing. Laboratories performing high complexity testing are required
to meet more stringent requirements than laboratories performing fewer complex tests. CLIA regulations require clinical laboratories like
ours to comply with various operational, personnel, facilities administration, quality, and proficiency testing requirements intended
to ensure that testing services are accurate, reliable, and timely. CLIA certification is a prerequisite for reimbursement eligibility
for services provided to state and federal health care program beneficiaries. CLIA is user-fee funded. Therefore, all costs of administering
the program must be covered by the regulated facilities, including certification and survey costs. To renew this certificate, we are subject
to survey and inspection every two years. Moreover, CLIA inspectors may make periodic inspections of our clinical laboratory outside of
the renewal process. The failure to comply with CLIA requirements can result in enforcement actions, including the revocation, suspension,
or limitation of our CLIA certificate of compliance, as well as a directed plan of correction, state on-site monitoring, civil money penalties,
civil injunctive suit and/or criminal penalties. We must maintain CLIA compliance and certification to be eligible to bill for assays
provided to Medicare beneficiaries. If we were to be found out of compliance with CLIA program requirements and subjected to sanctions,
our business and reputation could be harmed. Even if it were possible for us to bring our laboratory back into compliance, we could incur
significant expenses and potentially lose revenue in doing so.
Additionally, certain states
require laboratory licenses to test specimens from patients in those states or received from ordering physicians in those states. We may
also be subject to regulation in foreign jurisdictions if we seek to expand international distribution of our assays outside the United
States.
If we were to lose our CLIA
certification or state laboratory licenses, whether because of a revocation, suspension or limitation, we would no longer be able to offer
our assays (including our AditxtScore™ platform), which would limit our revenues and harm our business. If we were to lose, or fail
to obtain, a license in any other state where we are required to hold a license, we would not be able to test specimens from those states.
Our AditxtScore™ tests are currently
being offered as a LDTs. Should the FDA disagree that AditxtScore™ tests are LDTs, if our LDTs do not receive the required emergency
use authorizations, or if the FDA’s regulatory approach to LDTs should change in the future, our commercialization strategy may
be adversely affected, which would negatively affect our results of operations and financial condition.
The FDA has historically asserted
its authority to regulate Laboratory Developed Tests (LDTs) as medical devices under the Federal Food, Drug, and Cosmetic Act (the “FDCA”),
but it has generally exercised enforcement discretion regarding LDTs. This means that even though the FDA believes it can impose regulatory
requirements on LDTs, such as requirements to obtain premarket approval, de novo classification, or clearance of LDTs,
it has generally chosen not to enforce those requirements. The FDA has, on occasion, sent warning letters to laboratories offering LDTs
that the agency believed were not eligible for enforcement discretion because of how they were developed, validated, performed or marketed
and consequent risks to the public.
The FDA considers an LDT to
be a test that is developed, validated, and performed within a single laboratory. We are providing AditxtScore™ as a service as
a Laboratory Developed Test (LDT) to assess immunity status to COVID-19. Our AditxtScore™ tests are currently manufactured
in our Mountain View, CA facility and performed in our Richmond, VA facility. If the FDA believes that the AditxtScore™ is not regulated
as an LDT, we may be forced to stop performing AditxtScore™ while we worked to obtain the appropriate FDA authorizations which
could negative affect our business, results of operations and financial condition.
On November 15, 2021, FDA
revised its guidance document titled “Policy for Coronavirus Disease-2019 Tests During the Public Health Emergency (Revised)”
(“FDA COVID-19 Testing Guidance”) to require all COVID-19 diagnostic assays conducted as LDTs to apply for EUA authorization
within a 60-day period from the revised guidance’s issuance date. The FDA COVID-19 Testing Guidance states that FDA does not
intend to object to continued offering of LDTs that are the subject of submitted EUA requests while FDA reviews the EUA requests. The
FDA COVID-19 Testing Guidance further states that if FDA declines to issue an EUA or otherwise decides not to authorize a test for any
reason, including a determination that there is a lack of adequate data to support authorization, FDA generally expects developers to
cease marketing and offering their test within 15 calendar days. Moreover, if FDA identifies a significant problem or concern with a test,
based either on the provided information or external reports, FDA generally expects the developer to take appropriate steps to address
such problems, which could include a recall of the test and/or notification concerning corrected test reports indicating prior test results
may not be accurate. We have submitted EUA requests for our SARS-CoV-2 LDTs, and the applications are pending before FDA. There
can be no assurance that the EUA requests that we submitted for our SARS-CoV-2 LDTs will be granted on a timely basis or at
all. If FDA declines to issue a EUAs for our SARS-CoV-2 LDTs, we may be required to cease marketing the tests and our business, results
of operations and financial condition could be negatively affected. Regardless of if our EUA applications are granted by FDA, we may recall,
replace, or make corrections to our LDTs if we become aware of a product concern, which could negatively impact manufacturing, supply,
and customer relationships, and may result in adverse regulatory action, including revision or revocation of an EUA.
In addition, there have been
numerous legislative proposals to clarify the FDA’s regulatory authority over medical devices. These include two bills reintroduced
in 2021: the VALID Act, which would expressly grant the FDA authority to regulate LDTs under a risk-based framework; and the VITAL Act,
which would assign LDTs to regulation solely under CLIA and would direct CMS to update its CLIA regulations. We cannot predict if either
of these bills will be enacted in their current (or any other) form and cannot quantify the effect of these bills on our business. In
the meantime, the regulation by the FDA of LDTs remains uncertain. If FDA premarket review, classification or approval is required for
AditxtScore™, our laboratory could be forced to stop performing AditxtScore™ while we worked to obtain the appropriate
FDA authorizations which could negative affect our business, results of operations and financial condition.
We are subject
to various governmental regulations relating to the labeling, marketing and sale of our products.
Both before and after a product
is commercially released, we have ongoing responsibilities under regulations promulgated by the FDA, the Federal Trade Commission, and
similar U.S. and foreign regulations governing product labeling and advertising, distribution, sale, and marketing of our products.
Manufacturers of medical devices
are permitted to promote products solely for the uses and indications set forth in the device’s authorization. A number of enforcement
actions have been taken against manufacturers that promote products for “off-label” uses (i.e., uses that are not
described in the device’s authorization), including actions alleging that claims submitted to government healthcare programs for
reimbursement of products that were promoted for “off-label” uses are fraudulent in violation of the Federal False
Claims Act or other federal and state statutes and that the submission of those claims was caused by off-label promotion. The
failure to comply with prohibitions on “off-label” promotion can result in significant monetary penalties, revocation
or suspension of a company’s business license, suspension of sales of certain products, product recalls, civil or criminal sanctions,
exclusion from participating in federal healthcare programs, or other enforcement actions. In the United States, allegations of such wrongful
conduct could also result in a corporate integrity agreement with the U.S. government that imposes significant administrative obligations
and costs.
We and our employees and contractors are
subject, directly, or indirectly, to federal, state and foreign healthcare fraud and abuse laws, including false claims laws. If we are
unable to comply, or have not fully complied, with such laws, we could face substantial penalties.
Our operations are subject
to various federal, state, and foreign fraud and abuse laws. These laws may constrain our operations, including the financial arrangements
and relationships through which we market, sell and distribute our products.
U.S. federal and state laws
that affect our ability to operate include, but are not limited to:
| |
● |
the federal Anti-Kickback Statute, which prohibits, among other things, persons or entities from knowingly and willfully soliciting, receiving, offering or paying any remuneration (including any kickback, bribe, or rebate), directly or indirectly, overtly or covertly, in cash or in kind in return for, the purchase, recommendation, leasing or furnishing of an item or service reimbursable under a federal healthcare program, such as the Medicare and Medicaid programs; |
| |
● |
federal physician self-referral law, which prohibits a physician from referring a patient to an entity with which the physician (or an immediate family member) has a financial relationship, for the furnishing of certain designated health services for which payment may be made by Medicare or Medicaid, unless an exception applies; |
| |
● |
federal civil and criminal false claims laws and civil monetary penalty laws, which prohibit, among other things, individuals, or entities from knowingly presenting, or causing to be presented, claims for payment or approval from Medicare, Medicaid, or other government payers that are false or fraudulent; |
| |
● |
Section 242 of HIPAA codified at 18 U.S.C. § 1347, which created new federal criminal statutes that prohibit a person from knowingly and willfully executing a scheme or from making false or fraudulent statements to defraud any healthcare benefit program (i.e., public or private); |
| |
● |
federal transparency laws, including the Physician Payments Sunshine Act which requires the tracking and disclosure to the federal government by pharmaceutical and medical device manufacturers of payments and other transfers of value to physicians and teaching hospitals as well as ownership and investment interests that are held by physicians and their immediate family members; and |
| |
● |
state law equivalents of each of these federal laws, such as anti-kickback and false claims laws that may apply to items or services reimbursed by any third-party payer, including commercial insurers; state laws that require pharmaceutical and medical device companies to comply with their industry’s voluntary compliance guidelines and the applicable compliance guidance promulgated by the federal government or otherwise restrict certain payments that may be made to healthcare providers and other potential referral sources; state laws that require drug and medical device manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures; state laws that prohibit giving gifts to licensed healthcare professionals; and state laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts in certain circumstances, such as specific disease states. |
In particular, activities
and arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, waste and other abusive
practices. These laws and regulations may restrict or prohibit a wide range of activities or other arrangements related to the development,
marketing or promotion of products, including pricing and discounting of products, provision of customer incentives, provision of reimbursement
support, other customer support services, provision of sales commissions or other incentives to employees and independent contractors
and other interactions with healthcare practitioners, other healthcare providers and patients.
Because of the breadth of
these laws and the narrow scope of the statutory or regulatory exceptions and safe harbors available, our business activities could be
challenged under one or more of these laws.
Government expectations and
industry best practices for compliance continue to evolve and past activities may not always be consistent with current industry best
practices. Further, there is a lack of government guidance as to whether various industry practices comply with these laws, and government
interpretations of these laws continue to evolve, all of which create compliance uncertainties. Any non-compliance could result in regulatory
sanctions, criminal or civil liability and serious harm to our reputation. It is not always possible to identify and deter misconduct
concerning applicable laws, regulations, guidelines, policies and standards, and the precautions we take to detect and prevent this activity
may not be effective in preventing such conduct, mitigating risks, or reducing the chance of governmental investigations or other actions
or lawsuits stemming from a failure to comply with these laws or regulations.
If a government entity opens
an investigation into possible violations of any of these laws (which may include the issuance of subpoenas or civil investigative demands),
we would have to expend significant resources to defend ourselves against the allegations. Allegations that we, our officers, or our employees
violated any one of these laws can be made by individuals called “whistleblowers” who may be our employees, customers, competitors
or other parties. Government policy is to encourage individuals to become whistleblowers and file a complaint in federal court alleging
wrongful conduct. The government is required to investigate all of these complaints and decide whether to intervene. If the government
intervenes and we are required to pay money back to the government, the whistleblower, as a reward, is awarded a percentage of the collection.
If the government declines to intervene, the whistleblower may proceed on their own and, if they are successful, they will receive a percentage
of any judgment or settlement amount the company is required to pay. The government may also initiate an investigation on its own. Such
actions could have a significant impact on our business, including the imposition of significant fines, and other sanctions that may materially
impair our ability to run a profitable business. In particular, if our operations are found to be in violation of any of the laws described
above or if we agree to settle with the government without admitting to any wrongful conduct or if we are found to be in violation of
any other governmental regulations that apply to us, we, our officers and employees may be subject to sanctions, including civil and criminal
penalties, damages, fines, exclusion from participation in government health care programs, such as Medicare and Medicaid, imprisonment,
the curtailment or restructuring of our operations and the imposition of a corporate integrity agreement, any of which could adversely
affect our business, results of operations and financial condition.
Risks Related to our Company and our Business
Our technology is subject to licenses from
LLU and Stanford, each of which are revocable in certain circumstances, including in the event we do not achieve certain payments and
milestone deadlines. Without these licenses, we may not be able to continue to develop our product candidates.
The LLU License Agreement
may be terminated by LLU in the event of a breach by us of any non-payment provision (including the provision that requires us to meet
certain deadlines for milestone events (each, a “Milestone Deadline”)) not cured within 90 days after delivery of written
notice by LLU. Additional Milestone Deadlines include: (i) the requirement to have regulatory approval of an IND application to initiate
first-in-human clinical trials on or before March 31, 2022, (ii) the completion of first-in-human (phase I/II) clinical trials by March
31, 2024, (iii) the completion of Phase III clinical trials by March 31, 2026 and (iv) biologic licensing approval (BLA) by the FDA by
March 31, 2027. If the LLU License Agreement were to be terminated by LLU, we would lose our most significant asset and may no longer
be able to develop our product candidates, which would have a material adverse effect on our operations.
The February 2020 License
Agreement with Stanford may be terminated by Stanford if we (i) are delinquent on any report or payments; (ii) are not diligently developing
and commercializing Licensed Product (as defined in the February 2020 License Agreement); (iii) miss a milestone described in the agreement;
(iv) are in breach of any other provision of the agreement; or (v) if we provide a false report to Stanford. The Termination discussed
above will take effect only upon 30 days written notice by Stanford unless we remedy the breach within a 30-day cure period. If the February
2020 License Agreement were to be terminated by Stanford, we would lose a significant asset and may no longer be able to develop our product
candidates, which would have a material adverse effect on our operations.
Our results of operations will be affected
by the level of royalty and milestone payments that we are required to pay to third parties.
The LLU License Agreement
and February 2020 License Agreement with Stanford each require us to remit royalty payments and meet certain performance milestones related
to in-licensed intellectual property. Any failure on our part to pay royalties owed or meet milestones could lead to us losing rights
under our licenses and could thereby adversely affect our business. As our product sales increase, we may, from time-to-time, disagree
with our third-party collaborators as to the appropriate royalties owed and the resolution of such disputes may be costly and may consume
management’s time. Furthermore, we may enter into additional license agreements in the future, which may also include royalty payments.
We face substantial competition, which may
result in others discovering, developing or commercializing products before or more successfully than we do.
The development and commercialization
of drugs is highly competitive. We compete with a variety of multinational pharmaceutical companies and specialized biotechnology companies,
as well as products and processes being developed at universities and other research institutions. Our competitors have developed, are
developing or will develop product candidates and processes competitive with our product candidates. Competitive therapeutic treatments
include those that have already been approved and accepted by the medical community and any new treatments that may enter the market.
We believe that a significant number of products are currently available, under development, and may become commercially available in
the future, for the treatment of indications for which we may try to develop product candidates.
More established companies
may have a competitive advantage over us due to their greater size, cash flows and institutional experience. Compared to us, many of our
competitors may have significantly greater financial, technical and human resources. As a result of these factors, our competitors may
have an advantage in marketing their approved products and may obtain regulatory approval of their product candidates before we are able
to, which may limit our ability to develop or commercialize our product candidates. Our competitors may also develop drugs that are safer,
more effective, more widely used and less expensive than ours, and may also be more successful than us in manufacturing and marketing
their products.
Mergers and acquisitions in
the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of our competitors.
Smaller and other early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements
with large and established companies. These companies compete with us in recruiting and retaining qualified scientific, management and
commercial personnel, establishing clinical trial sites and subject registration for clinical trials, as well as in acquiring technologies
complementary to, or necessary for, our programs.
Our technologies and products under development,
and our business, may fail if we are not able to successfully commercialize them and ultimately generate significant revenues as a result.
Successful development of
technologies and our product candidates will require significant additional investment, including costs associated with additional development,
completing trials and obtaining regulatory approval, as well as the ability to manufacture or have others manufacture our products in
sufficient quantities at acceptable costs while also preserving product quality. Difficulties often encountered in scaling up production
include problems involving production yields, quality control and assurance, shortage of qualified personnel, production costs and process
controls. In addition, we are subject to inherent risks associated with new technologies and products. These risks include the possibility
that any of our technologies or future products may:
| |
● |
be ineffective or less effective than anticipated; |
| |
● |
fail to receive necessary regulatory approvals; |
| |
● |
be difficult to competitively price relative to alternative solutions; |
| |
● |
be harmful to consumers or the environment; |
| |
● |
be difficult to manufacture on an economically viable scale; |
| |
● |
be subject to supply chain constraints for raw materials; |
| |
● |
fail to be developed and accepted by the market prior to the successful marketing of alternative products by competitors; |
| |
● |
be difficult to market because of infringement on the proprietary rights of third parties; or |
| |
● |
be too expensive for commercial use. |
Furthermore, we may be faced
with lengthy market partner or distributor evaluation and approval processes. Consequently, we may incur substantial expenses and devote
significant management effort to customize products for market partner or distributor acceptance, though there can be no assurance of
such acceptance. As a result, we cannot accurately predict the volume or timing of any future sales.
Customers may not adopt our products quickly,
or at all.
Customers in the sector in
which we operate can be generally cautious in their adoption of new products and technologies. In addition, given the relative novelty
of our future planned products (including our AditxtScore™ platform), customers of those products may require education regarding
their utility and use, which may delay their adoption. There can be no assurance that customers will adopt our products quickly, or at
all.
The significant level of competition in
the markets for our products developed in the future may result in pricing pressure, reduced margins or the inability of our future products
to achieve market acceptance.
The markets for our future
products are intensely competitive and rapidly changing. We may be unable to compete successfully, which may result in price reductions,
reduced margins and the inability to achieve market acceptance for our products.
Our competitors may have longer
operating histories, significantly greater resources, greater brand recognition and large customer bases than we do. As a result, they
may be able to devote greater resources to the manufacture, promotion or sale of their products, receive greater resources and support
from market partners and independent distributors, initiate or withstand substantial price competition or more readily take advantage
of acquisition or other opportunities.
We rely on third parties for the distribution
of our current and future products, including our AditxtScore™ platform. If these parties do not distribute our products in
a satisfactory or timely manner, in sufficient quantities or at an acceptable cost, our sales and development efforts could be delayed
or otherwise negatively affected.
