UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM
10-K
| [X] |
ANNUAL
REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For
the fiscal year ended August 31, 2015
| [ ] |
TRANSITION
REPORT UNDER SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
Commission
file number: 000-52445
PETLIFE
PHARMACEUTICALS, INC,
(FORMERLY
CLEAR TV VENTURES, INC.)
(Name
of registrant as specified in its charter)
| Nevada |
|
33-1133537 |
| (State
or other jurisdiction of |
|
(I.R.S.
Employer |
| incorporation
or organization) |
|
Identification
No.) |
| |
|
|
433
N. Camden Dr.
Beverly
Hills, CA |
|
90210 |
| (Address
of principal executive offices) |
|
(Zip
Code) |
(310)
279-5152
(Registrant’s
telephone number, including area code)
Securities
registered pursuant to Section 12(b) of the Act: None
Securities
registered pursuant to Section 12(g) of the Act:
Common
Stock, $0.001 par value
Indicate
by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes [ ]
No [X]
Indicate
by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Exchange Act. Yes
[ ] No [X]
Indicate
by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange
Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports),
and (2) has been subject to such filing requirements for the past 90 days. Yes [X] No [ ]
Indicate
by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive
Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such
shorter period that the registrant was required to submit and post such files). Yes [ ] No [X]
Indicate
by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not
be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference
in Part III of this Form 10-K or any amendment to this Form 10-K. [ ]
Indicate
by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller
reporting company. See definition of “accelerated filer”, “large accelerated filer” and “smaller
reporting company” in Rule 12b-2 of the Exchange Act.
| Large
accelerated filer [ ] |
Accelerated
filer [ ] |
Non-accelerated
filer [ ] |
Smaller
reporting company [X] |
Indicate
by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes [ ] No
[X]
The
aggregate market value of the common stock of the registrant held by non-affiliates as of November 28, 2014 the last business
day of the registrant’s most recently completed first fiscal quarter based on the closing sale prices of the registrant’s
common stock on that date as reported on the OTCQB maintained by OTC Markets Group, Inc. was $15,338,480. For purposes of this
computation, all officers, directors, and 5 percent beneficial owners of the registrant are deemed to be affiliates. Such determination
should not be deemed an admission that such directors, officers, or 5 percent beneficial owners are, in fact, affiliates of the
registrant.
The
number of shares of the registrant’s common stock
outstanding as of December 24, 2015 was 38,115,356 shares.
TABLE
OF CONTENTS
The
document (including information incorporated herein by reference) contains “forward-looking statements” within the
meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, which involve a degree
of risk and uncertainty due to various factors affecting PetLife Pharmaceuticals, Inc. (Formerly Clear TV Ventures, Inc. and formerly
Eco Ventures Group, Inc.)
PART
I
Forward-Looking
Statements
This
Annual Report on Form 10-K contains forward-looking statements, including, without limitation, in the sections captioned “Business,”
“Risk Factors,” and “Management’s Discussion and Analysis of Financial Condition and Result of Operations,”
and elsewhere. Any and all statements contained in this Current Report that are not statements of historical fact may be deemed
forward-looking statements. Terms such as “may,” “might,” “would,” “should,” “could,”
“project,” “estimate,” “pro-forma,” “predict,” “potential,” “strategy,”
“anticipate,” “attempt,” “develop,” “plan,” “help,” “believe,”
“continue,” “intend,” “expect,” “future,” and terms of similar import (including
the negative of any of the foregoing) may be intended to identify forward-looking statements. However, not all forward-looking
statements may contain one or more of these identifying terms. Forward-looking statements in this Annual Report may include, without
limitation, statements regarding (i) the plans and objectives of management for future operations, (ii) a projection of income
(including income/loss), earnings (including earnings/loss) per share, capital expenditures, dividends, capital structure or other
financial items, (iii) our future financial performance, including any such statement contained in a discussion and analysis of
financial condition by management or in the results of operations included pursuant to the rules and regulations of the Securities
and Exchange Commission (the “SEC”), (iv) our beliefs regarding potential clinical and other health benefits of our
PetLife products, and (v) the assumptions underlying or relating to any statement described in points (i), (ii), (iii) or (iv)
above.
