Distribution agreements signed for the UK
May 03 2023 - 2:30AM

Distribution agreements signed for the
UK
- Direct distribution
in Scotland
- Indirect
distribution in England and Wales
|
| |
|
|
Éragny-sur-Oise,
Fleurieux-sur-l'Arbresle,
May
3rd,
2023 at 8:30
a.m. CET - Safe (FR001400F1V2 - ALSAF), a
company specializing in the design, manufacture and marketing of
ready-to-use technologies for back surgery, particularly safe for
spinal fractures treated in emergency (the "Company") announces
strategic sales agreement for the United Kingdom area.
Since acquiring its distributor in 2018, Safe
Orthopaedics has a direct sales force and is seeing long-term
customer adoption in Scotland.
Today, the company announces a dual distribution
agreement in the United Kingdom. In Scotland, the Safe Orthopaedics
sales team is now strengthened by local agents to provide enhanced
sales support to existing customers and to approach new surgery
centers.
In England and Wales, distribution will be
carried out indirectly. Contrary to England, Safe Orthopaedics had
never initiated a commercial approach in Wales. These direct and
indirect distributions are contracted with the same player, to
facilitate daily interactions with the Safe Orthopaedics sales
force.
Pierre
Dumouchel, Chairman of
Safe Group, said: " Our presence in the UK for several
years now has prompted us to set up a hybrid organization that
takes into account the local specificities and will enable us to
maintain double-digit growth in the region. The reinforcement of
our commercial resources in the UK aims to support an already
significant growth of 19% in 2022 and +25% in Q1 2023 and to
maximize the commercial contribution”.
About Safe GroupSafe Group is a
French medical technology group that brings together Safe
Orthopaedics, a pioneer in ready-to-use technologies for spine
pathologies, and Safe Medical (formerly LCI Medical), a medical
device subcontractor for orthopaedic surgery. The group employs
approximately 150 people.
Safe Orthopaedics develops and manufactures kits
combining sterile implants and single-use instruments, available at
any time to the surgeon. These technologies are part of a minimally
invasive approach aimed at reducing the risks of contamination and
infection, in the interest of the patient and with a positive
impact on hospitalization times and costs. Protected by 18 patent
families, SteriSpineTM kits are CE marked and FDA approved. Safe
Orthopaedics is headquartered in the Paris region (95610
Eragny-sur-Oise) and has subsidiaries in the United Kingdom,
Germany, the United States, and the Lyon region
(Fleurieux-sur-l'Arbresle).
For more information:
http://www.safeorthopaedics.com/
Safe Medical produces implantable medical
devices and ready-to-use instruments. It has an innovation center
and two production sites in France (Fleurieux-sur-l'Arbresle,
69210) and in Tunisia, offering numerous industrial services:
design, industrialization, machining, finishing and sterile
packaging. Supported by the French stimulus plan in 2020, the
company invests in additive printing and will be operational in
2022 on this new technology.
For more information:
http://www.safemedical.fr/
Contacts
Safe
GroupFrançois-Henri Reynaud
Chief
Financial and Administrative Officer
Tél.
: +33 (0)1 34 21 50
00 investors@safeorthopaedics.com
- 20230503 - PR_agent et distrib UK vDef
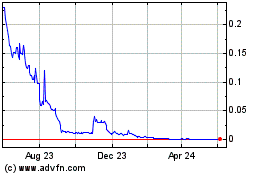
SAFE (EU:ALSAF)
Historical Stock Chart
From Jun 2024 to Jul 2024
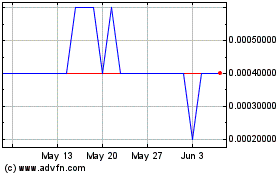
SAFE (EU:ALSAF)
Historical Stock Chart
From Jul 2023 to Jul 2024