We rely on third parties for
the distribution of our current and future products, including our AditxtScore™ platform. Our reliance on third parties to distribute
products may present significant risks to us, including the risk that should any of these third parties fail to adequately distribute
our products and services to end consumers and other market participants, our business may be materially harmed. Additionally, if we need
to enter into agreements for the distribution of our future products with other third parties, there can be no assurance we will be able
to do so on favorable terms, if at all.
We may rely on third parties to produce
our future products. If these parties do not produce our products at a satisfactory quality, in a timely manner, in sufficient quantities
or at an acceptable cost, our sales and development efforts could be delayed or otherwise negatively affected.
We may rely on third parties
for the manufacture of our future products. Our reliance on third parties to manufacture our future products may present significant risks
to us, including the following:
| |
● |
reduced control over delivery schedules, yields and product reliability; |
| |
● |
manufacturing deviations from internal and regulatory specifications; |
| |
● |
the failure of a key manufacturer to perform as we require for technical, market or other reasons; |
| |
● |
difficulties in establishing additional manufacturer relationships if we are presented with the need to transfer our manufacturing process technologies to them; |
| |
● |
misappropriation of our intellectual property; and |
| |
● |
other risks in potentially meeting our product development schedule or satisfying the requirements of our market partners, distributors, direct customers and end users. |
If we need to enter into agreements
for the manufacturing of our future products, there can be no assurance we will be able to do so on favorable terms, if at all.
If we are unable to establish successful
relations with third-party market partners or distributors, or these market partners or distributors do not focus adequate resources on
selling our products or are otherwise unsuccessful in selling them, sales of our products may not develop.
We anticipate relying on independent
market partners and distributors to distribute and assist us with the marketing and sale of our products. Our future revenue generation
and growth will depend in large part on our success in establishing and maintaining this sales and distribution channel. If our market
partners and distributors are unable to sell our products, or receive negative feedback from end users, they may not continue to purchase
or market our products. In addition, there can be no assurance that our market partners and distributors will focus adequate resources
on selling our products to end users or will be successful in selling them. Many of our potential market partners and distributors are
in the business of distributing and sometimes manufacturing other, possibly competing, products. As a result, these market partners
and distributors may perceive our products as a threat to various product lines currently being distributed or manufactured by them. In
addition, these market partners and distributors may earn higher margins by selling competing products or combinations of competing products.
If we are unable to establish successful relationships with independent market partners and distributors, we will need to further develop
our own sales and distribution capabilities, which would be expensive and time-consuming and might not be successful.
If we are not able to attract and retain
highly skilled employees and contractors, we may not be able to implement our business model successfully.
We will rely upon employees
and third-party consultant/contractors to effectively establish, manage and grow our business. Consequently, we believe that our future
viability will depend largely on our ability to attract and retain highly skilled personnel. In order to do so, we may need to pay
higher compensation, fees, and/or other incentives to our employees or consultants than we currently expect, and such higher compensation
payments would have a negative effect on our operating results. Competition for experienced, high-quality employees, consultants and contractors
is intense and we cannot assure that we will be able to recruit and retain such personnel. We may not be able to hire or retain the necessary
personnel to implement our business strategy. Our failure to hire and retain such personnel could impair our ability to develop new products
and manage our business effectively.
The loss of our management team or other
key personnel would have an adverse impact on our future development and impair our ability to succeed.
In the early stages of development,
our business will be significantly dependent on the Company’s management team and other key personnel. Our success will be particularly
dependent upon Mr. Amro Albanna and Dr. Shahrokh Shabahang. The loss of any one of these individuals or any other future key personnel
could have a material adverse effect on the Company and our ability to further execute our intended business.
The use of our products may be limited by
regulations, and we may be exposed to product liability and remediation claims.
The use of our planned products
may be regulated by various local, state, federal and foreign regulators. Even if we are able to comply with all such regulations
and obtain all necessary registrations, we cannot provide assurance that our future products will not cause injury to the environment,
people, or animals and/or otherwise have unintended adverse consequences, under all circumstances. For example, our products may be improperly
combined with other chemicals or, even when properly combined, our products may be blamed for damage caused by those other chemicals.
The costs of remediation or products liability could materially adversely affect our results, financial condition and operations.
We may be held liable for, or incur costs
to settle, liability and remediation claims if any products we develop, or any products that use or incorporate any of our technologies,
cause injury or are found unsuitable during product testing, manufacturing, marketing, sale or use. These risks exist even with respect
to products that have received, or may in the future receive, regulatory approval, registration or clearance for commercial use. We cannot
guarantee that we will be able to avoid product liability exposure.
At the stage customary to
do so, we expect to maintain product liability insurance at levels we believe are sufficient and consistent with industry standards for
like companies and products. However, we cannot guarantee that our product liability insurance will be sufficient to help us avoid product
liability-related losses. In the future, it is possible that meaningful insurance coverage may not be available on commercially reasonable
terms or at all. In addition, a product liability claim could result in liability to us greater than our assets or insurance coverage.
Moreover, even if we have adequate insurance coverage, product liability claims or recalls could result in negative publicity or force
us to devote significant time and attention to these matters, which could harm our business.
There may be limitations on the effectiveness
of our internal controls, and a failure of our control systems to prevent error or fraud may materially harm our Company.
We do not expect that internal
control over financial accounting and disclosure, even if timely and well established, will prevent all error and all fraud. A control
system, no matter how well designed and operated, can provide only reasonable, not absolute, assurance that the control system’s
objectives will be met. Further, the design of a control system must reflect the fact that there are resource constraints, and the benefits
of controls must be considered relative to their costs. Because of the inherent limitations in all control systems, no evaluation of controls
can provide absolute assurance that all control issues and instances of fraud, if any, have been detected. Failure of our control systems
to prevent error or fraud could materially adversely affect our business.
COVID-19 may impact our operations.
On January 30, 2020, the World
Health Organization declared the COVID-19 coronavirus outbreak a “Public Health Emergency of International Concern” and on
March 10, 2020, declared it to be a pandemic. Actions taken around the world to help mitigate the spread of the coronavirus include restrictions
on travel, and quarantines in certain areas, and forced closures for certain types of public places and businesses. The COVID-19 coronavirus
and actions taken to mitigate it have had and are expected to continue to have an adverse impact on the economies and financial markets
of many countries, including the geographical area in which the Company operates. While it is unknown how long these conditions will last
and what the complete financial effect will be to the Company, capital raise efforts and additional development of our technologies may
be negatively affected.
Risks Related to Our Acquisition Strategy
Our acquisition strategy exposes us to substantial
risk.
Our acquisition of companies
is subject to substantial risk, including but not limited to the failure to identify material problems during due diligence (for which
we may not be indemnified post-closing), the risk of over-paying for assets (or not making acquisitions on an accretive basis), the ability
to obtain or retain customers and the risks of entering markets where we have limited experience. While we perform due diligence on prospective
acquisitions, we may not be able to discover all potential operational deficiencies in such entities.
Our
acquisition targets may not perform as expected or the returns from such businesses may not support the financing utilized to acquire
them or maintain them. Furthermore, integration and consolidation of acquired businesses requires substantial human, financial and other
resources and may divert management’s attention from our existing business concerns, disrupt our ongoing business or not be successfully
integrated. Even if we consummate businesses that we believe will be accretive, those businesses may in fact result in a decrease in revenues
as a result of incorrect assumptions in our evaluation of such businesses, unforeseen consequences, or other external events beyond our
control. Furthermore, if we consummate any future acquisitions, our capitalization and results of operations may change significantly,
and stockholders will generally not have the opportunity to evaluate the economic, financial, and other relevant information that we will
consider in determining the application of these funds and other resources. As a result, the consummation of acquisitions may have a material
adverse effect on our business, financial condition, results of operations and cash flows.
We may experience difficulty as we evaluate,
acquire and integrate businesses that we may acquire, which could result in drains on our resources, including the attention of our management,
and disruptions of our on-going business.
We acquire small to mid-sized
businesses in various industry segments. Generally, because such businesses are privately held, we may experience difficulty in evaluating
potential target businesses as much of the information concerning these businesses is not publicly available. Therefore, our estimates
and assumptions used to evaluate the operations, management and market risks with respect to potential target businesses may be subject
to various risks and uncertainties. Further, the time and costs associated with identifying and evaluating potential target businesses
may cause a substantial drain on our resources and may divert our management team’s attention away from the operations of our businesses
for significant periods of time.
In addition, we may have difficulty
effectively integrating and managing acquisitions. The management or improvement of businesses we acquire may be hindered by a number
of factors, including limitations in the standards, controls, procedures and policies implemented in connection with such acquisitions.
Further, the management of an acquired business may involve a substantial reorganization of the business’ operations resulting in
the loss of employees and customers or the disruption of our ongoing businesses. We may experience greater than expected costs or difficulties
relating to an acquisition, in which case, we might not achieve the anticipated returns from any particular acquisition.
We may not be able to effectively integrate
the businesses that we acquire.
Our ability to realize the
anticipated benefits of acquisitions will depend on our ability to integrate those businesses with our own. The combination of multiple
independent businesses is a complex, costly and time-consuming process and there can be no assurance that we will be able to successfully
integrate businesses into our business, or if such integration is successfully accomplished, that such integration will not be costlier
or take longer than presently contemplated. Integration of future acquisitions may include various risks and uncertainties, including
the factors discussed in the paragraph below. If we cannot successfully integrate and manage the businesses within a reasonable time,
we may not be able to realize the potential and anticipated benefits of such acquisitions, which could have a material adverse effect
on our stock price, business, cash flows, results of operations and financial position.
We will consider acquisitions
that we believe will complement, strengthen and enhance our growth. We evaluate opportunities on a preliminary basis from time to time,
but these transactions may not advance beyond the preliminary stages or be completed. Such acquisitions are subject to various risks and
uncertainties, including:
| |
● |
the inability to integrate effectively the operations, products, technologies and personnel of the acquired companies (some of which are in diverse geographic regions) and achieve expected synergies; |
| |
● |
the potential disruption of existing business and diversion of management’s attention from day-to-day operations; |
| |
● |
the inability to maintain uniform standards, controls, procedures and policies; |
| |
● |
the need or obligation to divest portions of the acquired companies; |
| |
● |
the potential failure to identify material problems and liabilities during due diligence review of acquisition targets; |
| |
● |
the potential failure to obtain sufficient indemnification rights to fully offset possible liabilities associated with acquired businesses; and |
| |
● |
the challenges associated with operating in new geographic regions. |
The integration
of our acquisitions may result in significant accounting charges that adversely affect the announced results of our Company.
The financial results of our
Company may be adversely affected by cash expenses and non-cash accounting charges incurred in connection with our recent acquisitions.
In addition to the anticipated cash charges, costs associated with the amortization of intangible assets are expected. The price of our
common stock could decline to the extent our financial results are materially affected by the foregoing charges or if the foregoing charges
are larger than anticipated.
Our planned acquisitions
may result in unexpected consequences to our business and results of operations.
Although we believe that our
planned acquisitions will generally be subject to risks similar to those to which we are subject to in our existing operations, we may
not have discovered all risks applicable to these businesses during the due diligence process. Some of these risks could produce unexpected
and unwanted consequences for us. Undiscovered risks may result in us incurring financial liabilities, which could be material and have
a negative impact on our business operations.
Failure to manage our growing and changing
business could have a material adverse effect on our business, prospects, financial condition, and results of operations.
As we grow, we expect to encounter
additional challenges to our internal processes, capital commitment process, and acquisition funding and financing capabilities. Our existing
operations, personnel, systems, and internal control may not be adequate to support our growth and expansion and may require us to make
additional unanticipated investments in our infrastructure. To manage the future growth of our operations, we will be required to improve
our administrative, operational, and financial systems, procedures, and controls, and maintain, expand, train, and manage our growing
employee base. If we are unable to manage our growth effectively, we may not be able to take advantage of market opportunities, execute
our business strategies successfully or respond to competitive pressures. As a result, our business, prospects, financial condition, and
results of operations could be materially and adversely affected.
We face competition for businesses that
fit our acquisition strategy and, therefore, we may have to acquire targets at sub-optimal prices or, alternatively, forego certain acquisition
opportunities.
Our acquisition strategy is
focused on the acquisition of small to mid-sized businesses. In pursuing such acquisitions, we expect to face strong competition from
a wide range of other potential purchasers. Although the pool of potential purchasers for such businesses is typically smaller than for
larger businesses, those potential purchasers can be aggressive in their approach to acquiring such businesses. Furthermore, we expect
that we will need to use third-party financing in order to fund some or all of these potential acquisitions, thereby increasing our acquisition
costs. To the extent that other potential purchasers do not need to obtain third-party financing or are able to obtain such financing
on more favorable terms, they may be in a position to be more aggressive with their acquisition proposals. As a result, in order to be
competitive, our acquisition proposals may need to be aggressively priced, including at price levels that exceed what we originally determined
to be fair or appropriate. Alternatively, we may determine that we cannot pursue on a cost-effective basis what would otherwise be an
attractive acquisition opportunity.
We may not be able to successfully fund
acquisitions due to the unavailability of equity or debt financing on acceptable terms, which could impede the implementation of our acquisition
strategy.
We intend to finance acquisitions
primarily through additional debt and equity financings. Because the timing and size of acquisitions cannot be readily predicted, we may
need to be able to obtain funding on short notice to benefit fully from attractive acquisition opportunities. The sale of additional shares
of any class of equity will be subject to market conditions and investor demand for such shares at prices that may not be in the best
interest of our stockholders. The sale of additional equity securities could also result in dilution to our stockholders. The incurrence
of indebtedness would result in increased debt service obligations and could require us to agree to operating and financial covenants
that would restrict our operations. Financing may not be available in amounts or on terms acceptable to us, if at all. These risks may
materially adversely affect our ability to pursue our acquisition strategy.
We may change our management and acquisition
strategies without the consent of our stockholders, which may result in a determination by us to pursue riskier business activities.
We may change our strategy
at any time without the consent of our stockholders, which may result in our acquiring businesses or assets that are different from, and
possibly riskier than, the strategy described in this prospectus. A change in our strategy may increase our exposure to interest rate
and currency fluctuations, subject us to regulation under the Investment Company Act or subject us to other risks and uncertainties
that affect our operations and profitability.
In the future, we may seek to enter into
credit facilities to help fund our acquisition capital and working capital needs. These credit facilities may expose us to additional
risks associated with leverage and may inhibit our operating flexibility.
We may seek to enter into
credit facilities with third-party lenders to help fund our acquisitions. Such credit facilities will likely require us to pay a commitment
fee on the undrawn amount and will likely contain a number of affirmative and restrictive covenants. If we violate any such covenants,
our lenders could accelerate the maturity of any debt outstanding. Such debt may be secured by our assets, including the stock we may
own in businesses that we acquire. Our ability to meet our debt service obligations may be affected by events beyond our control and will
depend primarily upon cash produced by businesses that we currently manage and may acquire in the future and distributed or paid to us.
Any failure to comply with the terms of our indebtedness may have a material adverse effect on our financial condition.
In addition, we expect that
such credit facilities will bear interest at floating rates which will generally change as interest rates change. We will bear the risk
that the rates that we are charged by our lenders will increase faster than we can grow the cash flow from our businesses or businesses
that we may acquire in the future, which could reduce profitability, materially adversely affect our ability to service our debt, cause
us to breach covenants contained in our third-party credit facilities and reduce cash flow available for distribution.
If, in the future, we cease to control and
operate our businesses or other businesses that we acquire in the future or engage in certain other activities, we may be deemed to be
an investment company under the Investment Company Act.
We have the ability to make
investments in businesses that we will not operate or control. If we make significant investments in businesses that we do not operate
or control, or that we cease to operate or control, or if we commence certain investment-related activities, we may be deemed to be an
investment company under the Investment Company Act. Our decision to sell a business will be based upon financial, operating and
other considerations rather than a plan to complete a sale of a business within any specific time frame. If we were deemed to be an investment
company, we would either have to register as an investment company under the Investment Company Act, obtain exemptive relief from
the Securities and Exchange Commission, or the SEC, or modify our investments or organizational structure or our contract rights to fall
outside the definition of an investment company. Registering as an investment company could, among other things, materially adversely
affect our financial condition, business and results of operations, materially limit our ability to borrow funds or engage in other transactions
involving leverage and require us to add directors who are independent of us and otherwise will subject us to additional regulation that
will be costly and time-consuming.
If intangible assets
and goodwill that we recorded in connection with our acquisitions become impaired, we may have to take significant charges against earnings.
In
connection with the accounting for our completed acquisitions, we may be required to record a significant amount of intangible assets,
including developed technology, in-process research and development, and customer relationships relating to the acquired product lines,
and goodwill. Under generally accepted accounting principles in the United States, we must assess, at least annually and potentially more
frequently, whether the value of indefinite-lived intangible assets and goodwill have been impaired. Intangible assets and goodwill are
assessed for impairment in the event of an impairment indicator. Any reduction or impairment of the value of intangible assets and goodwill
will result in a charge against earnings, which could materially adversely affect our results of operations and shareholders’ equity
in future periods.
Risks Relating to Our Intellectual Property Rights
The failure to obtain or maintain patents,
licensing agreements and other intellectual property could materially impact our ability to compete effectively.