The
forward-looking statements are not meant to predict or guarantee actual results, performance, events or circumstances and may
not be realized because they are based upon our current projections, plans, objectives, beliefs, expectations, estimates and assumptions
and are subject to a number of risks and uncertainties and other influences, many of which we have no control over. Actual results
and the timing of certain events and circumstances may differ materially from those described by the forward-looking statements
as a result of these risks and uncertainties. Factors that may influence or contribute to the inaccuracy of the forward-looking
statements or cause actual results to differ materially from expected or desired results may include, without limitation, our
inability to obtain adequate financing, the significant length of time and resources associated with the development of our products
and related insufficient cash flows and resulting illiquidity, our inability to expand our business, significant government regulation
of our business and the healthcare industry, the results of clinical studies or trials, lack of product diversification, volatility
in the price of our raw materials, existing or increased competition, results of arbitration and litigation, stock volatility
and illiquidity, and our failure to implement our business plans or strategies. A description of some of the risks and uncertainties
that could cause our actual results to differ materially from those described by the forward-looking statements in this Annual
Report appears in the section captioned “Risk Factors” and elsewhere in this Annual Report.
Readers
are cautioned not to place undue reliance on forward-looking statements because of the risks and uncertainties related to them
and to the risk factors. We disclaim any obligation to update the forward-looking statements contained in this Annual Report to
reflect any new information or future events or circumstances or otherwise.
Readers
should read this Annual Report in conjunction with the discussion under the caption “Risk Factors,” our financial
statements and the related notes thereto in this Annual Report, and other documents which we may file from time to time with the
SEC.
All
references in this Form 10-K that refer to the “Company,” “PetLife Pharmaceuticals, Inc.”, “Registrant,”
“we,” “us” or “our” are to Petlife Pharmaceuticals, Inc., a Nevada formed corporation formerly
known as “Clear TV Ventures, Inc.” and “Eco Ventures Group, Inc.”
Item
1. Business
PetLife
Pharmaceuticals, Inc. (PetLife) has developed and is launching a new generation of potentiated veterinary cancer medications and
nutraceuticals, based on the same patented formula “Escozine” and production processes that have been scientifically
proven as an effective treatment for cancer in humans for years. Escozine (for humans) is currently sold as either a nutraceutical
or prescription drug in 40 countries including the United States.
Historical
Company Information
We
were incorporated on April 5, 2002 under the laws of the State of Nevada as “Aztek Ventures Inc.” Effective November
13, 2007, we filed a Certificate of Amendment to our Articles of Incorporation to change our name from “Aztek Ventures Inc.”
to “Genesis Uranium Corp.” Effective April 21, 2008, we amended our Articles of Incorporation to change our name from
“Genesis Uranium Corp.” to “Vault Technology Inc.” to reflect the change in our business focus beyond
solely that of uranium exploration. Effective July 10, 2009, we filed a Certificate of Amendment to our Articles of Incorporation
to change our name from “Vault Technology, Inc.” to “Modern Renewable Technologies, Inc.” (“Modern”).
On May 27, 2011, Modern, merged with Eco Ventures Group, Inc., and the name of the Company was changed to Eco Ventures Group,
Inc. On July 15, 2013, the Company entered into an Agreement and Plan of Merger with Clear TV Ventures, Inc. Under the terms of
the merger, Clear TV became the surviving corporation. On June 26, 2014, Eco Ventures Group, Inc. entered into an Agreement and
Plan of Merger with its subsidiary, PetLife Pharmaceuticals, Inc., a Nevada Corporation, with PetLife Pharmaceuticals, Inc. being
the surviving entity. As part of that merger, the name of the Company was changed to PetLife Pharmaceuticals, Inc. and each 15
shares of our common stock were exchanged for one share in the surviving company.
Business
of PetLife Pharmaceuticals, Inc.
PetLife
Pharmaceuticals, Inc. (PetLife) has developed and is launching a new generation of HP (high potency) veterinary cancer medications
and nutraceuticals, based on the same patented formula “Escozine” and production processes that have been scientifically
proven as an effective treatment for cancer in humans for years. Escozine (for humans) is currently sold as either a nutraceutical
or prescription drug in 40 countries including the United States.
