GAITHERSBURG, Md., Sept. 29, 2015 /PRNewswire/ -- Novavax,
Inc., (Nasdaq: NVAX) a clinical-stage vaccine company focused on
the discovery, development and commercialization of recombinant
nanoparticle vaccines and adjuvants, today announced positive
top-line data from a Phase 2 clinical trial of its RSV F-protein
recombinant nanoparticle vaccine candidate (RSV F Vaccine) to
protect infants via maternal immunization. Novavax also announced
today that it has been awarded a grant of up to $89 million by the Bill & Melinda Gates
Foundation to support development of the RSV F Vaccine Phase 3
clinical trial in pregnant women, planned to initiate during the
first quarter of 2016.
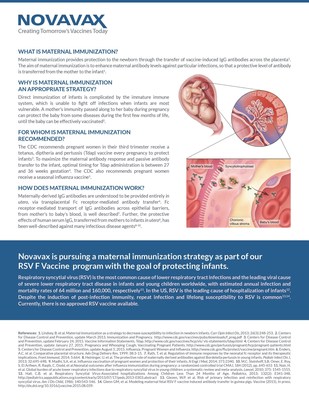
The purpose of the Phase 2 trial was to evaluate the safety and
immunogenicity of the RSV F Vaccine in healthy pregnant women. The
trial also assessed the transplacental transfer of maternal
antibodies induced by the vaccine, the impact of maternal
immunization on infant safety during the first year of life and
RSV-specific antibody levels through the infants' first six months
of life.
The trial was a randomized, blinded, placebo-controlled Phase 2
trial which enrolled 50 healthy pregnant women in their third
trimester. Women were randomized to receive either placebo or 120
micrograms of RSV F Vaccine adjuvanted with 0.4 mg of aluminum
phosphate. They were followed through the remainder of their
pregnancies, delivery and for an additional 180 days postpartum to
assess safety and immunogenicity as measured by serum RSV anti-F
IgG, microneutralizing, and palivizumab-competing antibody (PCA)
titers. Upon delivery, umbilical cord blood samples were obtained
to determine the titers of RSV PCA, anti-F IgG, and
microneutralizing titers in their infants. Additional serum samples
from the infant participants were obtained over the following six
month period to provide a preliminary estimate of the half-life of
vaccine-induced maternal antibodies.
Women in the vaccinated group demonstrated a geometric mean
14-fold rise in anti-F IgG, 29-fold rise in PCA, and 2-fold rise in
microneutralization titers. In contrast, women who received placebo
demonstrated no significant change in their antibody levels. At
delivery, the geometric mean anti-F IgG antibody titer in immunized
women was 7,244 Elisa Units (EU) and on average, the infants'
antibody titer equaled 100% of the mothers' anti-F IgG antibody
titer. The geometric mean PCA antibody titer was 212 micrograms/mL
in immunized mothers and the average PCA antibody transfer within
the mother-infant pairs was 90%. Finally, geometric mean
microneutralizing antibody titers in immunized mothers were 759 and
481 micrograms/mL for RSV/A and RSV/B respectively, and the average
microneutralizing antibody transfer within the mother-infant pairs
was 90% for RSV/A and 100% for RSV/B. The estimated half-lives of
infant PCA, anti-F IgG, RSV/A and RSV/B microneutralizing
antibodies, based on data through day 60, were 41, 30, 36 and 34
days, respectively.
"Maternal immunization is an elegant and accepted approach to
protect infants in the first few months of life, the time period
when they are most vulnerable to RSV," said Gregory Glenn, M.D., Senior Vice President,
Research and Development. "These data are consistent with immune
responses observed in our earlier trials of women of child-bearing
age. As anticipated, infants received a significant bolus of all
anti-RSV antibodies via transplacental transfer. Half of all
hospitalizations due to RSV occur within the first three months of
life and, based on these data, the RSV F Vaccine demonstrates the
potential to protect infants when they are most at risk of
infection."
"These data illustrate the promise of our RSV F Vaccine to
protect infants via maternal immunization, demonstrating robust
immune responses in women with antibody transfer to infants. We
look forward to advancing our maternal immunization program to a
Phase 3 clinical trial with the support of the Bill & Melinda
Gates Foundation," said Stanley C.
Erck, President and CEO. "We have now delivered positive
top-line data from multiple clinical trials this quarter, including
two from our RSV F Vaccine program. These announcements are not
only significant achievements for our company, but important
contributions to the field. We look forward to sustaining this
momentum in the coming months, as we intend to initiate two pivotal
Phase 3 trials."
A fact sheet on maternal immunization is available at the
Novavax website,
http://novavax.com/download/files/pipeline/151_Novavax_FactSheet_FIN_D_9x10.pdf
About RSV
Respiratory syncytial virus (RSV) is the most common cause of
lower respiratory tract infections and the leading viral cause of
severe lower respiratory tract disease in infants and young
children worldwide, with estimated annual infection and mortality
rates of 64 million and 160,000, respectively1. In the
US, RSV is the leading cause of hospitalization of
infants2. Despite the induction of post-infection
immunity, repeat infection and lifelong susceptibility to RSV is
common3,4. Currently, there is no approved RSV vaccine
available. Palivizumab is a monoclonal antibody, licensed and sold
by MedImmune as Synagis®, that targets the RSV F protein
and is used for prophylaxis against RSV disease in high risk
infants.
About Novavax
Novavax, Inc. (Nasdaq:NVAX) is a clinical-stage vaccine company
committed to delivering novel products to prevent a broad range of
infectious diseases. Our recombinant nanoparticles and Matrix-M™
adjuvant technology are the foundation for groundbreaking
innovation that improves global health through safe and effective
vaccines. Additional information about Novavax is available on the
company's website, novavax.com.
References:
- Nair, H. et al. Global burden of acute lower respiratory
infections due to respiratory syncytial virus in young children: a
systematic review and meta-analysis. Lancet, 2010; 375:
1545-1555.
- Hall, C.B. et al. Respiratory Syncytial Virus-Associated
hospitalizations Among Children Less Than 24 Months of Age.
Pediatrics, 2013; 132(2): E341-348.
- Glezen, W.P. et al. Risk of primary infection and
reinfection with respiratory syncytial virus. Am J Dis
Child, 1986; 140:543-546.
- Glenn GM, et al. Modeling maternal fetal RSV F vaccine
induced antibody transfer in guinea pigs. Vaccine, 2015; In
press. http://dx.doi.org/10.1016/j.vaccine.2015.08.039.
|
Contact:
|
Novavax,
Inc.
|
|
Barclay A.
Phillips
|
|
SVP, Chief Financial
Officer and Treasurer
|
|
|
|
Andrea N. Flynn,
Ph.D.
|
|
Senior Manager,
Investor Relations
|
|
ir@novavax.com
|
|
240-268-2000
|
|
|
|
Russo Partners,
LLC
|
|
David
Schull
|
|
Todd Davenport,
Ph.D.
|
|
david.schull@russopartnersllc.com
|
|
todd.davenport@russopartnersllc.com
|
|
212-845-4271
|
Photo - http://photos.prnewswire.com/prnh/20150925/271009
To view the original version on PR Newswire,
visit:http://www.prnewswire.com/news-releases/novavax-announces-positive-top-line-data-from-phase-2-clinical-trial-of-rsv-f-vaccine-to-protect-infants-via-maternal-immunization-300149808.html
SOURCE Novavax, Inc.