In order for our business
to be viable and to compete effectively, we need to develop and maintain, and we will heavily rely on, a proprietary position with respect
to our technologies and intellectual property. However, there are significant risks associated with our actual or proposed intellectual
property. The risks and uncertainties that we face with respect to our rights principally include the following:
| |
● |
pending patent applications, we have filed or will file may not result in issued patents or may take longer than we expect to result in issued patents; |
| |
● |
we may be subject to interference proceedings; |
| |
● |
we may be subject to reexamination proceedings; |
| |
● |
we may be subject to post grant review proceedings; |
| |
● |
we may be subject to inter partes review proceedings; |
| |
● |
we may be subject to derivation proceedings; |
| |
● |
we may be subject to opposition proceedings in the U.S. or in foreign countries; |
| |
● |
any patents that are issued to us may not provide meaningful protection; |
| |
● |
we may not be able to develop additional proprietary technologies that are patentable; |
| |
● |
other companies may challenge patents licensed or issued to us; |
| |
● |
other companies may have independently developed and patented (or may in the future independently develop and patent) similar or alternative technologies, or duplicate our technologies; |
| |
● |
other companies may design around technologies we have licensed or developed; |
| |
● |
enforcement of patents is complex, uncertain, and very expensive and we may not be able to secure, enforce and defend our patents; and |
| |
● |
if we were to ever seek to enforce our patents in ligation, there is some risk that they could be deemed invalid, not infringed, or unenforceable. |
We cannot be certain that
any patents will be issued because of any pending or future applications, or that any patents, once issued, will provide us with adequate
protection from competing products. For example, issued patents may be circumvented or challenged, declared invalid or unenforceable,
or narrowed in scope. In addition, since publication of discoveries in scientific or patent literature often lags actual discoveries,
we cannot be certain that we or our licensors were the first to invent or to file patent applications covering them.
It is also possible that others
may have or may obtain issued patents that could prevent us from commercializing our products or require us to obtain licenses requiring
the payment of significant fees or royalties to enable us to conduct our business. There is no guarantee that such licenses will be available
based on commercially reasonable terms. As to those patents that we have licensed, our rights depend on maintaining our obligations to
the licensor under the applicable license agreement, and we may be unable to do so.
If we are unable to obtain and maintain
patent protection for our products, or if the scope of the patent protection obtained is not sufficiently broad, competitors could develop
and commercialize products similar or identical to ours, and our ability to successfully commercialize our products could be impaired.
The patent prosecution process
is expensive and time-consuming, and we may not be able to file and prosecute all necessary or desirable patent applications at a reasonable
cost, in a timely manner, or in all jurisdictions. It is also possible that we will fail to identify patentable aspects of our development
output before it is too late to obtain patent protection.
The patent position of life
science companies generally is highly uncertain, involves complex legal and factual questions and has in past years been the subject of
much litigation. In addition, the laws of foreign countries may not protect our rights to the same extent as the laws of the United States
and we may fail to seek or obtain patent protection in all major markets. For example, unlike the U.S., European patent law restricts
the patentability of methods of treatment of the human body. Our pending and future patent applications may not result in patents being
issued which protect our technology or products, in whole or in part, or which effectively prevent others from commercializing competitive
technologies and products. Changes in either the patent laws or interpretation of the patent laws in the United States and other countries
may diminish the value of our patents or narrow the scope of our patent protection, even post-grant.
Recent patent reform legislation
has increased the uncertainties and costs surrounding the prosecution of patent applications and the enforcement or defense of issued
patents. On September 16, 2011, the Leahy-Smith America Invents Act, or the Leahy-Smith Act, was signed into law. The Leahy-Smith Act
includes a number of significant changes to United States patent law. These include provisions that affect the way patent applications
are prosecuted and may also affect patent litigation. The U.S. Patent and Trademark Office, or USPTO, recently developed new regulations
and procedures to govern administration of the Leahy-Smith Act, and many of the substantive changes to patent law associated with the
Leahy-Smith Act, and in particular, the first to file provisions, only became effective on March 16, 2013. Accordingly, it is not clear
what, if any, impact the Leahy-Smith Act will have on the operation of our business. However, the Leahy-Smith Act and its implementation
could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our
issued patents, all of which could have a material adverse effect on our business and financial condition.
Moreover, we may be subject
to a third-party pre-issuance submission of prior art to the USPTO, or become involved in opposition, derivation, reexamination, inter
partes review, post-grant review or interference proceedings challenging our patent rights (whether licensed or otherwise held)
or the patent rights of others. An adverse determination in any such submission, proceeding or litigation could reduce the scope of, or
invalidate, our patent rights (whether licensed or otherwise held), allow third parties to commercialize our technology or products and
compete directly with us, without payment to us, or result in our inability to manufacture or commercialize products without infringing
third-party patent rights. In addition, if the breadth or strength of protection provided by our patents and patent applications (whether
licensed or otherwise held) is threatened, it could dissuade companies from collaborating with us to license, develop or commercialize
current or future product candidates.
Even if our patent applications
(whether licensed or otherwise held) result in the issuance of patents, they may not issue in a form that will provide us with any meaningful
protection, prevent competitors from competing with us or otherwise provide us with any competitive advantage. Our competitors may be
able to circumvent our owned or licensed patents by developing similar or alternative technologies or products in a non-infringing manner.
The issuance of a patent is
not conclusive as to its inventorship, scope, validity or enforceability, and our licensed or owned patents may be challenged in the courts
or patent offices in the United States and abroad. Such challenges may result in loss of exclusivity or freedom to operate or in patent
claims being narrowed, invalidated, or held unenforceable, in whole or in part, which could limit our ability to stop others from using
or commercializing similar or identical products, or limit the duration of the patent protection of our products. Given the amount of
time required for the development, testing and regulatory review of new life science product candidates, patents protecting such candidates
might expire before or shortly after such candidates are commercialized. As a result, our intellectual property rights portfolio may not
provide us with sufficient rights to exclude others from commercializing products similar or identical to ours.
We may become involved in lawsuits to protect
or enforce our intellectual property rights, which could be expensive, time-consuming, and ultimately unsuccessful.
Competitors may infringe our
intellectual property. To counter infringement or unauthorized use, we may be required to file infringement claims, which can be expensive
and time-consuming. Any claims we assert against perceived infringers could provoke these parties to assert counterclaims against us alleging
that we infringe their intellectual property or that our intellectual property is invalid or unenforceable. In addition, in a patent infringement
proceeding, a court may decide that a licensed or owned patent of ours is invalid or unenforceable, in whole or in part, construe the
patent’s claims narrowly or refuse to stop the other party from using the technology at issue on the grounds that our patents do
not cover that technology. Moreover, lawsuits to protect or enforce our intellectual property rights could be expensive, time-consuming,
and ultimately unsuccessful.
Third parties may initiate legal proceedings
alleging that we are infringing their intellectual property rights, the outcome of which would be uncertain.
Our commercial success depends
upon our ability to develop, manufacture, market and sell our product candidates without infringing the proprietary rights of third parties.
There is considerable intellectual property litigation in the life sciences industry. We cannot guarantee that our product candidates
will not infringe third-party patents or other proprietary rights. We may become party to, or threatened with, future adversarial proceedings
or litigation regarding intellectual property rights with respect to our products and technology, including inter partes review,
interference, or derivation proceedings before the USPTO and similar bodies in other countries. Third parties may assert infringement
claims against us based on existing intellectual property rights and intellectual property rights that may be granted in the future.
If we are found to infringe
a third party’s intellectual property rights, we could be required to obtain a license from such third party to continue developing
and marketing our products. However, we may not be able to obtain any required license on commercially reasonable terms or at all. Even
if we were able to obtain a license, it could be non-exclusive, thereby giving our competitors access to the same technologies licensed
to us. We could be forced, including by court order, to cease commercializing the infringing technology or product. In addition, we could
be found liable for monetary damages, including treble damages and attorneys’ fees if we are found to have willfully infringed a
patent. A finding of infringement could prevent us from commercializing our product candidates or force us to cease some of our business
operations, which could materially harm our business. Claims that we have misappropriated the confidential information or trade secrets
of third parties could have a similar negative impact on our business.
Obtaining and maintaining
our patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by
governmental patent agencies, and our own patent protection could be reduced or eliminated for noncompliance with these requirements.
Periodic maintenance fees
and annuities on any issued patent are due to be paid to the USPTO and foreign patent agencies in several stages over the lifetime of
the patent. The USPTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee
payment and other similar provisions during the patent application process. While an inadvertent lapse can in many cases be cured by payment
of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment
or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Noncompliance
events that could result in abandonment or lapse of a patent or patent application include, but are not limited to, failure to respond
to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal documents. In
such an event, our competitors might be able to enter our markets, which could have a material adverse effect on our business.
We may be subject to claims by third parties
asserting that our employees or we have misappropriated their intellectual property or claiming ownership of what we regard as our own
intellectual property.
Certain employees and contractors
were previously employed at universities or other companies, including potential competitors. Although we try to ensure that our employees
and contractors do not use the proprietary information or know-how of others in their work for us, we may be subject to claims that these
employees or we have used or disclosed intellectual property, including trade secrets or other proprietary information, of any such employee’s
former employer. Litigation may be necessary to defend against these claims, and any such litigation could have an unfavorable outcome.
In addition, while it is our
policy to require our employees and contractors who may be involved in the development of intellectual property to execute agreements
assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who in fact develops
intellectual property that we regard as our own. Our and their assignment agreements may not be self-executing or may be breached, and
we may be forced to bring claims against third parties, or defend claims they may bring against us, to determine the ownership of what
we regard as our intellectual property.
If we fail in prosecuting
or defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel.
Even if we are successful in prosecuting or defending against such claims, litigation could result in substantial costs and adverse results,
and be a distraction to management.
Some intellectual property which we own
or have licensed may have been discovered through government funded programs such as, for example, the government funded programs referenced
in intellectual property licensed under the LLU License Agreement, and thus may be subject to federal regulations such as “march-in”
rights, certain reporting requirements, and a preference for United States industry. Compliance with such regulations may limit our exclusive
rights, subject us to expenditure of resources with respect to reporting requirements and limit our ability to contract with non-U.S.
manufacturers.
Some of the intellectual property
rights we own or have licensed have been generated using United States government funding and may therefore be subject to certain federal
regulations. As a result, the United States government may have certain rights to intellectual property embodied in our current or future
products and product candidates pursuant to the Bayh-Dole Act of 1980. These United States government rights in certain inventions developed
under a government-funded program include a non-exclusive, non-transferable, irrevocable worldwide license to use inventions for any governmental
purpose. In addition, the United States government has the right to require us to grant exclusive, partially exclusive, or non-exclusive
licenses to any of these inventions to a third party if it determines that: (i) adequate steps have not been taken to commercialize
the invention; (ii) government action is necessary to meet public health or safety needs; or (iii) government action is necessary
to meet requirements for public use under federal regulations (also referred to as “march-in rights”). The United States government
also has the right to take title to these inventions if we fail to disclose the invention to the government and fail to file an application
to register the intellectual property within specified time limits. In addition, the United States government may acquire title to these
inventions in any country in which a patent application is not filed within specified time limits. Intellectual property generated under
a government funded program is also subject to certain reporting requirements, compliance with which may require us to expend substantial
resources. In addition, the United States government requires that any products embodying the subject invention or produced using the
subject invention be manufactured substantially in the United States. The manufacturing preference requirement can be waived if the owner
of the intellectual property can show that reasonable but unsuccessful efforts have been made to grant licenses on similar terms to potential
licensees that would be likely to manufacture substantially in the United States or that under the circumstances domestic manufacture
is not commercially feasible. This preference for United States manufacturers may limit our ability to contract with non-U.S. product
manufacturers for products covered by such intellectual property. Any exercise by the government of any of the foregoing rights could
harm our competitive position, business, financial condition, results of operations and prospects.
Intellectual property litigation could cause
us to spend substantial resources and distract our personnel from their normal responsibilities.
Even if resolved in our favor,
litigation or other legal proceedings relating to intellectual property claims may cause us to incur significant expenses and could distract
our technical and management personnel from their normal responsibilities. In addition, there could be public announcements of the results
of hearings, motions or other interim proceedings or developments and if securities analysts or investors perceive these results to be
negative, it could have an adverse effect on the price of our common stock. Such litigation or proceedings could increase our operating
losses and reduce the resources available for development activities or any future sales, marketing or distribution activities. We may
not have sufficient financial or other resources to conduct such litigation or proceedings adequately. Some of our competitors may be
able to sustain the costs of such litigation or proceedings more effectively than we can because of their greater financial resources.
Uncertainties resulting from the initiation and continuation of patent litigation or other proceedings could compromise our ability to
compete in the marketplace.
We may spend considerable resources developing
and maintaining patents, licensing agreements and other intellectual property that may later be abandoned or may otherwise never result
in products brought to market.
Not all technologies and candidate
products that initially show potential as the basis for future products ultimately meet the rigors of our development process and as a
result may be abandoned and/or never otherwise result in products brought to market. In some cases, prior to abandonment we may be
required to incur significant costs developing and maintaining intellectual property and/or maintaining license agreements and our business
could be harmed by such costs.
We rely on information technology, and if
we are unable to protect against service interruptions, data corruption, cyber-based attacks or network security breaches, our operations
could be disrupted, and our business could be negatively affected.
We rely on information technology
networks and systems to process, transmit and store electronic and financial information; to coordinate our business; and to communicate
within our Company and with customers, suppliers, partners and other third parties. These information technology systems may be susceptible
to damage, disruptions or shutdowns, hardware or software failures, power outages, computer viruses, cyber-attacks, telecommunication
failures, user errors or catastrophic events. If our information technology systems suffer severe damage, disruption or shutdown, and
our business continuity plans do not effectively resolve the issues in a timely manner, our operations could be disrupted, and our business
could be negatively affected. In addition, cyber-attacks could lead to potential unauthorized access and disclosure of confidential information,
and data loss and corruption. There is no assurance that we will not experience these service interruptions or cyber-attacks in the future.
Risks Related to Our Common Stock
We received a written notice from Nasdaq
that we have failed to comply with certain listing requirements of the Nasdaq Stock Market, which could result in our Common Stock being
delisted from the Nasdaq Stock Market.
On May 23, 2023, we received
written notice from Nasdaq that, based upon the stockholders equity reported by the Company in its Form 10-Q for the period ended
March 31, 2023, and as of March 31, 2023, the Company was no longer in compliance with Nasdaq Listing Rule 5550(b)(1), which requires
a company to maintain a minimum of $2,500,000 in stockholders’ equity, a market value of listed securities of at least $35 million,
or net income from continuing operations of $500,000 in the most recently completed fiscal year or in two of the three most recently completed
fiscal years (the “Continued Listing Requirements”). The May notification letter further provided that the Company has 45
calendar days, or until July 7, 2023, to submit a plan to regain compliance and if the plan is accepted by Nasdaq, an extension of up
to 180 calendar days, or until November 19, 2023 to evidence compliance. On June 22, 2023, we received a letter from Nasdaq notifying
the Company that it has failed to maintain compliance with the minimum bid price rule in Nasdaq Listing Rule 5550(a)(2) (the “Minimum
Bid Price Rule”) as the closing price of Company’s common stock has remained below $1.00 for over 30 consecutive trading days.
On June 29, 2023, we submitted an appeal to Nasdaq, which stays the delisting and suspension of our securities pending the decision of
the Nasdaq Hearings Panel (the “Panel”) no later than 4:00 p.m. Eastern Time on June 29, 2023.
On August 17, 2023, we filed
a Certificate of Amendment to our Certificate of Incorporation, as amended, to effect a reverse stock split at a ratio of one-for-forty
(1:40) in order to regain compliance with the Minimum Bid Price Rule. However, we can provide no assurance that Nasdaq will accept our
plan of compliance or grant us any additional time to demonstrate our ability to regain and sustain compliance with the continued listing
requirements over the long term. If we are delisted from Nasdaq, our common stock may be eligible for trading on an over-the-counter market.
If we are not able to obtain a listing on another stock exchange or quotation service for our common stock, it may be extremely difficult
or impossible for stockholders to sell their shares.
The hearing was held on August
31, 2023, which represented the tenth trading day that the closing price of our Common Stock was above $1.00 per share. At the hearing,
we also presented our views and our plans to regain compliance with the stockholders’ equity requirement to the Panel. It is our
understanding that the Panel typically issues its decision within 30 days after the hearing.
If we are delisted from Nasdaq,
but obtain a substitute listing for our common stock, it will likely be on a market with less liquidity, and therefore experience potentially
more price volatility than experienced on Nasdaq. Stockholders may not be able to sell their shares of common stock on any such substitute
market in the quantities, at the times, or at the prices that could potentially be available on a more liquid trading market. As a result
of these factors, if our common stock is delisted from Nasdaq, the value and liquidity of our common stock, warrants and pre-funded warrants
would likely be significantly adversely affected. A delisting of our common stock from Nasdaq could also adversely affect our ability
to obtain financing for our operations and/or result in a loss of confidence by investors, employees and/or business partners.
We do not expect to pay dividends in the
foreseeable future.
We do not intend to declare
dividends for the foreseeable future, as we anticipate that we will reinvest any and all future earnings in the development and growth
of our business. Therefore, investors will not receive any funds unless they sell their securities, and holders may be unable to sell
their securities on favorable terms or at all. We cannot assure you of a positive return on your investment or that you will not lose
the entire amount of your investment.
We may engage in future acquisitions or
strategic transactions, including the transaction with Cellvera Global, which may require us to seek additional financing or financial
commitments, increase our expenses and/or present significant distractions to our management.