Given
the histology (the similarity of the cellular biology) between humans and dogs and cats and the growing body of new preclinical
research (UCSD) establishing the effectiveness of Escozine for the treatment of animals, PetLife has licensed the worldwide rights
to formulate, package and market a new product line, “Escozine for PetsTM.”
The
Company’s products have been demonstrated as a preventative and a treatment of cancer, and to dramatically improve the quality
of life, and extend the longevity of pets with cancer — without the painful, debilitating side effects of toxic, conventional
chemotherapy.
PetLife
also has studied and plans to introduce an Escozine for PetsTM product line for livestock, which suffer from high rates
of cancer at a cost of billions to the industry.
Summary
We
have developed and are launching a new generation of high potency veterinary cancer medications and nutraceuticals, based on the
same patented “Escozine” formula and its production processes that, for years, have shown scientific evidence as being
effective for cancer in humans.
For
humans, PetLife’s sister company, Medolife, has been successfully marketing its Escozine products through direct sales in
40 countries. Medolife has invested over $10 million in R&D in Escozine (for humans) over 10 years and amassed substantial
scientific documentation, worldwide of its effectiveness.
“Escozine”
has been used for years to treat humans with cancer in many countries and its positive results are backed by numerous scientific
research, pre-clinical and clinical observations that confirm its safety and efficacy. It is registered and certified for oncological
treatment by the Minister of Health in the Dominican Republic, and is registered and distributed in six countries (Dominican Republic,
Vietnam, Russia, Belarus, Nigeria and Kazakhstan).
At
the molecular level, the cancer cells of humans, cats and dogs are quite similar. With respected scientific research (UCSD and
others) establishing Escozine as an effective potential solution for pets, we have licensed the rights to use Medolife’s
patented formula and manufacturing process to launch “Escozine for Pets™” — a line of over-the-counter
nutraceuticals, infused dog and cat ‘treats’ and prescription strength pharmaceuticals for pets.
PetLife’s
main product, Escozine for Pets™ is a natural product containing serum derived from the Caribbean Blue Scorpion and polarized
using PetLife’s patented polarization technology. It is a nontoxic and is potentiated up to prescription drug strength levels
by efficiency.
PetLife’s
products are produced to the highest industry standards in a US FDA-approved manufacturing facility in southern California.
The
National Cancer Institute reported that in the United States alone, nearly 6 million dogs and 6.5 million cats are diagnosed with
cancer annually with dogs being 35 times and cats 40 times more likely to suffer from cancer than humans. Sixty percent of dogs
and cats over six years of age will be diagnosed with cancer.
In
the United States over 92 million households own a dog or cat, representing 83.3 million dogs and 95.6 million cats. Consumer
spending on domestic companion animals in the United States alone is expected to reach over $58 billion in 2014, with over $13
billion being spent on over the counter medications.
Concurrently,
PetLife plans to apply to the FDA in the second quarter of 2016 for approval of prescription strength versions for Pets, including
orally and intravenously administered as well as a version for direct administration into cancerous tumors. Although there can
be no assurance the FDA will work this quickly, the Company anticipates FDA approval of its prescription strength products in
the second half of 2016, and intends to roll out those products shortly thereafter.
The
Company has a developed a sophisticated, multi-channel marketing strategy that includes direct sales, retail, veterinarian vertical
market, affiliate sales and infomercials.
Business
Strategy
●
Initially, enter the market with an oral nutraceutical version of Escozine for Pets™ with
a focus on the oncology needs of dogs and cats.
●
Raise capital through traditional means, including through private placement of securities, public offerings or bank
financings. Apply for FDA approval of the Caribbean Blue
Scorpion animal pharmaceutical drug, Escozine for Pets GNP-1, for cancer treatment, a prescription strength oral
pharmaceutical as well as a concentrated intravenous and injectable version for direct administration to a tumor.
●
Concurrently develop edible dog and cat treats that are infused with the Escozine active ingredients that deliver preventative
benefits for both dogs and cats.
