Intercept Shares Tumble Premarket After Negative FDA AdCom Vote
May 22 2023 - 7:57AM
Dow Jones News
By Colin Kellaher
Intercept Pharmaceuticals shares moved lower in premarket
trading Monday after the biopharmaceutical company's latest setback
in its bid to win U.S. Food and Drug Administration approval of
obeticholic acid as a treatment for pre-cirrhotic liver fibrosis
due to nonalcoholic steatohepatitis, a chronic liver condition
commonly known as NASH.
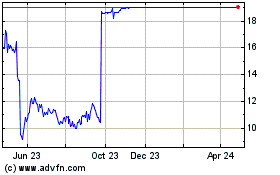
Shares of the Morristown, N.J., company, which closed Thursday
at $13.56, were recently down 14% at $11.66 in premarket trading.
Trading in the stock was halted all day Friday.
An FDA advisory committee meeting on Friday voted overwhelmingly
that the drug's benefits don't outweigh its risks, and that the
agency should defer approval under an accelerated pathway based on
currently available data until data from an ongoing study are
submitted and reviewed.
The FDA in 2020 rejected Intercept's initial application for
obeticholic acid, saying the company's efficacy data didn't
sufficiently outweigh potential risks.
Intercept conducted additional studies and resubmitted the
application in December, seeking an accelerated approval. The
agency has set a target action date of June 22 for a decision.
The FDA often relies on advisory committees to provide
independent advice when a scientific, technical or policy question
arises, such as whether an unapproved product is safe and
effective, and the panels make non-binding recommendations that the
agency generally follows, although it isn't legally bound to do
so.
There are currently no FDA-approved products for NASH, a
progressive disease that is a leading cause for the need for liver
transplants.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
May 22, 2023 07:42 ET (11:42 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
Intercept Pharmaceuticals (NASDAQ:ICPT)
Historical Stock Chart
From May 2024 to Jun 2024
Intercept Pharmaceuticals (NASDAQ:ICPT)
Historical Stock Chart
From Jun 2023 to Jun 2024