As described herein, on April
19, 2023 entered into an Asset Purchase Agreement with Cellvera Global, pursuant to which we agreed to acquire from Cellvera Ltd a 50%
membership interest in GRA, together with certain other intellectual property assets and all goodwill related thereto. We will need to
acquire additional financing to fund our obligations under the Asset Purchase Agreement or to fund other potential acquisitions or strategic
transactions (particularly if the acquired entity is not cash flow positive or does not have significant cash on hand). Obtaining financing
through the issuance or sale of additional equity and/or debt securities, if possible, may not be at favorable terms and may result in
additional dilution to our current stockholders. Additionally, any such transaction may require us to incur non-recurring or other charges,
may increase our near and long-term expenditures and may pose significant integration challenges or disrupt our management or business,
which could adversely affect our operations and financial results. For example, an acquisition or strategic transaction may entail numerous
operational and financial risks, including the risks outlined above and additionally:
| |
● |
exposure to unknown liabilities; |
| |
● |
disruption of our business and diversion of our management’s time and attention in order to develop acquired products or technologies; |
| |
● |
higher than expected acquisition and integration costs; |
| |
● |
write-downs of assets or goodwill or impairment charges; |
| |
● |
increased amortization expenses; |
| |
● |
difficulty and cost in combining the operations and personnel of any acquired businesses with our operations and personnel; |
| |
● |
impairment of relationships with key suppliers or customers of any acquired businesses due to changes in management and ownership; and |
| |
● |
inability to retain key employees of any acquired businesses. |
Accordingly, although there
can be no assurance that we will undertake or successfully complete any transactions of the nature described above, and any transactions
that we do complete could have a material adverse effect on our business, results of operations, financial condition and prospects.
While we have entered into an Asset Purchase
Agreement with Cellvera Global, we cannot assure you that the transaction contemplated by the Asset Purchase Agreement will be consummated
or, that if such transactions are consummated, they will be accretive to stockholder value.
As described herein, we entered
into an Asset Purchase Agreement with Cellvera Global, pursuant to which we agreed to acquire from Cellvera Ltd a 50% membership interest
in GRA, together with certain other intellectual property assets and all goodwill related thereto. We will be required to obtain additional
financing through the sale of additional equity and/or debt securities to fund such obligations under the Asset Purchase Agreement. We
can provide no assurance that we will be successful in securing such financing. In addition, such financing, if available, may not be
on favorable terms and may result in additional dilution to our current stockholders.
The closing of the transaction
contemplated by the Asset Purchase Agreement are subject to the satisfaction or waiver of a number of closing conditions. There is no
guarantee that the conditions to closing will be satisfied. Further, even if all conditions to closing are satisfied, there is no guarantee
that the transaction will be completed in the time frame or in the manner currently anticipated, or that we will recognize the anticipated
benefits of the transaction.
Upon dissolution of our Company, you may
not recoup all or any portion of your investment.
In the event of a liquidation,
dissolution or winding-up of our Company, whether voluntary or involuntary, our assets would be used to pay all of our debts and liabilities,
and only thereafter would any remaining assets be distributed to our stockholders, subject to rights of the holders of the Preferred Stock,
if any, on a pro rata basis. There can be no assurance that we will have assets available from which to pay any amounts
to our stockholders upon such a liquidation, dissolution or winding-up. In such an event, you would lose all of your investment.
Limitation of Liability and Indemnification
of Management.
The Delaware General Corporation
Law and the Company’s Amended and Restated Certificate of Incorporation provide for the limitation of the liability of directors
for monetary damages. Such provisions may discourage shareholders from bringing a lawsuit against directors for breaches of fiduciary
duty and may also have the effect of reducing the likelihood of derivative litigation against directors and officers even though such
action, if successful, might otherwise be a benefit to the Company’s shareholders. In addition, a shareholder’s investment
in the Company may be adversely affected to the extent that costs of settlement and damage awards against the Company’s officers
or directors are paid by the Company pursuant to such provisions. Additionally, in accordance with Delaware law and the Company’s
Amended and Restated Certificate of Incorporation, the Company shall indemnify, hold harmless and provide advancement of expenses, to
the fullest extent permitted by applicable law, directors, officers, employees, and agents that are made a party or threatened to be made
a party to legal proceedings by reason of the fact that such parties were working at the request of the Company. We direct you to
the Company’s Amended and Restated Certificate of Incorporation for more information.
Anti-takeover provisions under Delaware
law could discourage, delay or prevent a change in control of our Company and could affect the trading price of our securities.
We are a Delaware corporation,
and the anti-takeover provisions of the Delaware General Corporation Law may discourage, delay or prevent a change in control by prohibiting
us from engaging in a business combination with an interested stockholder for a period of three years after the person becomes an interested
stockholder, even if a change in control would be beneficial to our existing stockholders.
Our management team is required to devote
substantial time to public company compliance initiatives.
As a publicly reporting company,
we incur significant legal, accounting, and other expenses. Our management and other personnel devote a substantial amount of time to
comply with our reporting obligations. Moreover, these reporting obligations increase our legal and financial compliance costs and make
some activities more time-consuming and costly.
Failure to develop our internal controls
over financial reporting as we grow could have an adverse impact on us.
As our Company matures, we
will need to develop our current internal control systems and procedures to manage our growth. We are required to establish and maintain
appropriate internal controls over financial reporting. Failure to establish appropriate controls, or any failure of those controls once
established, could adversely impact our public disclosures regarding our business, financial condition, or results of operations. In addition,
management’s assessment of internal controls over financial reporting may identify weaknesses and conditions that need to be addressed
in our internal controls over financial reporting or other matters that may raise concerns for investors. Any actual or perceived weaknesses
and conditions that need to be addressed in our internal control over financial reporting, disclosure of management’s assessment
of our internal controls over financial reporting or disclosure of our public accounting firm’s attestation to or report on management’s
assessment of our internal controls over financial reporting may have an adverse impact on the price of our common stock.
We could issue “blank check” preferred
stock without stockholder approval with the effect of diluting interests of then-current stockholders and impairing their voting
rights, and provisions in our charter documents and under Delaware law could discourage a takeover that stockholders may consider favorable.
Our Amended and Restated Certificate
of Incorporation provides for the authorization to issue up to 3,000,000 shares of “blank check” preferred stock with
designations, rights and preferences as may be determined from time to time by our board of directors. Our board of directors is empowered,
without stockholder approval, to issue one or more series of preferred stock with dividend, liquidation, conversion, voting or other rights
which could dilute the interest of, or impair the voting power of, our common stockholders. The issuance of a series of preferred stock
could be used as a method of discouraging, delaying, or preventing a change in control. For example, it would be possible for our board
of directors to issue preferred stock with voting or other rights or preferences that could impede the success of any attempt to change
control of our company. In addition, advanced notice is required prior to stockholder proposals, which might further delay a change of
control.
Our Amended and Restated Certificate of
Incorporation provides that the Court of Chancery of the State of Delaware will be the sole and exclusive forum for substantially
all disputes between the Company and its stockholders, which could limit stockholders’ ability to obtain a favorable judicial forum
for disputes with the Company or its directors, officers or employees.
Our Amended and Restated Certificate
of Incorporation provides that unless the Company consents in writing to the selection of an alternative forum, the State of Delaware
is the sole and exclusive forum for: (i) any derivative action or proceeding brought on behalf of the Company, (ii) any action asserting
a claim of breach of a fiduciary duty owed by any director, officer or other employee of the Company to the Company or the Company’s
stockholders, (iii) any action asserting a claim against the Company, its directors, officers or employees arising pursuant to any provision
of the Delaware General Corporation Law (the “DGCL”) or our Amended and Restated Certificate of Incorporation or
the Company’s Amended and Restated Bylaws, or (iv) any action asserting a claim against the Company, its directors, officers, employees
or agents governed by the internal affairs doctrine, except for, as to each of (i) through (iv) above, any claim as to which the Court
of Chancery determines that there is an indispensable party not subject to the jurisdiction of the Court of Chancery (and the indispensable
party does not consent to the personal jurisdiction of the Court of Chancery within ten days following such determination), which is vested
in the exclusive jurisdiction of a court or forum other than the Court of Chancery, or for which the Court of Chancery does not have subject
matter jurisdiction. This exclusive forum provision would not apply to suits brought to enforce any liability or duty created by the Securities
Act or the Exchange Act or any other claim for which the federal courts have exclusive jurisdiction. To the extent that any such claims
may be based upon federal law claims, Section 27 of the Exchange Act creates exclusive federal jurisdiction over all suits brought to
enforce any duty or liability created by the Exchange Act or the rules and regulations thereunder.
Section 22 of the Securities
Act creates concurrent jurisdiction for federal and state courts over all suits brought to enforce any duty or liability created by the
Securities Act or the rules and regulations thereunder. However, our Amended and Restated Bylaws contain a federal forum provision which
provides that unless the Company consents in writing to the selection of an alternative forum, the federal district courts of the United
States of America will be the exclusive forum for the resolution of any complaint asserting a cause of action arising under the Securities
Act. Any person or entity purchasing or otherwise acquiring any interest in shares of capital stock of the Corporation are deemed to have
notice of and consented to this provision. The Supreme Court of Delaware has held that this type of exclusive federal forum provision
is enforceable. There may be uncertainty, however, as to whether courts of other jurisdictions would enforce such a provision, if applicable.
These choice of forum provisions
may limit a stockholder’s ability to bring a claim in a judicial forum that it finds favorable for disputes with the Company or
its directors, officers or other employees, which may discourage such lawsuits against us and our directors, officers and other employees.
Alternatively, if a court were to find our choice of forum provisions contained in either our Amended and Restated Certificate of Incorporation
or Amended and Restated Bylaws to be inapplicable or unenforceable in an action, we may incur additional costs associated with resolving
such action in other jurisdictions, which could harm our business, results of operations, and financial condition.
We are an “emerging growth company”
and will be able to avail ourselves of reduced disclosure requirements applicable to emerging growth companies, which could make our common
stock less attractive to investors.
We are an “emerging
growth company,” as defined in the Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”), and we intend to take
advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not “emerging
growth companies” including not being required to comply with the auditor attestation requirements of Section 404(b) of the
Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and
exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden
parachute payments not previously approved. In addition, pursuant to Section 107 of the JOBS Act, as an “emerging growth company”
we intend to take advantage of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act, for complying
with new or revised accounting standards. In other words, an “emerging growth company” can delay the adoption of certain accounting
standards until those standards would otherwise apply to private companies. As a result, our financial statements may not be comparable
to those of companies that comply with public company effective dates for complying with new or revised accounting standards.
We cannot predict if investors
will find our common stock less attractive because we may rely on these exemptions. If some investors find our common stock less attractive
as a result, there may be a less active trading market for our common stock and our stock price may be more volatile. We may take advantage
of these reporting exemptions until we are no longer an “emerging growth company.” We will remain an “emerging growth
company” until the earliest of (i) the last day of the fiscal year in which we have total annual gross revenues of $1.07 billion
or more; (ii) the last day of our fiscal year following the fifth anniversary of the date of the completion of our initial public
offering; (iii) the date on which we have issued more than $1 billion in nonconvertible debt during the previous three years;
or (iv) the date on which we are deemed to be a large accelerated filer under the rules of the SEC.
USE OF PROCEEDS
We will not receive any of the proceeds from the
sale by the Selling Stockholders of the Common Stock. Upon any exercise of the Warrants by payment of cash, however, we will receive the
exercise price of the Warrants, which, if exercised in cash with respect to the 2,060,000 shares of Common Stock offered hereby, would
result in gross proceeds to us of approximately $10.75 million. However, we cannot predict when and in what amounts or if the Warrants
will be exercised by payments of cash and it is possible that the Warrants may expire and never be exercised, in which case we would not
receive any cash proceeds. In addition, upon exercise of any of the Common Warrants, we will
pay the Placement Agent a cash fee equal to 7.75% of the gross proceeds received from the exercise of the Common Warrants and will also
issue to the Placement Agent (or its designees) additional Placement Agent Warrants to purchase a number of shares of Common Stock equal
to 6.0% of the aggregate number of shares of Common Stock issued upon such exercise of the Common Warrants.
DIVIDEND POLICY
We
have never paid cash dividends on our Common Stock and we do not anticipate paying cash dividends in the foreseeable future, but intend
to retain our capital resources for reinvestment in our business. Any future determination to pay cash dividends on our Common Stock will
be at the discretion of our board of directors and will be dependent upon our financial condition, results of operations, capital requirements
and other factors as the board of directors deems relevant.
DETERMINATION OF THE
OFFERING PRICE
The prices at which the
shares of Common Stock covered by this prospectus may actually be sold will be determined by the prevailing public market price for shares
of our Common Stock or by negotiations between the Selling Stockholders and buyers of our Common Stock in private transactions or as otherwise
described in “Plan of Distribution.”
SELLING STOCKHOLDERS
The Common Stock being offered
by the Selling Stockholders are those issuable to the Selling Stockholders, upon exercise of the Warrants. For additional information
regarding the issuances of those shares of Common Stock and Warrants, see “Prospectus Summary—Recent Developments—August
2023 Private Placement” above. We are registering the shares of Common Stock in order to permit the Selling Stockholders to
offer the shares for resale from time to time. Except for the ownership of the Warrants and as noted below, the Selling Stockholders have
not had any material relationship with us within the past three years.
The table below lists the
Selling Stockholders and other information regarding the beneficial ownership of the shares of Common Stock by each of the Selling Stockholders.
The second column lists the number of shares of Common Stock beneficially owned by each Selling Stockholder, based on its ownership of
the shares of Common Stock and Warrants, as of September 14, 2023, assuming exercise of the Warrants held by the Selling Stockholders
on that date, without regard to any limitations on exercises.
The third column lists the
shares of Common Stock being offered by this prospectus by the Selling Stockholders.
In accordance with the terms
of a registration rights agreement with the Selling Stockholders, this prospectus generally covers the resale of the maximum number of
shares of Common Stock issuable upon exercise of the Warrants, determined as if the outstanding Warrants were exercised in full as of
the trading day immediately preceding the date this registration statement was initially filed with the SEC, each as of the trading day
immediately preceding the applicable date of determination and all subject to adjustment as provided in the registration rights agreement,
without regard to any limitations on the exercise of the Warrants. The fourth column assumes the sale of all of the shares offered by
the Selling Stockholders pursuant to this prospectus.
Under the terms of the Warrants,
a Selling Stockholder may not exercise the Warrants to the extent such exercise would cause such Selling Stockholder, together with its
affiliates and attribution parties, to beneficially own a number of shares of Common Stock which would exceed 4.99% or 9.99%, as applicable,
of our then outstanding Common Stock following such exercise, excluding for purposes of such determination shares of Common Stock issuable
upon exercise of such Warrants which have not been exercised. The number of shares in the second and fourth columns do not reflect this
limitation. The Selling Stockholders may sell all, some or none of their shares in this offering. See “Plan of Distribution.”
| Name of Selling Stockholder | |
Number of
Shares
of Common
Stock
Beneficially
Owned
Prior to
Offering(1) | | |
Maximum
Number of
Shares
of Common
Stock
to be Sold
in this
Offering | | |
Number of
Shares
of Common
Stock
Beneficially
Owned After
Offering | | |
Percentage
of Shares
Beneficially
Owned after
Offering(1) | |
| Armistice Capital, LLC.(2) | |
| 2,030,385 | | |
| 2,000,000 | | |
| 30,385 | | |
| 1.4 | % |
| Michael Vasinkevich(3) | |
| 38,475 | | |
| 38,475 | | |
| 0 | | |
| 0 | % |
| Michael Mirsky(3) | |
| 11,400 | | |
| 11,400 | | |
| 0 | | |
| 0 | % |
| Noam Rubinstein(3) | |
| 7,500 | | |
| 7,500 | | |
| 0 | | |
| 0 | % |
| Craig Schwabe(3) | |
| 2,025 | | |
| 2,025 | | |
| 0 | | |
| 0 | % |
| Charles Worthman(3) | |
| 600 | | |
| 600 | | |
| 0 | | |
| 0 | % |
| (1) | The ability to exercise
the Warrants and Pre-Funded Warrants, as applicable, held by the Selling Stockholders is subject to a beneficial ownership limitation
that, at the time of initial issuance of the Warrants and Pre-Funded Warrants, as applicable, was capped at either 4.99% or 9.99% beneficial
ownership of the Company’s issued and outstanding Common Stock (post-exercise). These beneficial ownership limitations may be adjusted
up or down, subject to providing advanced notice to the Company. Beneficial ownership as reflected in the selling stockholder table reflects
the total number of shares potentially issuable underlying the Warrants and Pre-Funded Warrants, as applicable, and does not give
effect to these beneficial ownership limitations. Accordingly, actual beneficial ownership, as calculated in accordance with Section
13(d) and Rule 13d-3 thereunder may be lower than as reflected in the table. |
| (2) | The securities are directly held by Armistice Capital Master
Fund Ltd., a Cayman Islands exempted company (the “Master Fund”), and may be deemed to be beneficially owned by: (i) Armistice
Capital, LLC (“Armistice Capital”), as the investment manager of the Master Fund; and (ii) Steven Boyd, as the Managing Member
of Armistice Capital. The warrants are subject to a beneficial ownership limitation of 4.99%, which such limitation restricts the Selling
Stockholder from exercising that portion of the warrants that would result in the Selling Stockholder and its affiliates owning, after
exercise, a number of shares of Common Stock in excess of the beneficial ownership limitation. The address of Armistice Capital Master
Fund Ltd. is c/o Armistice Capital, LLC, 510 Madison Avenue, 7th Floor, New York, NY 10022. |
| (3) | The selling stockholder
is affiliated with H.C. Wainwright & Co., LLC, a registered broker dealer with a registered address of H.C. Wainwright & Co.,
LLC, 430 Park Ave, 3rd Floor, New York, NY 10022, and has sole voting and dispositive power over the securities held. The number of shares
beneficially owned prior to this offering consist of shares of Common Stock issuable upon
exercise of placement agent warrants, which were received as compensation in connection with the concurrent Registered Direct
Offering and the Private Placements consummated by us in June 2023. The selling stockholder acquired
the placement agent warrants in the ordinary course of business and, at the time the placement agent warrants were acquired, the selling
stockholder had no agreement or understanding, directly or indirectly, with any person to distribute such securities. |
Plan
of Distribution
Each Selling Stockholder of
the securities and any of their pledgees, assignees and successors-in-interest may, from time to time, sell any or all of their securities
covered hereby on the principal Trading Market or any other stock exchange, market or trading facility on which the securities are traded
or in private transactions. These sales may be at fixed or negotiated prices. A Selling Stockholder may use any one or more of the following
methods when selling securities:
| ● | ordinary
brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
| ● | block
trades in which the broker-dealer will attempt to sell the securities as agent but may position and resell a portion of the block as
principal to facilitate the transaction; |
| ● | purchases
by a broker-dealer as principal and resale by the broker-dealer for its account; |
| ● | an
exchange distribution in accordance with the rules of the applicable exchange; |
| ● | privately
negotiated transactions; |
| ● | settlement
of short sales; |
| ● | in
transactions through broker-dealers that agree with the Selling Stockholders to sell a specified number of such securities at a stipulated
price per security; |
| ● | through
the writing or settlement of options or other hedging transactions, whether through an options exchange or otherwise; |
| ● | a
combination of any such methods of sale; or |
| ● | any
other method permitted pursuant to applicable law. |
The Selling Stockholders may
also sell securities under Rule 144 or any other exemption from registration under the Securities Act of 1933, as amended (the “Securities
Act”), if available, rather than under this prospectus. Broker-dealers engaged by the Selling Stockholders may arrange for other
brokers-dealers to participate in sales. Broker-dealers may receive commissions or discounts from the Selling Stockholders (or, if any
broker-dealer acts as agent for the purchaser of securities, from the purchaser) in amounts to be negotiated, but, except as set forth
in a supplement to this prospectus, in the case of an agency transaction not in excess of a customary brokerage commission in compliance
with FINRA Rule 2121; and in the case of a principal transaction a markup or markdown in compliance with FINRA Rule 2121.