●
Develop a line of nutraceuticals and drugs in the second half of 2016 for livestock, based on the same molecule and patented production
process to prevent disease, to lower the industry’s reliance on antibiotics, and to improve the quality and confidence in
the livestock products sold around the world.
The
Company’s Escozine Product
Escozine™
was originally developed and commercialized by Medolife, Corp. in 2007. Until 2007, this natural medicine has only been available
in Cuba, where it is known as Escozul, or its more diluted homeopathic version called Vidatox. The founder of Medolife holds through
a trust the exclusive patent license, (United States patent # 8,097,284 B2) of the bioactive peptide and polarization technology
used to make the Polarized Dilute Blue Scorpion Venom. Escozine™, is registered and certified for oncological treatment
by the Minister of Health in the Dominican Republic. Escozine™ is currently registered and distributed in six countries
(Dominican Republic, Vietnam, Russia, Belarus, Nigeria and Kazakhstan).
Studies
show that the patented polarization technique (US Patent # 8097284 B2) increases the delivery of the scorpion venom into cancer
cells which amplifies the effectiveness of Escozine™ dramatically compared to Cuban products Escozul or Vidatox. PetLife’s
Chief Executive officer, Dr. Mikaelian, is currently the only scientist who has completed all research and the development process
and has attained the legal registration to commercialize Blue Scorpion venom derived products under the name of Escozine. The
product, Escozul, is not produced or sold by Medolife or PetLife and neither Medolife nor PetLife rely on this product in any
way. Medolife has its own independent source of Blue Scorpion venom in the Dominican Republic and has established the world’s
first Scorpion reservation which is 50,000 square meters and can produce millions of doses to meet global demand. Products are
currently sold direct, through online marketing to over 40 countries, as well as to six countries where the product has been registered
(Dominican Republic, Vietnam, Russia, Belarus, Nigeria and Kazakhstan). In 2014, Medolife granted an exclusive global license
for production, marketing and sales to PetLife, for all applications of Escozine for Pets, including the nutraceutical and FDA
approved versions.
Escozine™
serum has demonstrated promising results in human preclinical studies in 2012 at UCSD Moores Cancer Center, as well as human clinical
observations; results from UCSD also showed that Escozine had significant synergy with traditional chemotherapy agents cyclophosphamide,
Fludarabine, Etoposide and others. Many Escozine users have combined the product with chemotherapy, radiation, hormone therapy
and other conventional treatments with positive results. In 2007, Medolife collaborated with Atheris Laboratory in Switzerland,
one of the world leaders in the study and analysis of crude venoms, to confirm that the venom of Rhopalurus Junceus from Cuba
and Rhopalurus Princeps from Dominican Republic both share similar properties and have similar mechanisms of action in cancer
cells.
Polarized
Dilute Blue Scorpion Venom solution has proven to lead to more apoptosis (cancer cell death) in humans and animals, in comparison
to non-polarized Dilute Blue Scorpion Venom solution. Additionally, polarized Dilute Blue Scorpion Venom solution can be used
with gold nanoparticles as an intermediary agent to increase the targeting and binding preferences of Polarized Dilute Blue Scorpion
Venom solution with abnormal or malignant cells in humans as well as in animals. The inclusion of gold nanoparticles, as the intermediary
agent for the drug version further enhances the targeting and binding preferences of the Polarized Dilute Blue Scorpion Venom
solution with abnormal or malignant cells in humans as well as animals.
