- Next-generation antigen test system built on more than eight
years of research and backed by industry leading microfluidic
experts
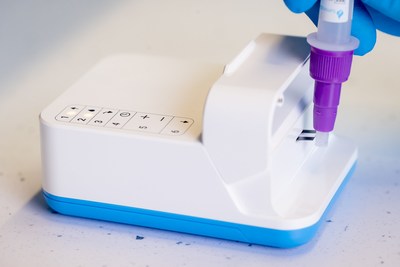
- The Amira System includes the Amira Analyzer, Amira SARS-CoV-2
Ag test strips, swabs and sample preparation materials
- Designed to provide lab-comparable performance at a lateral
flow-comparable price
LONDON, June 9, 2022 /PRNewswire/ -- LumiraDx Limited
(Nasdaq: LMDX), a next-generation point of care diagnostics
company, today announced that it has achieved CE Mark for the Amira
System, its new testing system that provides low-cost, highly
sensitive COVID-19 testing. The Amira Analyzer reads the included
Amira SARS-CoV-2 Ag test strips and returns a result in just 15
minutes. The Amira Analyzer condenses technology from large
laboratory analyzers into a portable instrument, comparable in size
and weight to a deck of playing cards, that delivers high
performing testing in minutes. The initial CE Mark for the Amira
System covers professional use at the point of care. However, the
company plans to seek additional regulatory approvals and
authorizations for the Amira System, including those required for
non-professional, over the counter use, to provide value for home,
events, workplace, and travel testing.
The microfluidic technology in the Amira SARS-CoV-2 Ag test
delivers excellent testing performance. The test has a positive
percent agreement of 90.00% and a negative percent agreement of
99.31% versus RT-PCR in symptomatic individuals, based on clinical
data collected 0-10 days since symptom onset. Within this cohort,
the Amira SARS-CoV-2 Ag test showed sensitivity of 95.6% up to a CT
of 30 indicating high coverage of potentially infectious
individuals.
Ron Zwanziger, LumiraDx's Chief Executive Officer commented,
"The Amira System brings a unique combination of features to the
market. It is one of the only COVID-19 testing solutions on
the market that can offer highly accurate, objectively read
results with the rapid turnaround time and low price of many
lateral flow tests. Frequent testing has become a part of many of
our lives in order to limit the spread of the virus. However,
without trusted results, many testing protocols do not accurately
protect patients, employees, and families."
The Amira System will be commercially available in Europe shortly as a starter kit with one
analyzer and 200 Amira SARS CoV-2 Ag test strips.
About LumiraDx
LumiraDx (Nasdaq: LMDX) is a next-generation point of care
diagnostics company that is transforming community-based
healthcare. Founded in 2014, LumiraDx manufactures and
commercializes an innovative diagnostic Platform that supports a
broad menu of tests with lab comparable performance at the point of
care. LumiraDx diagnostic testing solutions are being deployed by
governments and leading healthcare institutions across
laboratories, urgent care, physician offices, pharmacies, schools,
and workplaces to screen, diagnose, and monitor wellness as well as
disease. LumiraDx has, on the market and in development, 30+ tests
covering infectious diseases, cardiovascular diseases, diabetes,
and coagulation disorders, all on the LumiraDx Platform. In
addition, LumiraDx has a comprehensive portfolio of fast, accurate,
and cost-efficient COVID-19 testing solutions from the lab to point
of need.
LumiraDx is based in the UK with more than 1600 employees
worldwide. Further information on LumiraDx is available
at www.lumiradx.com
Cautionary Note Regarding
Forward-Looking Statements
This press release contains forward-looking statements within
the meaning of the U.S. Private Securities Litigation Reform Act of
1995, including statements regarding the performance and benefits
of the Amira System, the possibility of seeking additional
regulatory approvals and authorizations for the Amira System, and
the expected commercialization timeline for the Amira System. These
statements involve risks, uncertainties and other factors that may
cause actual results, levels of activity, performance or
achievements to be materially different from the information
expressed or implied by these forward-looking statements,
including, among others, general economic, political and business
conditions; regulatory changes; the ability of LumiraDx to maintain
CE marking for its Amira Analyzer and Amira SARS-CoV-2 Ag test; and
those factors discussed under the header "Risk Factors" in the
Annual Report on Form 20-F for the year ended December 31, 2021, which was filed by LumiraDx
with the Securities and Exchange Commission ("SEC") on April 13, 2022, and in other filings made by
LumiraDx with the SEC. Although LumiraDx believes that it has a
reasonable basis for each forward-looking statement contained in
this press release, LumiraDx cautions you that these statements are
based on a combination of facts and factors currently known by it
and its projections of the future, about which it cannot be
certain. LumiraDx undertakes no obligation to update or revise the
information contained in this press release, whether as a result of
new information, future events or circumstances or otherwise,
except as required by applicable law.
Media
Contact:
Colleen.McMillen@lumiradx.com
Commercial
Contact:
Giffin.Daughtridge@lumiradx.com
Photo -
https://mma.prnewswire.com/media/1836100/AmiraImage.jpg