- Design enhancements allow for faster
preparation of Floseal with same trusted safety and
efficacy
- Reinforces Baxter’s efforts to develop
products that support operating room nurses and surgeons
Baxter International Inc. (NYSE:BAX), a global leader in
advancing surgical innovation, today announced that it has received
U.S. Food and Drug Administration (FDA) approval for faster
preparation of its leading hemostatic product, Floseal
Hemostatic Matrix, at the 2019 Association of periOperative
Registered Nurses (AORN) Global Surgical Conference and Expo. This
next generation of Floseal has 20 percent fewer components
and steps to prepare,1 making it easier and faster for operating
room (OR) nurses to get Floseal in the hands of surgeons to
help stop bleeding during procedures. The new design will be on
display at Baxter’s AORN booth #1624.
“We are focused on advancing the art of healing in the operating
room with our innovative and dependable surgical products that
address intraoperative bleeding,” said Wil Boren, president of
Baxter’s Advanced Surgery business. “When creating the next
generation of Floseal, we had OR nurses in mind. We listened
to their feedback so that we could deliver on our promise of
customer-inspired innovation.”
With 20 years of leadership in hemostasis and as a frequently
chosen advanced hemostatic agent,2 Floseal has been proven
to perform quickly and consistently across a range of bleeds in
surgical procedures.3 A 13cm Malleable Applicator is included with
every Floseal kit and allows surgeons to maneuver the
product into the proper position.
“I’ve spent my entire career involved with surgery in various
capacities, and I’ve seen firsthand the importance that speed and
precision play when it comes to preventing bleeding complications
for patients,” said Mary Anne Sanford, BSN, RN, CNOR and senior
manager, Global Medical Affairs at Baxter. “The latest design
enhancements to Floseal make it easier and faster to
prepare, enhancing the ability of perioperative nurses to focus
their efforts to coordinate and support the surgical team’s
activities throughout a procedure.”
Floseal has been ranked by registered nurses in ORs
across the U.S. as the flowable configuration of choice because of
its simple and easy preparation.4 In this latest design, the
diluent ampoule has been replaced by pre-filling the existing
mixing syringe so that Floseal can be prepared more quickly
than the current configuration.
Both active and passive adjunctive hemostatic agents are
available to help control bleeding in surgical procedures when
ligature or conventional methods are ineffective or impractical.
Baxter’s broad portfolio of hemostatic and sealing agents
effectively work to stop bleeding in a variety of anatomies to help
lower bleeding-related complications and to reduce overall costs.
To learn more about Floseal and the rest of Baxter’s
portfolio, please visit https://advancedsurgery.baxter.com/.
About Baxter’s Surgery Portfolio
Baxter is committed to partnering with clinicians to make a
meaningful impact on patient care in operating rooms (OR) in nearly
60 countries. Surgeons rely on our hemostats to stop bleeding
during surgery, our sealants to close wounds, and our repair
patches and biologics to promote healing. We are focused on
pioneering innovative and dependable surgical tools and programs
that help to improve clinical outcomes while reducing the total
cost of care.
About Baxter
Every day, millions of patients and caregivers rely on Baxter’s
leading portfolio of critical care, nutrition, renal, hospital and
surgical products. For more than 85 years, we’ve been
operating at the critical intersection where innovations that save
and sustain lives meet the healthcare providers that make it
happen. With products, technologies and therapies available in
more than 100 countries, Baxter’s employees worldwide are now
building upon the company’s rich heritage of medical breakthroughs
to advance the next generation of transformative healthcare
innovations. To learn more, visit www.baxter.com and follow us
on Twitter, LinkedIn and Facebook.
Rx Only. For safe and proper use of this device, refer to
the full Instructions for Use.
About Floseal
Important Safety Information
Floseal Matrix is indicated in surgical procedures (other
than ophthalmic) as an adjunct to hemostasis when control of
bleeding by ligature or convention procedure is ineffective or
impractical.
Important Risk Information for Floseal Matrix
Do not inject or compress Floseal Matrix into blood
vessels. Do not apply Floseal Matrix in the absence of
active blood flow, e.g., while the vessel is clamped or bypassed,
as extensive intravascular clotting and even death may result.
Do not use Floseal Matrix in patients with known
allergies to materials of bovine origin. Do not use Floseal
Matrix in the closure of skin incisions because it may interfere
with the healing of the skin edges.
Floseal Matrix contains Thrombin made from human plasma.
It may carry a risk of transmitting infectious agents, e.g.,
viruses, and theoretically, the Creutzfeldt-Jakob disease (CJD)
agent.
Floseal Matrix is not intended as a substitute for
meticulous surgical technique and the proper application of
ligatures or other conventional procedures for hemostasis.
Excess Floseal Matrix (material not incorporated in the
hemostatic clot) should always be removed by gentle irrigation from
the site of application. Floseal Matrix swells by
approximately 10% to 20% after product is applied. Maximum swell
volume is achieved within about 10 minutes.
The safety and effectiveness of Floseal Matrix has not
been established in children under 2 years of age and pregnant
women.
Do not use air to remove residual Floseal Matrix from
Applicator tip. The Applicator tips should not be cut. Do not use
Floseal Matrix on bone surfaces where adhesives, such as
methylmethacrylate or other acrylic adhesives, will be required to
attach a prosthetic device.
This release includes forward-looking statements concerning
Floseal, including potential benefits associated with its
use. The statements are based on assumptions about many important
factors, including the following, which could cause actual results
to differ materially from those in the forward-looking statements:
satisfaction of regulatory and other requirements; actions of
regulatory bodies and other governmental authorities; product
quality, manufacturing or supply, or patient safety issues; changes
in law and regulations; and other risks identified in Baxter's most
recent filing on Form 10-K and other SEC filings, all of which are
available on Baxter's website. Baxter does not undertake to update
its forward-looking statements.
Baxter and Floseal are registered trademarks of Baxter
International Inc.
1 Floseal Hemostatic Matrix Instructions for Use. Hayward, CA:
Baxter Healthcare Corporation.2 2017 total unit sales (DRG data)3
D. Makhija, M. Rock, Y. Xiong, J. D. Epstein, M. R. Arnold, O. M.
Lattouf & D. Calcaterra (2017) Cost-consequence analysis of
different active flowable hemostatic matrices in cardiac surgical
procedures, Journal of Medical Economics, 20:6, 565-573,
DOI:10.1080/13696998.2017.12840794 Data on file – Time Motion Study
- Study Number: BXU528129
View source
version on businesswire.com: https://www.businesswire.com/news/home/20190409005409/en/
Media ContactJessica Szramiak, (224)
948-5353media@baxter.comInvestor ContactClare Trachtman, (224)
948-3085
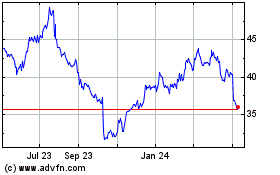
Baxter (NYSE:BAX)
Historical Stock Chart
From Aug 2024 to Sep 2024
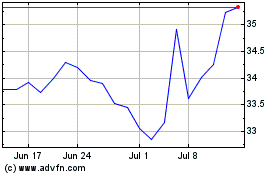
Baxter (NYSE:BAX)
Historical Stock Chart
From Sep 2023 to Sep 2024