One
component of PetLife’s licensed patent is for the process of polarizing the dilute Blue Scorpion venom; the patented technology
involves the circulation of the dilute solution past a series of electromagnets, which are generating resonating field with the
Scorpion Venom therapeutic components, such as small molecular peptide, chlorotoxin (CLTX). The benefits of ingesting polarized
Escozine dilute scorpion venom solution and its main ingredient are well documented through the clinical observations of well-known
scientists in this field like, Dr. Omran, Dr. Stöcklin, Dr. Philippe Favrea as well as Dr. Mikaelian and Dr. Ramon Feliz
and include:
| |
● |
Reduces
pain. |
| |
|
|
| |
● |
Enhances
the recipient’s immune-system. |
| |
|
|
| |
● |
Reduces
cancerous cell multiplication. |
| |
|
|
| |
● |
Inhibits
new blood vessels formation form pre-existing cancerous cells. (Angiogenesis) |
| |
|
|
| |
● |
Improves
quality of sleep. |
| |
|
|
| |
● |
Reduces
inflammation. |
| |
|
|
| |
● |
Dramatically
reduces the painful side-effects of chemotherapy and radiation treatment. |
| |
|
|
| |
● |
Improves
appetite |
From
statements in the patent document, polarized blue scorpion venom delivers an array of health benefits. According to extensive
research contained in the 2012 UCSD Moore’s Cancer Center report, cancer cells treated with Escozine showed more apoptosis
than many traditional chemotherapy agents. In studies by oncologists Dr Ramon Feliz and Dr. Vargas as well as in other studies,
patients administered with polarized blue scorpion serum solution frequently experienced the following health benefits: improved
quality of life, increased survival rate, and tumor remission. Many patients who have used polarized blue scorpion venom in their
treatment have experienced different degrees of these stated benefits. The degree of benefits of any particular patient is impacted
by the patient’s stage of cancer, genetic issues, condition of vital organs, continued exposure to environmental toxicity,
lifestyle choices and other factors.
Dr.
Mikaelian further determined through research that the cellular reproduction challenges of animal cancers are similar or closely
mirror those occurring in human cancers. His early tests on animals (pets and livestock) using polarized Dilute Scorpion Serum
solution suggest that they will experience similar positive health related benefits.
PetLife
will initially develop dedicated cancer preventative products, such as animal treats. These products are designed to prolong pets’
lives when taken consistently. The Company anticipates this will drive sales for PetLife as clients adopt these types of products
as part of their pet’s normal daily diets. Part of PetLife’s sales and marketing framework is to not only sell the
products to animals post cancer diagnosis, but to educate owners on cancer and autoimmune prevention. PetLife’s preventative
animal treats, for example, are an excellent way for pet owners to promote lasting health and viability for their companions.
PetLife
anticipates developing the following products for the cancer preventative products:
| |
● |
Escotreat
– a natural pet treat preventative |
| |
|
|
| |
● |
Escotab
– a natural daily pet preventative tablet |
| |
|
|
| |
● |
Zine
Pet Foods –natural preventative foods |
Escozine
for Pets has a universal effect on different cancer types. With inexpensive modifications, PetLife can create cancer specific
products that may increase sales. The following are the top veterinary cancer types that PetLife intends to target:
| |
● |
Lymphoma
or Lymph sarcoma |
| |
|
|
| |
● |
Hemangiosarcoma |
| |
|
|
| |
● |
Osteosarcoma |
| |
|
|
| |
● |
Mast
Cell Tumor |
| |
|
|
| |
● |
Melanoma |
| |
|
|
| |
● |
Squamous
Cell Carcinoma |
| |
|
|
| |
● |
Mammary
Carcinoma |
| |
|
|
| |
● |
Apocrine
Gland Carcinoma (Anal Sac) |
| |
|
|
| |
● |
Transitional
Cell Carcinoma |
| |
|
|
| |
● |
Soft
Tissue Sarcoma |
| |
|
|
| |
● |
Lymphoma |
| |
|
|
| |
● |
Squamous
carcinoma |
| |
|
|
| |
● |
Fibrosarcoma |
| |
|
|
| |
● |
Lung
tumors |
| |
|
|
| |
● |
Brain
tumors |
| |
|
|
| |
● |
Nasal
tumors |
| |
|
|
| |
● |
Liver
tumors |
When
surgery is difficult to perform, PetLife has designed three products for differentiated intake protocols for home and veterinary
usage.
●
Oral / Rectal - Home oral and rectal administration of the Escozine for Pets liquid makes the animal easy to treat. The solution
is colorless and odorless and can be administered through a droplet or enema. This is an effective tool which clients can use
in the convenience of their home.
●
Patch - A patch can be used at home and positioned in the area of the tumor for effective, targeted treatment.
