Femasys Hosts Congresswoman Lucy McBath at Georgia Headquarters
August 27 2024 - 8:30AM

Femasys, Inc., (Nasdaq: FEMY), a leading biomedical company focused
on addressing significant unmet needs of women worldwide with a
broad portfolio of in-office, accessible, and innovative
therapeutic and diagnostic products, hosted Congresswoman Lucy
McBath, Georgia’s 7th district representative to the U.S. House of
Representatives, for an informative tour of the Company’s facility
headquartered in Suwanee, Georgia.
Femasys Founder, President and Chief Executive Officer Kathy
Lee-Sepsick said, “Women’s reproductive health continues to be
underserved and underfunded across the country. We are encouraged
by the continued dialogue with leaders like Congresswoman McBath
around our improved solutions for accessible, effective, and
affordable reproductive health options for women. We look forward
to continued visits and new and extended conversations with
stakeholders across government, healthcare, and advocates as we
continue to push for better and less-invasive options for women in
their reproductive journeys.”
Femasys provides minimally-invasive solutions that share
delivery platforms. It has four commercially available products:
FemaSeed® – FDA-cleared, intratubal insemination, FemVue® –
contrast-generating device, FemCath® – selective delivery catheter,
and FemCerv® – endocervical tissue sampler. These products have
also received regulatory approval in Canada and Europe under the EU
MDR. The Company’s current lead product candidate in late-stage
clinical development is FemBloc® – permanent birth control.
About Femasys
Femasys is a leading biomedical company focused on addressing
significant unmet needs of women worldwide with a broad portfolio
of in-office, accessible, and innovative therapeutic and diagnostic
solutions, including a lead revolutionary product candidate and
FDA-cleared products. FemaSeed® Intratubal Insemination, an
innovative infertility treatment designed to deliver sperm directly
where conception occurs, is FDA-cleared and has received regulatory
approval in Canada and Europe under the EU MDR.
FemBloc® permanent birth control in late-stage clinical
development is the first and only non-surgical, in-office,
permanent birth control method intended to be a safer option for
women at substantially less cost than the long-standing surgical
alternative. The Company has developed diagnostic products that are
complementary for which it has achieved regulatory approvals to
market in the U.S., Canada, Europe, and other ex-U.S. territories,
and which are commercial-ready due to its in-house manufacturing
capabilities. Its diagnostic products include FemVue® for
fallopian tube assessment by ultrasound, which can be used in
conjunction with FemCath®, an intrauterine catheter for selective
fallopian tube evaluation, and FemCerv®, an endocervical tissue
sampler for cervical cancer diagnosis. Learn more at
www.femasys.com, or follow us on X, Facebook and LinkedIn.
Forward-Looking Statements
This press release contains forward-looking statements that are
subject to substantial risks and uncertainties. Forward-looking
statements can be identified by terms such as “may,” “will,”
“should,” “expect,” “plan,” “anticipate,” “could,” “pending,”
“intend,” “believe,” “suggests,” “potential,” “hope,” or “continue”
or the negative of these terms or other similar expressions,
although not all forward-looking statements contain these words.
Forward-looking statements are based on our current expectations
and are subject to inherent uncertainties, risks and assumptions,
many of which are beyond our control, difficult to predict and
could cause actual results to differ materially from what we
expect. Further, certain forward-looking statements are based on
assumptions as to future events that may not prove to be
accurate. Factors that could cause actual results to differ
include, among others: our ability to develop and advance our
current product candidates and programs into, and successfully
initiate, enroll and complete, clinical trials; the ability of our
clinical trials to demonstrate safety and effectiveness of our
product candidates and other positive results; estimates regarding
the total addressable market for our products and product
candidates; our ability to commercialize our products and product
candidates, or the effect of delays in commercializing our
products, including FemaSeed; our business model and strategic
plans for our products, technologies and business, including our
implementation thereof; and those other risks and uncertainties
described in the section titled "Risk Factors" in our Annual Report
on Form 10-K for the year ended December 31, 2023, and other
reports as filed with the SEC. Forward-looking statements contained
in this press release are made as of this date, and Femasys
undertakes no duty to update such information except as required
under applicable law.
Contacts:
Investors:Gene MannheimerIR@femasys.com
Media Contact:Kati WaldenburgMedia@femasys.com
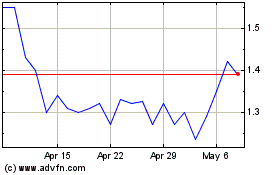
Femasys (NASDAQ:FEMY)
Historical Stock Chart
From Jul 2024 to Aug 2024
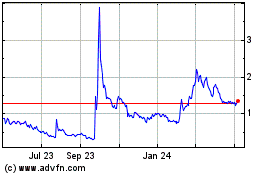
Femasys (NASDAQ:FEMY)
Historical Stock Chart
From Aug 2023 to Aug 2024