In connection with the sale
of the securities or interests therein, the Selling Stockholders may enter into hedging transactions with broker-dealers or other financial
institutions, which may in turn engage in short sales of the securities in the course of hedging the positions they assume. The Selling
Stockholders may also sell securities short and deliver these securities to close out their short positions, or loan or pledge the securities
to broker-dealers that in turn may sell these securities. The Selling Stockholders may also enter into option or other transactions with
broker-dealers or other financial institutions or create one or more derivative securities which require the delivery to such broker-dealer
or other financial institution of securities offered by this prospectus, which securities such broker-dealer or other financial institution
may resell pursuant to this prospectus (as supplemented or amended to reflect such transaction).
The Selling Stockholders and
any broker-dealers or agents that are involved in selling the securities may be deemed to be “underwriters” within the meaning
of the Securities Act in connection with such sales. In such event, any commissions received by such broker-dealers or agents and any
profit on the resale of the securities purchased by them may be deemed to be underwriting commissions or discounts under the Securities
Act. Each Selling Stockholder has informed us that it does not have any written or oral agreement or understanding, directly or indirectly,
with any person to distribute the securities.
We are required to pay certain
fees and expenses incurred by us incident to the registration of the securities. We have agreed to indemnify the Selling Stockholders
against certain losses, claims, damages and liabilities, including liabilities under the Securities Act.
We agreed to keep this prospectus
effective until the earlier of (i) the date on which the securities may be resold by the Selling Stockholders without registration and
without regard to any volume or manner-of-sale limitations by reason of Rule 144, without the requirement for us to be in compliance with
the current public information under Rule 144 under the Securities Act or any other rule of similar effect or (ii) all of the securities
have been sold pursuant to this prospectus or Rule 144 under the Securities Act or any other rule of similar effect. The resale securities
will be sold only through registered or licensed brokers or dealers if required under applicable state securities laws. In addition, in
certain states, the resale securities covered hereby may not be sold unless they have been registered or qualified for sale in the applicable
state or an exemption from the registration or qualification requirement is available and is complied with.
Under
applicable rules and regulations under the Exchange Act, any person engaged in the distribution of the resale securities may not simultaneously
engage in market making activities with respect to the Common Stock for the applicable restricted period, as defined in Regulation M,
prior to the commencement of the distribution. In addition, the Selling Stockholders will be subject to applicable provisions of the Exchange
Act and the rules and regulations thereunder, including Regulation M, which may limit the timing of purchases and sales of the Common
Stock by the Selling Stockholders or any other person. We will make copies of this prospectus available to the Selling Stockholders and
have informed them of the need to deliver a copy of this prospectus to each purchaser at or prior to the time of the sale (including by
compliance with Rule 172 under the Securities Act).
DESCRIPTION OF CAPITAL
STOCK
Common Stock
General Matters
The following description
summarizes the most important terms of our capital stock. Because it is only a summary of the provisions of our certificate of incorporation,
as amended (the “Certificate of Incorporation”), and bylaws, as amended (the “Bylaws”), it does
not contain all of the information that may be important to you. For a complete description of the matters set forth in this “Description
of Capital Stock,” you should refer to our Certificate of Incorporation and Bylaws, each of which are included as exhibits to the
registration statement of which this prospectus is a part, and to the applicable provisions of Delaware law.
As of September 14, 2023,
our authorized capital stock consisted of 100,000,000 shares of Common Stock and 3,000,000 shares of preferred stock, $0.001 par value
per share.
Common Stock
Voting
The holders of our common
stock are entitled to one vote for each share held on all matters to be voted on by the Company’s stockholders. There shall be no
cumulative voting.
Dividends
The holders of shares of our
common stock are entitled to dividends when and as declared by the Board from funds legally available therefor if, as and when determined
by the Board of Directors of the Company in their sole discretion, subject to provisions of law, and any provision of the Company’s
Amended and Restated Certificate of Incorporation, as amended from time to time. There are no preemptive, conversion or redemption privileges,
nor sinking fund provisions with respect to the common stock.
Liquidation
In the event of any voluntary
or involuntary liquidation, dissolution or winding up of our affairs, the holders of our common stock will be entitled to share ratably
in the net assets legally available for distribution to stockholders after the payment of or provision for all of our debts and other
liabilities.
Fully Paid and Non-assessable
All outstanding shares of
common stock are duly authorized, validly issued, fully paid and non-assessable.
Preferred Stock
We are authorized to issue
up to 3,000,000 shares of preferred stock. This preferred stock may be issued in one or more series, the terms of which may be determined
at the time of issuance by our board of directors without further action by stockholders. The terms of any series of preferred stock may
include voting rights (including the right to vote as a series on particular matters), preferences as to dividend, liquidation, conversion
and redemption rights and sinking fund provisions. No preferred stock is currently outstanding. The issuance of any preferred stock could
materially adversely affect the rights of the holders of our common stock, and therefore, reduce the value of our common stock and the
Notes. In particular, specific rights granted to future holders of preferred stock could be used to restrict our ability to merge with,
or sell our assets to, a third party and thereby preserve control by the present management.
Exclusive Forum
Our Amended and Restated Certificate
of Incorporation provides that unless the Company consents in writing to the selection of an alternative forum, the State of Delaware
is the sole and exclusive forum for: (i) any derivative action or proceeding brought on behalf of the Company, (ii) any action asserting
a claim of breach of a fiduciary duty owed by any director, officer or other employee of the Company to the Company or the Company’s
stockholders, (iii) any action asserting a claim against the Company, its directors, officers or employees arising pursuant to any provision
of the DGCL or our Amended and Restated Certificate of Incorporation or the Amended and Restated Bylaws, or (iv) any action asserting
a claim against the Company, its directors, officers, employees or agents governed by the internal affairs doctrine, except for, as to
each of (i) through (iv) above, any claim as to which the Court of Chancery determines that there is an indispensable party not subject
to the jurisdiction of the Court of Chancery (and the indispensable party does not consent to the personal jurisdiction of the Court of
Chancery within ten days following such determination), which is vested in the exclusive jurisdiction of a court or forum other than the
Court of Chancery, or for which the Court of Chancery does not have subject matter jurisdiction.
Additionally, our Amended
and Restated Bylaws provide that unless the Company consents in writing to the selection of an alternative forum, the federal district
courts of the United States of America will be the exclusive forum for the resolution of any complaint asserting a cause of action arising
under the Securities Act. Any person or entity purchasing or otherwise acquiring any interest in shares of capital stock of the Corporation
are deemed to have notice of and consented to this provision. The Supreme Court of Delaware has held that this type of exclusive federal
forum provision is enforceable. There may be uncertainty, however, as to whether courts of other jurisdictions would enforce such a provision,
if applicable.
Transfer Agent
The transfer agent and registrar
for our common stock is VStock Transfer, LLC.
Changes in Authorized Number
The number of authorized shares
of common stock may be increased or decreased subject to the Company’s legal commitments at any time and from time to time to issue
them, by the affirmative vote of the holders of a majority of the stock of the Company entitled to vote.
Delaware Anti-Takeover Statute
We may become subject to Section
203 of the Delaware General Corporation Law, which prohibits persons deemed to be “interested stockholders” from engaging
in a “business combination” with a publicly held Delaware corporation for three years following the date these persons become
interested stockholders unless the business combination is, or the transaction in which the person became an interested stockholder was,
approved in a prescribed manner or another prescribed exception applies. Generally, an “interested stockholder” is a person
who, together with affiliates and associates, owns, or within three years prior to the determination of interested stockholder status
did own, 15% or more of a corporation’s voting stock. Generally, a “business combination” includes a merger, asset or
stock sale, or other transaction resulting in a financial benefit to the interested stockholder. The existence of this provision may have
an anti-takeover effect with respect to transactions not approved in advance by the Board of Directors. A Delaware corporation may “opt
out” of these provisions with an express provision in its original certificate of incorporation or an express provision in its certificate
of incorporation or bylaws resulting from a stockholders’ amendment approved by at least a majority of the outstanding voting shares.
We have not opted out of these provisions. As a result, mergers or other takeover or change in control attempts of us may be discouraged
or prevented.
The Amended and Restated Bylaws
establish an advance notice procedure for stockholder proposals to be brought before an annual meeting of our stockholders, including
proposed nominations of persons for election to our board of directors. At an annual meeting, stockholders may only consider proposals
or nominations specified in the notice of meeting or brought before the meeting by or at the direction of our board of directors. Stockholders
may also consider a proposal or nomination by a person who was a stockholder at the time of giving notice and at the time of the meeting,
who is entitled to vote at the meeting and who has complied with the notice requirements of the Amended and Restated Bylaws in all respects.
The Amended and Restated Bylaws do not give our board of directors the power to approve or disapprove stockholder nominations of candidates
or proposals regarding other business to be conducted at a special or annual meeting of our stockholders. However, the Amended and Restated
Bylaws may have the effect of precluding the conduct of certain business at a meeting if the proper procedures are not followed. These
provisions may also discourage or deter a potential acquirer from conducting a solicitation of proxies to elect the acquirer’s own
slate of directors or otherwise attempting to obtain control of our company.
The Amended and Restated Bylaws
provide that a special meeting of our stockholders may be called only by our Chairman or by resolution adopted by a majority of our board
of directors. Because our stockholders do not have the right to call a special meeting, a stockholder could not force stockholder consideration
of a proposal over the opposition of our board of directors by calling a special meeting of stockholders prior to such time as a majority
of our board of directors, the chairperson of our board of directors, the president or the chief executive officer believed the matter
should be considered or until the next annual meeting provided that the requestor met the notice requirements. The restriction
on the ability of stockholders to call a special meeting means that a proposal to replace our board of directors also could be delayed
until the next annual meeting.
Transfer Agent and Registrar
The transfer agent and registrar
for our Common Stock is VStock Transfer LLC. The transfer agent and registrar’s address is 18 Lafayette Place, Woodmere, NY 11598.
Listing
Our shares of common stock
are listed on The Nasdaq Capital Market under the symbol “ADTX.”
LEGAL MATTERS
Sheppard, Mullin, Richter
& Hampton LLP, New York, New York, will pass upon the validity of the shares of our common stock offered hereby.
EXPERTS
dbbmckennon, an independent
registered public accounting firm, has audited our financial statements included in our Annual Report on Form 10-K for the year ended
December 31, 2022, as set forth in their report, which includes an explanatory paragraph as to our ability to continue as a going concern,
dated April 17, 2023, which is incorporated by reference in this prospectus and elsewhere in the registration statement. Our financial
statements are incorporated by reference in reliance on dbbmckennon report, given on the authority of such firm as experts in accounting
and auditing.
WHERE YOU CAN FIND MORE INFORMATION
We have filed with the SEC
a registration statement on Form S-1 under the Securities Act with respect to the securities offered by this prospectus. This prospectus,
which constitutes a part of the registration statement, does not contain all of the information set forth in the registration statement,
some of which is contained in exhibits to the registration statement as permitted by the rules and regulations of the SEC. For further
information with respect to us and our securities, we refer you to the registration statement, including the exhibits filed as a part
of the registration statement. Statements contained in this prospectus concerning the contents of any contract or any other document is
not necessarily complete. If a contract or document has been filed as an exhibit to the registration statement, please see the copy of
the contract or document that has been filed. Each statement is this prospectus relating to a contract or document filed as an exhibit
is qualified in all respects by the filed exhibit. We are subject to the informational requirements of the Exchange Act and in accordance
therewith file annual, quarterly and current reports, proxy statements and other information with the SEC. The SEC maintains an Internet
website that contains reports, proxy statements and other information about issuers, like us, that file electronically with the SEC. The
address of that website is www.sec.gov. The registration statement and the documents referred to below under “Incorporation
of Documents By Reference” are also available on our website, www.astrotechcorp.com. We have not incorporated by reference
into this prospectus the information on our website, and you should not consider it to be a part of this prospectus.
INCORPORATION OF DOCUMENTS BY REFERENCE
This prospectus is part of
the registration statement but the registration statement includes and incorporates by reference additional information and exhibits.
The SEC permits us to “incorporate by reference” the information contained in documents we file with the SEC, which means
that we can disclose important information to you by referring you to those documents rather than by including them in this prospectus.
Information that is incorporated by reference is considered to be part of this prospectus and you should read it with the same care that
you read this prospectus. Information that we file later with the SEC will automatically update and supersede the information that is
either contained, or incorporated by reference, in this prospectus, and will be considered to be a part of this prospectus from the date
those documents are filed. We have filed with the SEC, and incorporate by reference in this prospectus:
| |
● |
Current Reports on Form 8-K, filed with the SEC on January 6, 2023, January 20, 2023, February 24, 2023, March 21, 2023, April 7, 2023, April 17, 2023, April 24, 2023, April 25, 2023, April 28, 2023, May 26, 2023, May 31, 2023, June 16, 2023, June 21, 2023, June 30, 2023, July 7, 2023, July 14, 2023, July 27, 2023, July 28, 2023, August 16, 2023, August 17, 2023, August 18, 2023, August 21, 2023, August 28, 2023, and September 6, 2023; |
| |
● |
Annual Report on Form 10-K for the year ended December 31, 2022 originally filed with the SEC on April 17, 2023 and amended on April 28, 2023; |
| |
● |
Quarterly Report on Form 10-Q for the three months ended March 31, 2023 filed with the SEC on May 15, 2023; |
| |
● |
Quarterly Report on Form 10-Q for the six months ended June 30, 2023 filed with the SEC on August 14, 2023; |
| |
● |
Proxy Statement on Schedule 14A filed on July 20, 2023, as amended; and |
| |
● |
the
description of our common stock and our preferred stock contained in our Registration Statement on Form 8-A12B/A filed with the Commission on June 17,
2020, and any amendments or reports filed updating such description. |
In addition, all documents
subsequently filed by us pursuant to Sections 13(a), 13(c), 14 or 15(d) of the Securities Exchange Act of 1934, as amended, prior to the
termination of the offering (excluding any information furnished rather than filed) shall be deemed to be incorporated by reference into
this prospectus.
Notwithstanding the statements
in the preceding paragraphs, no document, report or exhibit (or portion of any of the foregoing) or any other information that we have
“furnished” to the SEC pursuant to the Securities Exchange Act of 1934, as amended shall be incorporated by reference into
this prospectus.
We will furnish without charge
to you, on written or oral request, a copy of any or all of the documents incorporated by reference in this prospectus, including exhibits
to these documents. You should direct any requests for documents to:
Aditxt, Inc.
737 N. Fifth Street, Suite 200
Richmond, VA 23219
Phone: (650) 870-1200
You also may access these
filings on our website at http://www.aditxt.com. We do not incorporate the information on our website into this prospectus or any supplement
to this prospectus and you should not consider any information on, or that can be accessed through, our website as part of this prospectus
or any supplement to this prospectus (other than those filings with the SEC that we specifically incorporate by reference into this prospectus
or any supplement to this prospectus).
Any statement contained in
a document incorporated or deemed to be incorporated by reference in this prospectus will be deemed modified, superseded or replaced for
purposes of this prospectus to the extent that a statement contained in this prospectus modifies, supersedes or replaces such statement.
Any statement contained herein or in any document incorporated or deemed to be incorporated by reference shall be deemed to be modified
or superseded for purposes of the registration statement of which this prospectus forms a part to the extent that a statement contained
in any other subsequently filed document which also is or is deemed to be incorporated by reference modifies or supersedes such statement.
Any such statement so modified or superseded shall not be deemed to constitute a part of the registration statement of which this prospectus
forms a part, except as so modified or superseded.
Aditxt, Inc.
Up to 2,060,000 Shares
of Common Stock
PROSPECTUS
, 2023
Part II
INFORMATION NOT REQUIRED IN PROSPECTUS
Item 13. Other Expenses of Issuance and Distribution.
