AstraZeneca Gets EU Marketing Authorization Recommendation
July 26 2021 - 2:37AM
Dow Jones News
By Sabela Ojea
AstraZeneca PLC said Monday that its Ultomiris drug has been
recommended for marketing authorization in the European Union by
the EMA Committee for Medicinal Products for Human Use, or
CHMP.
The pharmaceutical major said the use of the drug has been
expanded to include children and adolescents with paroxysmal
nocturnal haemoglobinuria.
The opinion is based on results from an Ultomiris phase 3 trial,
which showed an established efficacy and safety profile with
reduced treatment burden for children with PNH and their families,
the FTSE 100 listed company added.
Write to Sabela Ojea at sabela.ojea@wsj.com; @sabelaojeaguix
(END) Dow Jones Newswires
July 26, 2021 02:27 ET (06:27 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
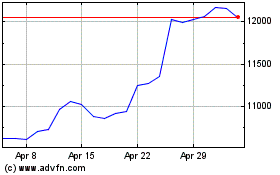
Astrazeneca (LSE:AZN)
Historical Stock Chart
From Aug 2024 to Sep 2024
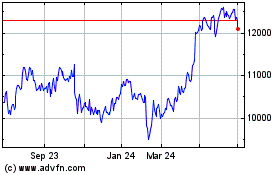
Astrazeneca (LSE:AZN)
Historical Stock Chart
From Sep 2023 to Sep 2024