EXTON, Pa., Oct. 25, 2021 /PRNewswire/ -- West
Pharmaceutical Services, Inc. (NYSE: WST), a global leader in
innovative solutions for injectable drug administration, is pleased
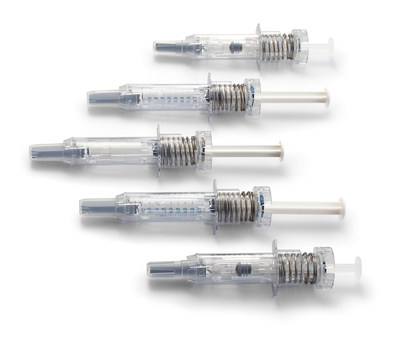
to announce that its NovaGuard® SA Pro safety system – a single-use
accessory for prefilled ISO standard 0.5 mL and 1mL long
staked-needle syringes – has won the INTERPHEX Best Technologies
Innovation Award. The Best Technologies Innovation Award recognizes
cutting-edge technologies, either new technologies or a novel
implementation of existing technologies, that have the potential to
change the way pharmaceutical and biotechnology companies
operate.
"West is honored to receive this award for our NovaGuard SA Pro
needle safety system, which is designed with innovative safety
features to help eliminate accidental needlestick injuries," said
Chris Ryan, Senior Vice President,
Commercial Products and Emerging Markets at West. "We recognize
that needlestick injuries are a serious hazard for healthcare
practitioners, and we developed the NovaGuard system to provide
greater control and enhanced safety when administering injections.
West is proud of this continued innovation with our NovaGuard SA
Pro safety device technology platform, and we continue to look for
ways to deliver greater innovation in the marketplace to ultimately
help our customers help their patients."
To help mitigate these needle stick injuries, the NovaGuard® SA
Pro safety system has recently expanded its technology platform to
include a 0.5mL device for standard 0.5mL glass syringes, in
addition to the 1mL device that was already available for 1mL long
glass syringes. This is important for injectable drug therapies
such as low molecular weight heparin where drug manufacturers
package and go to market with both 0.5mL standard glass syringes
and 1 mL long glass syringes.
This safety system offers a low snap-in force during
manufacturer assembly with the syringe, helps prevent
pre-activation of the syringe, and provides a low activation for
greater user comfort during administration. The NovaGuard SA
Pro safety system also now has the C-clip integrated into the top
portion of the design of the device enabling a one-step syringe
assembly process for our customers in the biopharmaceutical
industry.
West and the diamond logo and
NovaGuard® are trademarks and registered
trademarks of West Pharmaceutical Services, Inc., in the United States and other
jurisdictions.
Forward-Looking Statements
Certain forward-looking statements are included in this press
release. They use words such as "designed," "innovative,"
"eliminate," "provide," "enhanced," "help," "mitigate," "expanded,"
and other similar terminology. These statements reflect
management's current expectations regarding future events and
operating performance and speak only as of the date of this
release. There is no certainty that actual results will
be achieved in-line with current expectations. Specifically,
there is no certainty that the NovaGuard® SA Pro safety device,
either the newly offered 0.5mL device or the already available
1.0mL device, will achieve any particular result in the market.
These forward-looking statements involve a number of risks and
uncertainties. The following are some of the factors that could
cause our actual results to differ materially from those expressed
in or underlying our forward-looking statements: customer decisions
to move forward with the product offering and dependence on
third-party suppliers and partners. These important factors are not
all inclusive. For a description of certain additional factors that
could cause West's future results to differ from those expressed in
any such forward-looking statements, see Item 1A, entitled "Risk
Factors," in West's Annual Report on Form 10-K for the year ended
December 31, 2020. Except as required
by law or regulation, West undertakes no obligation to publicly
update any forward-looking statements, whether as a result of new
information, future events, or otherwise.
About West
West Pharmaceutical Services, Inc. is a leading provider of
innovative, high-quality injectable solutions and services. As a
trusted partner to established and emerging drug developers, West
helps ensure the safe, effective containment and delivery of
life-saving and life-enhancing medicines for patients. With
almost 10,000 team members across 50 sites worldwide, West helps
support our customers by delivering over 40 billion components and
devices each year.
Headquartered in Exton,
Pennsylvania, and in business for nearly a century, West in
its fiscal year 2020 generated over $2.15
billion in sales. West is traded on the New York Stock
Exchange (NYSE: WST) and is included on the Standard & Poor's
500 index. For more information, visit
www.westpharma.com.
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/wests-novaguard-sa-pro-safety-system-recognized-for-best-technologies-innovation-at-interphex-301407212.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/wests-novaguard-sa-pro-safety-system-recognized-for-best-technologies-innovation-at-interphex-301407212.html
SOURCE West Pharmaceutical Services, Inc.