We estimate that expenses
in connection with the distribution described in this registration statement (other than brokerage commissions, discounts or other expenses
relating to the sale of the shares in this offering) will be as set forth below. We will pay all of the expenses with respect to the distribution,
and such amounts, with the exception of the SEC registration fee and FINRA fee, are estimates.
| SEC expenses | |
$ | | |
| Legal fees and expenses | |
| | |
| Accounting fees and expenses | |
| | |
| Miscellaneous expenses | |
| | |
| Total offering expenses | |
$ | | |
Item 14. Indemnification of Directors and Officers
Section 102 of Delaware General
Corporation Law (the “DGCL”) permits a corporation to eliminate the personal liability of directors of a corporation to the
corporation or its stockholders for monetary damages for a breach of fiduciary duty as a director, except where the director breached
his duty of loyalty, failed to act in good faith, engaged in intentional misconduct or knowingly violated a law, authorized the payment
of a dividend or approved a stock repurchase in violation of Delaware corporate law or obtained an improper personal benefit. Our amended
and restated certificate of incorporation provides that no director of Aditxt shall be personally liable to it or its stockholders for
monetary damages for any breach of fiduciary duty as a director, notwithstanding any provision of law imposing such liability, except
to the extent that the DGCL prohibits the elimination or limitation of liability of directors for breaches of fiduciary duty.
Section 145 of the DGCL provides
that a corporation has the power to indemnify a director, officer, employee, or agent of the corporation, or a person serving at the request
of the corporation for another corporation, partnership, joint venture, trust or other enterprise in related capacities against expenses
(including attorneys’ fees), judgments, fines and amounts paid in settlement actually and reasonably incurred by the person in connection
with an action, suit or proceeding to which he was or is a party or is threatened to be made a party to any threatened, ending or completed
action, suit or proceeding by reason of such position, if such person acted in good faith and in a manner he reasonably believed to be
in or not opposed to the best interests of the corporation, and, in any criminal action or proceeding, had no reasonable cause to believe
his conduct was unlawful, except that, in the case of actions brought by or in the right of the corporation, no indemnification shall
be made with respect to any claim, issue or matter as to which such person shall have been adjudged to be liable to the corporation unless
and only to the extent that the Court of Chancery or other adjudicating court determines that, despite the adjudication of liability but
in view of all of the circumstances of the case, such person is fairly and reasonably entitled to indemnity for such expenses which the
Court of Chancery or such other court shall deem proper.
Our Certificate of Incorporation
as amended and Bylaws provide indemnification for our directors and officers to the fullest extent permitted by the DGCL.
Item 15. Recent Sales of Unregistered Securities
In the three years preceding the filing of this
registration statement, the Registrant has issued the following securities that were not registered under the Securities Act.
On January 31, 2022, the Company
issued a consultant 2 shares of common stock for services rendered.
On February 28, 2022, the
Company issued a consultant 2 shares of common stock for services rendered.
On June 27, 2022, the Company
issued a consultant 768 shares of common stock for services rendered.
On December
7, 2022, the Company issued a consultant 3,279 shares of common stock for services rendered.
On December
27, 2022, the Company issued a consultant 246 shares of common stock for services rendered.
On March 17, 2023, the Company
issued a consultant 4,675 shares of common stock for services rendered.
The issuances above were made
pursuant to Section 4(a)(2) of the Securities Act.
On June 15, 2022, the Company
entered into a letter agreement (the “Letter Agreement”) with a holder of certain of the Series C Warrants (the “Holder”).
Pursuant to the Letter Agreement, the Holder has agreed to exercise in cash 4,486 of its Series C Warrants at a reduced exercise price
of $300.00 per Share (reduced from $2,300.00 per Share), for gross proceeds to the Company of approximately $1.35 million. As an inducement
to such exercise, the Company has agreed to reduce the exercise price of the Holder’s remaining Series C Warrants to purchase up
to 1,229 Shares from $2,300.00 to $495.80 per share (the “Amended Series C Warrant”). The Amended Series C Warrant will be
non-exercisable for a period of six months following the closing date. In addition, the Company shall issue to the Holder a new warrant
(the “New Warrant”) to purchase up to 10,200 shares of the Company’s common stock at an exercise price of $495.80 per
share. The New Warrant will be non-exercisable for a period of six months following issuance date and have a term of five and one-half
years. The Shares of common stock issuable upon exercise of the Amended Series C Warrants are registered pursuant to the Company’s
Registration Statement on Form S-3 (Registration No. 333-257645), which was initially filed with the Securities and Exchange Commission
on July 2, 2021, and declared effective on July 13, 2021, and prospectus supplement thereto. The New Warrants were issued pursuant to
Section 4(a)(2) of the Securities Act.
On August 4, 2022, the Company
entered into a Securities Purchase Agreement (the “SPA”) with certain accredited investors providing for the issuance and
sale by the Company to the purchasers signatory thereto, of: (i) $1,477,777.78 in principal
amount 10% Senior Secured Promissory Notes (the “August 2022 Notes”), resulting in gross proceeds to the Company of $1,330,000.00,
exclusive of placement agent commission and fees and other offering expenses; (ii) 739 shares of common stock as commitment
fees (the “August 2022 Commitment Shares”); and warrants (the “Warrants”)
to purchase up to 3,138 shares (the “August 2022 Warrant Shares”) of the Company’s common stock (together with the August
2022 Notes, the August 2022 Commitment Shares and the August 2022 Warrant Shares, the “August 2022 Securities”).
The August 2022 Notes have
a maturity date of twelve (12) months from the date of issuance and are convertible at the option of the Investor at any time prior to
maturity in shares of Common Stock (the “Conversion Shares”) at an initial conversion price of $471.00 per share, subject
to adjustment under certain circumstances. The holders of the August 2022 Notes have the right, following any calendar day following the
Commencement Date (as defined therein) to convert all or any portion of the then outstanding and unpaid principal amount and interest
into fully paid and non-assessable shares of common stock at the conversion price. The Company is prohibited from effecting a conversion
of the Note to the extent that, as a result of such exercise, the Investor, together with the its affiliates, would beneficially own more
than 4.99% of the number of shares of common stock outstanding immediately after giving effect to the issuance of such shares. In addition,
the sum of the aggregate number of shares of common stock that may be issued to all Investors under the August 2022 Securities is limited
to 19.99% of the Company’s then outstanding shares of common stock as of the date of issuance unless Shareholder Approval (as defined
in the SPA) is obtained to issue more than the 19.99%.
The August 2022 Warrants are
exercisable for a period of five (5) years from period commencing on the Commencement Date (as defined therein) and ending on 5:00 p.m.
eastern standard time on the date that is five (5) years after the date of issuance, at an exercise price of $471.00, subject to adjustment
provided therein (including cashless exercise). The Company is prohibited from effecting an exercise of the Warrants to the extent that,
as a result of such exercise, the holder of the Warrant together with the holder’s affiliates, would beneficially own more than
4.99% of the number of shares of Common Stock of the Company outstanding immediately after giving effect to the issuance of the such shares.
In addition, the sum of the aggregate number of shares of common stock that may be issued to all Investors under the August 2022 Securities
is limited to 19.99% of the Company’s then outstanding shares of common stock as of the date of issuance unless Shareholder Approval
(as defined in the SPA) is obtained to issue more than the 19.99%.
In connection with the Offering,
the Company also entered into a registration rights agreement (the “Registration Rights Agreement”) with the Investors pursuant
to which the Company shall prepare and file with the U.S. Securities and Exchange Commission (the “SEC”) a registration statement
(the “Registration Statement”) covering the Note, the Conversion Shares, the Warrant, and the Warrant Shares and any additional
shares of Common Stock issued and issuable in connection with any stock split, dividend or other distribution, recapitalization or similar
event with respect to the foregoing (collectively, the “Registrable Securities”) on or prior to the 90th calendar
day following the Closing Date (the “Filing Date”).
The Company shall use its
best efforts to cause the registration statement covering the Registrable Securities to be declared effective (the “Effectiveness
Date”) by the SEC within one hundred twenty (120) calendar days from the date hereof (or at the earliest possible date if prior
to one hundred twenty (120) calendar days from the date of the Registration Rights Agreement.
In connection with the Offering,
the Company will issue 157 shares (the “Placement Agent Shares”) to Crito Capital LLC.
The Company estimates that
the net proceeds from the transaction will be approximately $1,244,000 after deducting estimated transaction fees and expenses. The
net proceeds received by the Company from the transaction will be used for business development, working capital and other general
corporate purposes.
The
August 2022 Notes contain certain covenants, events of default and triggering events, which would require repayment of the obligations
outstanding pursuant to such instruments. The obligations of the Company pursuant to the August 2022 Notes are secured by certain assets
of the Company pursuant to the Security Agreement dated August 4, 2022, by and among the Company and the holders of the August 2022 Notes.
On
August 11, 2022 and September 12, 2022, the Company entered into additional SPAs with certain additional accredited investors (collectively,
the “Subsequent Investors” and together with the August Investors, the “Investors”), pursuant to which the Company
issued and sold an additional: (i) $1,111,110 in principal amount of the August 2022 Notes, resulting in gross proceeds to the Company
of $1,000,000, exclusive of placement agent fees and other offering expenses; (ii) 556 August 2022 Commitment Shares; and (iii) August
2022 Warrants to purchase 2,384 shares of the Company’s common stock.
On
August 31, 2022, the Company entered into a First Amendment and Waiver with each of the August Investors (the “August Amendment”).
Pursuant to the August Amendment, the exercise price of the August 2022 Warrants was reduced to $300.00 per share and the Commencement
Date (as defined in the SPA) was amended to mean the date on which the Company obtains shareholder approval for the issuance of any shares
of common stock upon exercise of the August 2022 Warrants. The Warrant is exercisable for a period of five (5) years from period commencing
on the Commencement Date (as defined therein) and ending on 5:00 p.m. eastern standard time on the date that is five (5) years after the
date of issuance, at an exercise price of $300.00, subject to adjustment provided therein (including cashless exercise). The Company is
prohibited from effecting an exercise of the Warrant to the extent that, as a result of such exercise, the holder of the Warrant together
with the holder’s affiliates, would beneficially own more than 4.99% of the number of shares of Common Stock of the Company outstanding
immediately after giving effect to the issuance of such shares. In addition, the sum of the aggregate number of shares of common stock
that may be issued under the Warrant is limited to 19.99% of the Company’s then outstanding shares of common stock as of the date
of issuance unless Shareholder Approval (as defined in the Agreement for the Purchase and Sale of Future Receipts) is obtained to issue
more than the 19.99%.
On August 31, 2022, the Company
entered into an Agreement for the Purchase and Sale of Future Receipts with a commercial funding source (the “Funder”) pursuant
to which the Company agreed to sell to the Funder certain future trade receipts in the aggregate amount $288,000 (the “Purchased
Amount”) for gross proceeds to the Company of $200,000, less origination fees of $20,000. Pursuant to the Agreement, the Company
granted the Funder a security interest in all of the Company’s present and future accounts receivable in an amount not to exceed
the Purchased Amount. The Purchased Amount shall be repaid by the Company in 20 weekly installments of approximately $14,400. In connection
with the Agreement, the Company also issued a warrant to purchase 667 shares of the Company’s common stock.
On
April 20, 2023, the Company entered into a securities purchase agreement (the “Purchase Agreement”) with an institutional
investor, pursuant to which the Company agreed to sell to such investor pre-funded warrants (the “Pre-Funded Warrants”) to
purchase up to 39,634 shares of common stock of the Company (the “Common Stock”) at a purchase price of $48.76 per Pre-Funded
Warrant. The Pre-Funded Warrants (and shares of common stock underlying the Pre-Funded Warrants) were offered by the Company pursuant
to its shelf registration statement on Form S-3 (File No. 333-257645), which was declared effective by the Securities and Exchange Commission
on July 13, 2021.
Concurrently
with the sale of the Pre-Funded Warrants, pursuant to the Purchase Agreement in a concurrent private placement, for each Pre-Funded
Warrant purchased by the investor, such investor received from the Company an unregistered warrant (the “Warrant”) to purchase
two shares of Common Stock. The warrants have an exercise price of $34.40 per share, and are exercisable for a three year period. The
Warrant was issued were made pursuant to Section 4(a)(2) of the Securities Act.
On
July 3, 2023, Aditxt, Inc. we entered into a Securities Purchase Agreement (the “First Tranche Securities Purchase Agreement”)
with an accredited investor pursuant to which we issued and sold a secured promissory note in the principal amount of $375,000 (the “First
Tranche Note”) resulting in gross proceeds of $250,000. In connection with the issuance of the Note, we issued 3,906 shares of common
stock (the “First Tranche Commitment Shares”) as a commitment fee to the investor. Pursuant to the Securities Purchase Agreement,
we are obligated to obtain approval of our shareholders (“Shareholder Approval”) with respect to the issuance of any securities
in connection with the Securities Purchase Agreement and the Note in excess of 19.99% of our issued and outstanding shares on the closing
date, which is equal to 33,791 shares of our common stock. The First Tranche Note has a maturity date of December 31, 2023 and is convertible
following Shareholder Approval and the occurrence of an Event of Default (as defined in the Note) at a conversion price of $18.00 per
share.
In
connection with the Securities Purchase Agreement and the issuance of the First Tranche Note, we and certain of our subsidiaries also
entered into a Security Agreement with the investor (the “First Tranche Security Agreement”) pursuant to which we granted
the investor a security interest in certain Collateral (as defined in the First Tranche Security Agreement) to secure our obligations
under the First Tranche Note. In addition, we entered into a Registration Rights Agreement with the investor (the “First Tranche
Registration Rights Agreement”) pursuant to which we agreed to prepare and file with the U.S. Securities and Exchange Commission
a registration statement covering the resale of the First Tranche Commitment Shares and any shares of our common stock issuable upon conversion
of the First Tranche Note within 120 days of the closing date and to have such registration statement declared effective within 150 days
of the closing date.
On
July 3, 2023, we entered into a Business Loan and Security Agreement (the “July Loan Agreement”) with a commercial funding
source (the “July Lender”), pursuant to which we obtained a loan from the July Lender in the principal amount of $215,000,
which includes origination fees of $10,750 (the “July Loan”). Pursuant to the July Loan Agreement, we granted the Lender a
continuing secondary security interest in certain collateral (as defined in the Loan Agreement). The total amount of interest and fees
payable by us to the July Lender under the July Loan will be (i) $322,285 and will be repaid in 13 weekly installments of $24,500 with
a final payment of $3,785 in the fourteenth week.
On
July 11, 2023, we entered into a Subscription and Investment Representation Agreement (the “Subscription Agreement”) with
Amro Albanna, our Chief Executive Officer, who is an accredited investor (the “Purchaser”), pursuant to which we agreed to
issue and sell one (1) share of the Company’s Series C Preferred Stock, par value $0.001 per share (the “Preferred Stock”),
to the Purchaser for $1,000.00 in cash. The sale closed on July 11, 2023.
On
July 11, 2023, we filed a certificate of designation (the “Certificate of Designation”) with the Secretary of State of Delaware,
effective as of the time of filing, designating the rights, preferences, privileges and restrictions of the share of Preferred Stock.
The Certificate of Designation provides that the share of Preferred Stock will have 250,000,000 votes and will vote together with the
outstanding shares of our common stock as a single class exclusively with respect to any proposal to amend the Company’s Amended
and Restated Certificate of Incorporation to effect a reverse stock split of our common stock. The Preferred Stock will be voted, without
action by the holder, on any such proposal in the same proportion as shares of common stock are voted. The Preferred Stock otherwise has
no voting rights except as otherwise required by the General Corporation Law of the State of Delaware.
The
Preferred Stock is not convertible into, or exchangeable for, shares of any other class or series of stock or other securities of the
Company. The Preferred Stock has no rights with respect to any distribution of assets of the Company, including upon a liquidation, bankruptcy,
reorganization, merger, acquisition, sale, dissolution or winding up of the Company, whether voluntarily or involuntarily. The holder
of the Preferred Stock will not be entitled to receive dividends of any kind.
The
outstanding share of Preferred Stock shall be redeemed in whole, but not in part, at any time (i) if such redemption is ordered by the
Board of Directors in its sole discretion or (ii) automatically upon the effectiveness of the amendment to the Certificate of Incorporation
implementing a reverse stock split. Upon such redemption, the holder of the Preferred Stock will receive consideration of $1,000.00 in
cash.
On
July 24, 2023, we entered into a Securities Purchase Agreement (the “Second Tranche Securities Purchase Agreement”) with an
accredited investor pursuant to which the Company issued and sold a secured promissory note in the principal amount of $2,625,000 (the
“Second Tranche Note”) resulting in gross proceeds to the Company of $1,750,000. In connection with the issuance of the Note,
we agreed to issue a total of 27,343 shares of common stock (the “Second Tranche Commitment Shares”) as a commitment fee to
the investor. At the request of the investor, we issued 17,277 Second Tranche Commitment Shares and will issue the remaining 10,066 Second
Tranche Commitment Shares within 120 days, subject to the investor’s discretion. Pursuant to the Second Tranche Securities Purchase
Agreement, we are obligated to obtain approval of our shareholders with respect to the issuance of any securities in connection with the
Second Tranche Securities Purchase Agreement and the Second Tranche Note in excess of 19.99% of our issued and outstanding shares on the
closing date, which is equal to 38,026 shares of the Company’s common stock. The Second Tranche Note has a maturity date of December
31, 2023 and is convertible following shareholder approval and the occurrence of an Event of Default (as defined in the Note) at a conversion
price of $15.60 per share.
In
connection with the Second Tranche Securities Purchase Agreement and the issuance of the Second Tranche Note, we and certain of our subsidiaries
also entered into a Security Agreement with the investor (the “Second Tranche Security Agreement”) pursuant to which we granted
the investor a security interest in certain Collateral (as defined in the Second Tranche Security Agreement) to secure its obligations
under the Second Tranche Note. In addition, we entered into a Registration Rights Agreement with the investor (the “Second Tranche
Registration Rights Agreement”) pursuant to which we agreed to prepare and file with the U.S. Securities and Exchange Commission
a registration statement covering the resale of the Second Tranche Commitment Shares and any shares of our common stock issuable upon
conversion of the Second Tranche Note within 90 days of the closing date and to have such registration statement declared effective within
120 days of the closing date.
On
August 23, 2023, we entered into a Business Loan and Security Agreement (the “August Loan Agreement”) with a commercial funding
source (the “August Lender”) pursuant to which we obtained a loan from the August Lender in the principal amount of $1,400,000,
which will satisfy the outstanding balances on loans that we originally obtained From the August Lender in April 2023 and July 2023, and
includes origination fees of $70,000 (the “August Loan”). Pursuant to the August Loan Agreement, we granted the August Lender
a continuing secondary security interest in certain collateral (as defined in the August Loan Agreement). The total amount of interest
and fees payable by us to the August Lender under the August Loan will be $2,079,000, which will be repaid in 21 weekly installments of
$99,000.
On
August 31, 2023, we entered into a securities purchase agreement (the “Purchase Agreement”) with
an institutional investor for the issuance and sale in a private placement (the “Private Placement”) of (i) pre-funded warrants
(the “Pre-Funded Warrants”) to purchase up to 1,000,000 shares of the Company’s common stock, par value $0.001 (the
“Common Stock”), at an exercise price of $0.001 per share, and (ii) warrants (the “Common Warrants”) to purchase
up to 1,000,000 shares of the Company’s Common Stock at an exercise price of $10.00 per share.
The
Common Warrants are exercisable immediately upon issuance and have a term of exercise equal to five and one-half years from the date of
issuance. The Pre-Funded Warrants are exercisable immediately and may be exercised at any time until the Pre-Funded Warrants are exercised
in full. A holder of Pre-Funded Warrants or Warrants (together with its affiliates) may not exercise any portion of a warrant to the extent
that the holder would own more than 4.99% (or, at the election of the holder 9.99%) of the Company’s outstanding common stock immediately
after exercise.
In
connection with the Private Placement, we entered into a registration rights agreement (the “Registration Rights Agreement”),
dated as of August 31, 2023, with the investor, pursuant to which the Company agreed to prepare and file a registration statement with
the Securities and Exchange Commission (the “SEC”) registering the resale of the shares of Common Stock underlying the Pre-Funded
Warrants and the Common Warrants no later than 15 days after the date of the Registration Rights Agreement, and to use best efforts to
have the registration statement declared effective as promptly as practical thereafter, and in any event no later than 45 days following
the date of the Registration Rights Agreement (or 75 days following the date of the Registration Rights Agreement in the event of a “full
review” by the SEC).
The
Private Placement closed on September 6, 2023. We received net proceeds from the Private Placement of approximately $9 million, after
deducting placement agent fees and expenses and estimated offering expenses payable by us.
H.C.
Wainwright & Co., LLC (“Wainwright”) served as our exclusive placement agent in connection with the Private Placement,
pursuant to those certain engagement letters, dated as of March 27, 2023 and April 25, 2023, as amended, between the Company and Wainwright
(the “Engagement Letter”). Pursuant to the Engagement Letter, we paid Wainwright (i) a total cash fee equal to 7.75% of the
aggregate gross proceeds of the Private Placement, (ii) a management fee of 1.0% of the aggregate gross proceeds of the Private Placement,
(iii) a non-accountable expense allowance of $50,000, and (iv) $100,000 for legal fees and other out-of-pocket expenses. In addition,
we issued to Wainwright or its designees warrants (the “Placement Agent Warrants”) to purchase up to an aggregate of 60,000
shares of Common Stock at an exercise price equal to $12.50 per share. The Placement Agent Warrants are exercisable immediately upon issuance
and have a term of exercise equal to five and one-half years from the date of issuance.
Item 16. Exhibits and Financial Statement Schedules.
(a) Exhibits.
| Exhibit No. |
|
Description |
| 2.1 |
|
Share Exchange Agreement, dated as of December 28, 2021 by and between AiPharma Group Ltd. and Aditxt, Inc. (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on December 28, 2021) |
| 2.2 |
|
Amendment to Share Exchange Agreement by and between AiPharma Group Ltd. and Aditxt, Inc. (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on May 16, 2022) |
| 2.3 |
|
Second Amendment to Share Exchange Agreement by and between AiPharma Group Ltd. and Aditxt, Inc. (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on June 16, 2022) |
| 2.4 |
|
Asset Purchase Agreement dated as of April 19, 2023 by and between the Company and Cellvera Ltd., et al. (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on April 25, 2023) |
| 3.1 |
|
Amended and Restated Certificate of Incorporation (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 3.2 |
|
Certificate of Amendment, dated June 29, 2020 (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q, filed with the SEC on August 13, 2020) |
| 3.3 |
|
Amended and Restated Bylaws (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 3.4 |
|
Certificate of Designation Series A Preferred Stock (incorporated by reference to the Registrant’s Registration Statement on Form S-1 (File No. 333-248491) |
| 3.5 |
|
Certificate of Amendment, filed with the Secretary of State of the State of Delaware on May 24, 2021 (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on May 25, 2021) |
| 3.6 |
|
Certificate of Amendment, dated July 6, 2021 (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 8, 2021) |
| 3.7 |
|
Amendment No. 1 to Amended and Restated Bylaws of Aditxt, Inc. (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 8, 2022) |
| 3.8 |
|
Certificate of Designation of Series B Preferred Stock, dated July 19, 2022 (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 20, 2022) |
| 3.9 |
|
Certificate of Amendment to Certificate of Incorporation of Aditxt, Inc. (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on September 14, 2022) |
| 3.10 |
|
Certificate of Designation of Series C Preferred Stock, dated July 11, 2023 (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 14, 2023) |
| 3.11 |
|
Certificate of Amendment to Certificate of Incorporation of Aditxt, Inc. (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on August 17, 2023) |
| 4.1 |
|
Description of Securities Registered Under Section 12 of the Exchange Act (incorporated by reference to the Registrant’s Annual Report on Form 10-K filed on March 25, 2021) |
| 4.2 |
|
Form the Company’s common stock certificate (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 4.3 |
|
Form of Series A-1 Warrant Agent Agreement (including the terms of the Series A-1 Warrant) (incorporated by reference to the Registrant’s Registration Statement on Form S-1 (File No. 333-248491) |
| 4.4 |
|
Form of Series B-1 Warrant Agent Agreement (including the terms of the Series B-1 Warrant) (incorporated by reference to the Registrant’s Registration Statement on Form S-1 (File No. 333-248491) |
| 4.5 |
|
Form of Warrant (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on August 30, 2021) |
| 4.6 |
|
Form of Pre-Funded Warrant (incorporated by reference to the Registrant’s filed on April 24, 2023) Current Report on Form 8-K |
| 4.7 |
|
Form of Warrant (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on April 24, 2023) |
| 4.8 |
|
Form of Placement Agent Warrant (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on April 24, 2023) |
| 4.9 |
|
Form of Warrant (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on September 6, 2023) |
| 4.10 |
|
Form of Placement Agent Warrant (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on September 6, 2023) |
| 5.1* |
|
Opinion of Sheppard, Mullin, Richter & Hampton LLP |
| 10.1 |
|
Form of Promissory Note issued to Sekris Biomedical, Inc. (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.2 |
|
Warrant, dated March 8, 2018, issued to Sekris Biomedical, Inc. (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.3 |
|
Form of Private Placement Subscription Agreement (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.4 |
|
Patent Licensing Agreement, dated February 3, 2020 (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.5 |
|
Patent and Technology License Agreement, dated March 15, 2018 between Loma Linda University and Aditx Therapeutics, Inc. (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.6 |
|
Amendment Agreement to the Patent and Technology License Agreement, dated July 1, 2020 by and between Loma Linda University and Aditx Therapeutics, Inc. (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q, filed with the SEC on August 13, 2020) |
| 10.7 |
|
2017 Equity Incentive Plan and forms of award agreements thereunder (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.8 |
|
Consulting Agreement, dated March 1, 2018 between Aditx Therapeutics, Inc. and Canyon Ridge Development LLC d/b/a Mission Critical Solutions International (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.9 |
|
Form of July 2018 Securities Purchase Agreement (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.10 |
|
Form of July 2018 Note (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.11 |
|
Form of April 2018 Promissory Note (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.12 |
|
Form of March 2019 Promissory Note (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.13 |
|
Form of October 2019 Securities Purchase Agreement (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.14 |
|
Form of October 2019 Note (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.15 |
|
Form of January 2020 Note Purchase Agreement (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.16 |
|
Form of January 2020 Private Placement Promissory Note (incorporated by reference to the Registrant’s Registration Statement on Form S-1/A (File No. 333-235933) |
| 10.17 |
|
Consulting Agreement by and between the Company and Salveo Diagnostics, Inc., dated November 18, 2020 (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on November 23, 2020) |
| 10.18 |
|
Form of Senior Secured Convertible Promissory Note (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on January 26, 2021) |
| 10.19 |
|
Form of Warrant (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on January 26, 2021) |
| 10.20 |
|
Form of Securities Purchase Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on January 26, 2021) |
| 10.21 |
|
Form of Registration Rights Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on January 26, 2021) |
| 10.22 |
|
Employment Agreement, dated as of February 24, 2021, by and between the Company and Amro Albanna (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on February 26, 2021) |
| 10.23 |
|
2021 Omnibus Equity Incentive Plan (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on February 26, 2021) |
| 10.24 |
|
Lease Agreement, dated as of May 4, 2021, by and between LS Biotech Eight, LLC as Landlord, and Aditxt Therapeutics, Inc., as Tenant (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on May 10, 2021) |
| 10.25 |
|
Form of Securities Purchase Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on August 30, 2021) |
| 10.26 |
|
Placement Agency Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on August 30, 2021) |
| 10.27 |
|
Form of Placement Agent Warrant (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on August 30, 2021) |
| 10.28 |
|
Waiver and Defeasance Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on August 30, 2021) |
| 10.29 |
|
Secured Credit Agreement, dated as of August 27, 2021, by and among AiPharma, AiPharma Holdings Limited, AiPharma Asia Limited and the Company (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on November 15, 2021) |
| 10.30 |
|
Security Agreement, dated as of August 27, 2021 by and between AiPharma Asia Limited and the Company (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on November 15, 2021) |
| 10.31 |
|
Security Agreement, dated as of August 27, 2021 by and between AiPharma Limited and the Company (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on November 15, 2021) |
| 10.32 |
|
Security Agreement – AiPharma Limited and Aditxt (BVI Law) (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on November 15, 2021) |
| 10.33 |
|
Floating Charge (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on November 15, 2021) |
| 10.34 |
|
Transaction Agreement, dated as of October 4, 2021, by and between the Company and AiPharma Global Holdings LLC (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on November 15, 2021) |
| 10.35 |
|
First Amendment to Secured Credit Agreement with AiPharma Global Holdings LLC dated October 18, 2021 (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on November 15, 2021) |
| 10.36 |
|
Second Amendment to Secured Credit Agreement with AiPharma Global Holdings LLC dated October 27, 2021(incorporated by reference to the Registrant’s Annual Report on Form 10-K filed on March 31, 2022) |
| 10.37 |
|
Employment Agreement, dated as of November 14, 2021 between Aditxt, Inc. and Amro Albanna, Chief Executive Officer (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on November 15, 2021) |
| 10.38 |
|
Employment Agreement, dated as of November 14, 2021 between Aditxt, Inc. and Corinne Pankovcin, President and Secretary (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on November 15, 2021) |
| 10.39 |
|
Employment Agreement, dated as of November 14, 2021 between Aditxt, Inc. and Thomas Farley, Chief Financial Officer (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on November 15, 2021) |
| 10.40 |
|
Employment Agreement, dated as of November 14, 2021 between Aditxt, Inc. and Shahrokh Shabahang, Chief Innovation Officer (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on November 15, 2021) |
| 10.41 |
|
Employment Agreement, dated as of November 14, 2021 between Aditxt, Inc. and Rowena Albanna, Chief Operating Officer (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on November 15, 2021) |
| 10.42 |
|
Form of Warrant Reduction and Release Agreement dated as of November 24, 2021 (incorporated by reference to the Registrant’s Annual Report on Form 10-K filed on March 31, 2022) |
| 10.43 |
|
First Amendment to Transaction Agreement dated November 30, 2021, by and between the Company and AiPharma Global Holdings LLC (incorporated by reference to the Registrant’s Annual Report on Form 10-K filed on March 31, 2022) |
| 10.44 |
|
Third Amendment to Secured Credit Agreement dated November 30, 2021, by and among AiPharma, AiPharma Holdings Limited, AiPharma Asia Limited and the Company (incorporated by reference to the Registrant’s Annual Report on Form 10-K filed on March 31, 2022) |
| 10.45 |
|
Second Amendment to Transaction Agreement dated December 7, 2021, by and between the Company and AiPharma Global Holdings LLC (incorporated by reference to the Registrant’s Annual Report on Form 10-K filed on March 31, 2022) |
| 10.46 |
|
Secured Credit Agreement, dated as of December 8, 2021, by and among the Company and the Target Company (incorporated by reference to the Registrant’s Annual Report on Form 10-K filed on March 31, 2022) |
| 10.47 |
|
Third Amendment to Transaction Agreement dated December 17, 2021, by and between the Company and AiPharma Global Holdings LLC (incorporated by reference to the Registrant’s Annual Report on Form 10-K filed on March 31, 2022) |
| 10.48 |
|
Fifth Amendment to Secured Credit Agreement dated December 22, 2021, by and among AiPharma, AiPharma Holdings Limited, AiPharma Asia Limited and the Company (incorporated by reference to the Registrant’s Annual Report on Form 10-K filed on March 31, 2022) |
| 10.49 |
|
Sixth Amendment to Secured Credit Agreement dated December 28, 2021, by and among AiPharma, AiPharma Holdings Limited, AiPharma Asia Limited and the Company (incorporated by reference to the Registrant’s Annual Report on Form 10-K filed on March 31, 2022) |
| 10.50 |
|
Employment Agreement between Aditxt, Inc. and Matthew Shatzkes, Chief Legal Officer and General Counsel (incorporated by reference to the Registrant’s Annual Report on Form 10-K filed on March 31, 2022) |
| 10.51 |
|
Forbearance Agreement and Seventh Amendment to Secured Credit Agreement dated as of February 14, 2022 by and among the Company, Cellvera Global Holdings LLC, Cellvera Holdings Ltd., Cellvera Asia Limited (incorporated by reference to the Registrant’s Annual Report on Form 10-K filed on March 31, 2022) |
| 10.52 |
|
Fourth Amendment to Transaction Agreement dated December 22,2021, by and between the Company and AiPharma Global Holdings LLC (incorporated by reference to the Registrant’s Annual Report on Form 10-K filed on March 31, 2022) |
| 10.53 |
|
Series C Warrant Agent Agreement (incorporated by reference to the Registrant’s Annual Report on Form 10-K/A filed on April 15, 2022) |
| 10.54 |
|
Form of Placement Agent Warrant dated January 25, 2021 (incorporated by reference to the Registrant’s Annual Report on Form 10-K/A filed on April 15, 2022) |
| 10.55 |
|
Forbearance Agreement and Eighth Amendment to Secured Credit Agreement dated as of March 31, 2022 (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on May 16, 2022) |
| 10.56 |
|
Security Agreement between Cellvera Holdings and Aditxt, Inc. dated as of March 31, 2022 (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on May 16, 2022) |
| 10.57 |
|
Security Agreement between Cellvera Development LLC and Aditxt, Inc. dated as of March 31, 2022 (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on May 16, 2022) |
| 10.58 |
|
Security Agreement between Cellvera Global Holdings and Aditxt, Inc. dated as of March 31, 2022 (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on May 16, 2022) |
| 10.59 |
|
Amended and Restated Security Agreement between Cellvera Asia Limited and Aditxt, Inc. dated as of March 31, 2022 (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on May 16, 2022) |
| 10.60 |
|
Revenue Sharing Agreement by and among Aditxt, Inc., Cellvera Global Holdings LLC and Cellvera Asia Limited dated as of March 31, 2022 (incorporated by reference to the Registrant’s Quarterly Report on Form 10-Q filed on May 16, 2022) |
| 10.61 |
|
Form of Agreement for the Purchase and Sale of Future Receipts (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on June 3, 2022) |
| 10.62 |
|
Amendment No. 1 to Series C Warrant Agent Agreement dated June 15, 2022 (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on June 15, 2022) |
| 10.63 |
|
Inducement Offer to Exercise Series C Common Stock Purchase Warrants (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on June 15, 2022) |
| 10.64 |
|
Form of New Warrant (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on June 15, 2022) |
| 10.65 |
|
Form of Placement Agent Warrant (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on June 15, 2022) |
| 10.66 |
|
Subscription and Investment Representation Agreement, dated July 19, 2022 (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 20, 2022) |
| 10.67 |
|
Unsecured Promissory Note dated July 21, 2022 (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 26, 2022) |
| 10.68 |
|
Form of Securities Purchase Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on August 10, 2022) |
| 10.69 |
|
Form of August 2022 Note (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on August 10, 2022) |
| 10.70 |
|
Form of August 2022 Warrant (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on August 10, 2022) |
| 10.71 |
|
Form of Registration Rights Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on August 10, 2022) |
| 10.72 |
|
Form of Security Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on August 10, 2022) |
| 10.73 |
|
Form of First Amendment and Waiver effective as of August 31, 2022 (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on September 7, 2022) |
| 10.74 |
|
Form of Warrant (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on September 7, 2022) |
| 10.75 |
|
Form of Securities Purchase Agreement (incorporated by reference to the Registrant’s Registration Statement on Form S-1 filed on September 15, 2022) |
| 10.76 |
|
Form of Warrant (incorporated by reference to the Registrant’s Registration Statement on Form S-1 filed on September 15, 2022) |
| 10.77 |
|
Form of Placement Agent’s Warrant (incorporated by reference to the Registrant’s Registration Statement on Form S-1 filed on September 15, 2022) |
| 10.78 |
|
Form of Pre-Funded Warrant (incorporated by reference to the Registrant’s Registration Statement on Form S-1 filed on September 15, 2022) |
| 10.79 |
|
Amendment No. 2 to Series C Warrant Agent Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on December 23, 2022) |
| 10.80 |
|
Form of Amended and Restated Unit Purchase Option (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on December 23, 2022) |
| 10.81 |
|
Form of Consulting Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on March 21, 2023) |
| 10.82 |
|
Form of Business Loan and Security Agreement dated April 4, 2023(incorporated by reference to the Registrant’s Current Report on Form 8-K filed on April 7, 2023) |
| 10.83 |
|
Form of Securities Purchase Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on April 24, 2023) |
| 10.84 |
|
Form of Unsecured Promissory Note (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on April 25, 2023) |
| 10.85 |
|
Form of Business Loan and Security Agreement, dated April 24, 2023 (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on April 25, 2023) |
| 10.86 |
|
Form of Agreement for the Purchase and Sale of Future Receipts (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on May 31, 2023) |
| 10.87 |
|
Unsecured Promissory Note dated May 25, 2023 (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on May 31, 2023) |
| 10.88 |
|
Form of Unsecured Promissory Note (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on June 16, 2023) |
| 10.89 |
|
At The Market Offering Agreement dated December 20, 2022 between Aditxt, Inc. and H.C. Wainwright & Co., LLC (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on December 20, 2022) |
| 10.90 |
|
Form of Securities Purchase Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 7, 2023) |
| 10.91 |
|
Form of Note (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 7, 2023) |
| 10.92 |
|
Form of Security Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 7, 2023) |
| 10.93 |
|
Form of Registration Rights Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 7, 2023) |
| 10.94 |
|
Form of Business Loan and Security Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 7, 2023) |
| 10.95 |
|
Subscription and Investment Representation Agreement, dated July 11, 2023, by and between Aditxt, Inc., and the purchaser signatory thereto (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 14, 2023) |
| 10.96 |
|
Separation Agreement and General Release by and between Matthew Shatzkes and Aditxt, Inc. (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 27, 2023) |
| 10.97 |
|
Form of Securities Purchase Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 28, 2023) |
| 10.98 |
|
Form of Note (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 28, 2023) |
| 10.99 |
|
Form of Security Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 28, 2023) |
| 10.100 |
|
Form of Registration Rights Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on July 28, 2023) |
| 10.101 |
|
Amendment to Separation Agreement and General Release dated August 15, 2023 (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on August 21, 2023) |
| 10.102 |
|
Form of Business Loan and Security Agreement dated August 23, 2023 (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on August 28, 2023) |
| 10.103 |
|
Form of Securities Purchase Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on September 6, 2023) |
| 10.104 |
|
Form of Registration Rights Agreement (incorporated by reference to the Registrant’s Current Report on Form 8-K filed on September 6, 2023) |
| 23.1* |
|
Consent of dbbmckennon, independent registered public accounting firm |
| 23.2* |
|
Consent of Sheppard, Mullin, Richter & Hampton LLP (Included in Exhibit 5.1) |
| 24.1 |
|
Power of Attorney |
| 107* |
|
Filing Fee Table |
| † | Executive
Compensation Plan or Agreement |
| # | Portions
of this exhibit (indicated by asterisks) have been redacted in compliance with Regulation S-K Item 601(b)(10)(iv). |
Item 17. Undertakings.
The undersigned registrant hereby undertakes:
| |
1. |
To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement to: |
| |
i. |
Include any prospectus required by Section 10(a)(3) of the Securities Act. |
| |
ii. |
Reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the SEC pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than a 20% change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement; and |
| |
iii. |
Include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information in the registration statement; |
provided, however,
that paragraphs (1)(i), (ii) and (iii) do not apply if the information required to be included in a post-effective amendment by those
paragraphs is contained in reports filed with or furnished to the Commission by the registrant pursuant to Section 13 or Section 15(d)
of the Securities Exchange Act of 1934 that are incorporated by reference in the registration statement, or is contained in a form of
prospectus filed pursuant to Rule 424(b) that is part of the registration statement.
| |
2. |
That, for the purpose of determining any liability under the Securities Act, each such post-effective amendment shall be deemed to be a new registration statement relating to securities offered therein, and the offering of the securities at that time shall be deemed to be the initial bona fide offering thereof; |
| |
3. |
To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering; |
| |
4. |
That, for the purpose of determining liability under the Securities Act to any purchaser, each prospectus filed pursuant to Rule 424(b) as part of a registration statement relating to an offering, other than registration statements relying on Rule 430B or other than prospectuses filed in reliance on Rule 430A, shall be deemed to be part of and included in the registration statement as of the date it is first used after effectiveness. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such date of first use. |
The undersigned registrant
hereby undertakes that, for purposes of determining any liability under the Securities Act of 1933, each filing of the registrant’s
annual report pursuant to section 13(a) or section 15(d) of the Securities Exchange Act of 1934 (and, where applicable, each filing of
an employee benefit plan’s annual report pursuant to section 15(d) of the Securities Exchange Act of 1934) that is incorporated
by reference in the registration statement shall be deemed to be a new registration statement relating to the securities offered therein,
and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof.
Insofar as indemnification
for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant pursuant
to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the SEC such indemnification is against
public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against
such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of
the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person
in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled
by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public
policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
SIGNATURES
Pursuant to the requirements
of the Securities Act of 1933, as amended, the registrant has duly caused this Registration Statement on Form S-1 to be signed on
its behalf by the undersigned, thereunto duly authorized in the City of Richmond, State of Virginia, on the 15th day of September,
2023.
| |
Aditxt, Inc. |
| |
|
| |
By: |
/s/ Amro Albanna |
| |
|
Name: |
Amro Albanna |
| |
|
Title: |
Chief Executive Officer |
POWER OF ATTORNEY
We, the undersigned officers
and directors of Aditxt, Inc., hereby severally constitute and appoint Amro Albanna, our true and lawful attorney-in-fact and agent, with
full power of substitution and resubstitution, for us and in our stead, in any and all capacities, to sign any and all amendments (including
post-effective amendments) to this Registration Statement and all documents relating thereto, and to file the same, with all exhibits
thereto, and other documents in connection therewith, with the Securities and Exchange Commission, granting unto said attorney-in-fact
and agent full power and authority to do and perform each and every act and thing necessary or advisable to be done in and about the premises,
as fully to all intents and purposes as he might or could do in person, hereby ratifying and confirming all that said attorney-in-fact
and agent or his substitute or substitutes, may lawfully do or cause to be done by virtue thereof.
WITNESS our hands and common
seal on the dates set forth below.
Pursuant to the requirements
of the Securities Act of 1933, as amended, this Registration Statement on Form S-1 has been signed by the following persons in the
capacities and on the dates indicated below.
| Signature |
|
Title |
|
Date |
| |
|
|
|
|
| /s/
Amro Albanna |
|
Chief Executive Officer
|
|
September
15, 2023 |
| Amro Albanna |
|
(Principal Executive Officer) |
|
|
| |
|
|
|
|
| /s/
Thomas J. Farley |
|
Chief Financial Officer
|
|
September
15, 2023 |
| Thomas J. Farley |
|
(Principal Financial and Accounting Officer) |
|
|
| |
|
|
|
|
| /s/ Brian
Brady |
|
Director |
|
September
15, 2023 |
| Brian Brady |
|
|
|
|
| |
|
|
|
|
| /s/ Namvar
Kiaie |
|
Director |
|
September
15, 2023 |
| Namvar Kiaie |
|
|
|
|
| |
|
|
|
|
| /s/
Jeffrey W. Runge |
|
Director |
|
September
15, 2023 |
| Jeffrey W. Runge, M.D. |
|
|
|
|
| |
|
|
|
|
| /s/
Shahrokh Shabahang |
|
Chief Innovation Officer
and Director |
|
September 15, 2023 |
| Shahrokh Shabahang |
|
|
|
|
II-13
Exhibit 5.1
 |
Sheppard, Mullin, Richter & Hampton LLP
30 Rockefeller Plaza
New York, New York 10112-0015
212.653.8700 main
212.653.8701 fax
www.sheppardmullin.com |
September 15, 2023
VIA EDGAR
Aditxt, Inc.
737 N. Fifth Street, Suite 200
Richmond, VA 23219
| Re: | Registration Statement on Form S-1 |
Ladies and Gentlemen:
We have acted as counsel to Aditxt, Inc., a Delaware
corporation (the “Company”), in connection with the preparation and filing by the Company of a Registration
Statement on Form S-1 (the “Registration Statement”) with the U.S. Securities and Exchange Commission (the “Commission”)
under the Securities Act of 1933, as amended (the “Securities Act”), on or about the date hereof, with respect
to the resale from time to time by the selling stockholders of the Company, as detailed in the Registration Statement (collectively, the
“Selling Stockholders”), of up to 2,060,000 shares of the Company’s common stock, par value $0.001 per
share (“Common Stock”), which consists of (a) up to an aggregate of 1,000,000 shares of Common Stock that are
issuable upon exercise of unregistered pre-funded warrants (the “Pre-Funded Warrants”), (b) up to an aggregate
of 1,000,000 shares of Common Stock that are issuable upon exercise of warrants (the “Common Warrants”), and
(c) up to 60,000 shares of Common Stock that are issuable upon the exercise of certain placement agent warrants (the “Placement
Agent Warrants”, and, collectively with the Pre-Funded Warrants and the Common Warrants, the “Warrants”)
issued to designees of H.C. Wainwright & Co., LLC, the Company’s
placement agent (the “Placement Agent”) pursuant to an
engagement letter, dated as of March 27, 2023 and April 25, 2023 (the “Engagement Letter”). The shares of Common
Stock issuable upon exercise of the Warrants described in clauses (a) through (c) above are referred to herein as the “Warrant
Shares”.
This opinion is being furnished in accordance
with the requirements of Item 601(b)(5)(i) of Regulation S-K.
In connection with this opinion, we have reviewed
and relied upon the following:
| ● | the Registration Statement; |
| ● | the Securities Purchase Agreement
dated August 31, 2023; |
| ● | the form of Pre-Funded Warrant; |
| ● | the form of Placement Agent
Warrant; |
| ● | the Amended and Restated Certificate
of Incorporation of the Company in effect on the date hereof; |
| ● | the Amended and Restated Bylaws
of the Company in effect on the date hereof; |
| ● | the resolutions of the Board
of Directors of the Company, adopted on August 31, 2023 authorizing/ratifying the execution and delivery of the Securities Purchase Agreement,
the issuance and sale of the Pre-Funded Warrants, the Warrants and the Warrant Shares; and |
| ● | such other documents, records,
certificates, memoranda and other instruments as we deem necessary as a basis for this opinion. |
In connection with the issuance
of this opinion letter, we have examined originals or copies, certified or otherwise identified to our satisfaction, of such records of
the Company and such agreements, certificates and receipts of public officials, certificates of officers or other representatives of the
Company and others, and such other documents as we have deemed necessary or appropriate as a basis for the opinions stated below. As to
any facts relevant to the opinions stated herein that we did not independently establish or verify, we have relied upon statements and
representations of officers and other representatives of the Company and of public officials.
In our examination, we have
assumed (a) the genuineness of all signatures, including endorsements, (b) the legal capacity and competency of all natural persons and,
with respect to all parties to agreements or instruments relevant hereto other than the Company, that such parties had the requisite power
and authority (corporate or otherwise) to execute, deliver and perform such agreements or instruments, that such agreements or instruments
have been duly authorized by all requisite action (corporate or otherwise), executed and delivered by such parties and that such agreements
or instruments are the valid, binding and enforceable obligations of such parties, (c) the authenticity of all documents submitted to
us as originals, (d) the conformity to original documents of all documents submitted to us as facsimile, electronic, certified or photostatic
copies, and the authenticity of the originals of such copies; (e) the accuracy, completeness and authenticity of certificates of public
officials; (f) the truth, accuracy and completeness of the information, representations and warranties contained in the instruments, documents,
certificates and records we have reviewed; and (g) the legal capacity for all purposes relevant hereto of all natural persons and, with
respect to all parties to agreements or instruments relevant hereto other than the Company, that such parties had the requisite power
and authority (corporate or otherwise) to execute, deliver and perform such agreements or instruments, that such agreements or instruments
have been duly authorized by all requisite action (corporate or otherwise), executed and delivered by such parties and that such agreements
or instruments are the valid, binding and enforceable obligations of such parties.
Based upon the foregoing and
subject to the qualifications and assumptions stated herein, we are of the opinion that:
1. The Warrant Shares have
been duly authorized by all necessary corporate action on the part of the Company and, assuming a sufficient number of authorized but
unissued shares of Common Stock are available for issuance when the Warrants are exercised, the Warrant Shares, when and if issued, delivered
and paid for in accordance with the terms of the respective Warrants, will be validly issued, fully paid and nonassessable.
Our
opinion set forth in paragraph 1 above is subject to (i) the effects of bankruptcy, insolvency, fraudulent conveyance, reorganization,
moratorium and other similar laws relating to or affecting creditors’ rights generally, (ii) general equitable principles (whether
considered in a proceeding in equity or at law) and (iii) an implied covenant of good faith and fair dealing.
This opinion letter is rendered
solely in connection with the registration of the Warrant Shares for resale by the Selling Stockholders under the Registration Statement.
This opinion letter is rendered as of the date hereof, and we assume no obligation to advise you or any other person with regard to any
change after the date hereof in the circumstances or the law that may bear on the matters set forth herein after the effectiveness of
the Registration Statement, even if the change may affect the legal analysis or a legal conclusion or other matters in this opinion letter.
The opinion we render herein
is limited to those matters governed by New York law as of the date hereof and we disclaim any obligation to revise or supplement the
opinion rendered herein should the above-referenced laws be changed by legislative or regulatory action, judicial decision, or otherwise.
We express no opinion as to whether, or the extent to which, the laws of any particular jurisdiction apply to the subject matter hereof.
We express no opinion as to matters governed by any laws other than New York law.
This opinion letter is rendered
as of the date first written above, and we disclaim any obligation to advise you of facts, circumstances, events, or developments that
hereafter may be brought to our attention or that may alter, affect, or modify the opinion expressed herein.
We hereby consent to the filing
of this opinion as an exhibit to the Registration Statement. We also hereby consent to the reference to our firm under the heading “Legal
Matters” in the Registration Statement. In giving this consent, we do not thereby admit that we are within the category of persons
whose consent is required under Section 7 of the Securities Act or the General Rules and Regulations under the Securities Act. It is understood
that this opinion is to be used only in connection with the offer and sale of the Warrant Shares being registered while the Registration
Statement is effective under the Securities Act.
| |
Respectfully submitted, |
| |
|
| |
/s/ Sheppard, Mullin, Richter & Hampton LLP |
| |
|
| |
SHEPPARD, MULLIN, RICHTER & HAMPTON LLP |
-3-
Exhibit
23.1

Consent
of Independent Registered Public Accounting Firm
We
consent to the incorporation by reference, in this Registration Statement on Form S-1, of our report dated April 17, 2023, related
to the financial statements of Aditxt, Inc (the “Company”) as of December 31, 2022 and 2021, and for the years then ended,
which includes an explanatory paragraph regarding the substantial doubt about the Company’s ability to continue as a going concern.
We also consent to the reference to us under the heading “Experts” in such Registration Statement.
/s/
dbbmckennon
Newport
Beach, California
September
15, 2023

Exhibit 107
Calculation of Filing Fee Tables
FORM S-1
(Form Type)
ADITXT, INC.
(Exact Name of Registrant as Specified in its Charter)
Table 1: Newly Registered Securities
| | |
Security
Type | |
Security
Class
Title | |
Fee
Calculation
Rule | | |
Amount
Registered(1) | | |
Proposed Maximum Offering Price per Share (2) | | |
Maximum
Aggregate
Offering
Price | | |
Amount of
Registration
Fee | |
| Fees to Be Paid | |
Equity | |
Common Stock, $0.001 par value (2) | |
457(c) | | |
| 2,060,000 | | |
$ | 23.88 | | |
$ | 49,192,800 | | |
$ | 5,421.05 | |
| | |
| |
| |
| | |
| | | |
| | | |
| | | |
| | |
| | |
| |
| |
| | |
| | | |
| | | |
| | | |
| | |
| | |
| |
| |
| | |
| | | |
| | | |
| | | |
| | |
| | |
| |
| |
| | |
| | | |
| | | |
| | | |
| | |
| | |
| |
| |
| | |
| | | |
| | | |
| | | |
| | |
| | |
| |
| |
| | |
| | | |
| | | |
| | | |
| | |
| | |
| |
| |
| | |
| | | |
| | | |
| | | |
| | |
| | |
Total Offering Amounts | | |
| | | |
| | | |
| | | |
$ | 5,421.05 | |
| | |
Total Fees Previously Paid | | |
| | | |
| | | |
| | | |
$ | 0 | |
| | |
Total Fee Offsets | | |
| | | |
| | | |
| | | |
$ | 0 | |
| | |
Net Fee Due | | |
| | | |
| | | |
| | | |
$ | 5,421.05 | |
| (1) | Pursuant
to Rule 416 under the Securities Act of 1933, as amended, or the Securities Act, this registration statement also covers any additional
securities that may be offered, issued or become issuable in connection with any stock split, stock dividend or similar transaction or
pursuant to anti-dilution provisions of any of the securities. |
| (2) | Estimated
solely for the purpose of calculation of the registration fee pursuant to Rule 457(c) under the Securities Act based on a per share price
of $23.88, the average of the high and low reported sales prices of the registrant’s common stock on the Nasdaq Capital Market
on September 12, 2023. |
Aditxt (NASDAQ:ADTX)
Historical Stock Chart
From Jun 2024 to Jul 2024
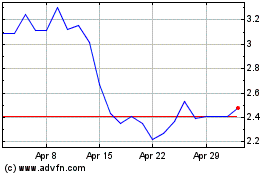
Aditxt (NASDAQ:ADTX)
Historical Stock Chart
From Jul 2023 to Jul 2024