●
Injectable - Escozine can be used by veterinarians to directly inject the product into the tumor for effective, targeted high
dose treatments. This will increase sales with vets who will have an effective in clinic, targeted product will require clients
to bring their pets into the vet clinic for treatment.
PetLife,
through the work of its analytical scientists, has identified a diversity of diseases that it believes will respond positively
from treatments with the Escozine for Pets™ nutraceuticals and drugs. The diseases below illustrate those areas of potential
expansion beyond the current focus on veterinary oncology related illnesses. The expansion of the application of PetLife’s
products and technical knowledge into other health related areas will require the addition of skilled research scientists and
pharmacological experts. PetLife will initially develop the new PetLife products with the use of Medolife’s research and
development expertise. PetLife will expand its market reach and increase its revenue by treating other diseases.
PetLife
intends to also develop products for pets in the following areas:
●Arthritis:
Arthritis (or osteoarthritis) is a slowly progressive, degenerative disease of the joints for which there is no cure. However,
Escozine for Pets can help to prevent, delay, and manage arthritis in dogs or cats. Arthritis affects 1 in 5 dogs over the age
of 7 and over 90% of geriatric cats have arthritis; currently 12 million cats have arthritis.
●
Hepatitis: As with humans, Hepatitis in dogs and cats affects the liver. This contagious disease includes symptoms such as fever,
vomiting and diarrhea accompanied by abdominal pain. Furthermore, Hepatitis may lead to kidney damage. Approximately, 12% of dogs
and 40% of cats get Hepatitis.
●
Lyme disease: Lyme disease is a dangerous bacterial disease that can cause irreversible damage to a companion animal’s health.
About 5% of dogs contract Lyme disease while the statistics for cats is nominal.
The
anticipated products in development in these areas are:
| |
● |
Escozine
for Pets™ Arthritis |
| |
|
|
| |
● |
Escozine
for Pets™ Hepatitis |
| |
|
|
| |
● |
Escozine
for Pets™ Lyme |
Scientific
Research on EscozineTM
In
2012, UCSD (University of California San Diego, Moores Cancer Center, together with Medolife Corp.) conducted research using polarized
dilute Blue Scorpion venom utilizing the Mikaelian patented process # US 8097284 B2. The results demonstrated up to 95% apoptotic
(programmed cell death) effect in a variety of cancer cell lines incubated for 48 hours, and showed a binding preference to cancer
cells only while not affecting normal cells. Additionally, the study showed a synergistic effect with traditional chemotherapy
treatments. Research anticipates that PetLife’s Escozine for Pets™ will also extend the life of pets suffering from
cancer and improve their quality of life.
PetLife’s
main product, Escozine for Pets™ is a natural product containing serum derived from the Caribbean Blue Scorpion and polarized
using PetLife’s patented polarization technology. PetLife’s Escozine for Pets™ is nontoxic and is potentiated
up to prescription drug strength levels.
Global
Market for Escozine and the Company’s Products
The
global veterinary pharmaceutical industry is composed of various veterinary health products including biological, veterinary pharmaceuticals
and medicated food additives. Over the past few years, the range of animal health products has diversified, currently encompassing
metabolic drugs, reproductive aids, anti-ineffectives, feed additives, imaging diagnostics, vaccines, parasite control, and topical
solutions. Veterinary products have emerged for treating chronic diseases such as cancer, osteoarthritis and cardiovascular disease.
Pet owners, more than ever before, are spending a larger proportion of their income to take care of their pets’ health.
In
the future, the global market for pet health products is expected to grow by an average of 4 - 6% annually and become more specialized.
The major drivers will be: (1) the continued strengthening of the bond between owners and their animal companions, (2) increasing
companion animal owner awareness and willingness to pay for care, (3) increasing occurrence of cancer in dogs and cats than people
while only a fraction are treated because of prohibitive costs, (4) offering an FDA drug at competitive prices, and potentially
more effective, as an incentive for more pet owners to treat their pets and extend their duration and quality of life. Companion
animals have come to play an important part in the lives of many people. Unsurprisingly the bond between companion animals and
people continues to strengthen in parallel to the market for products that contribute significantly to the health and well-being
of these animals, such as the products offered by PetLife.
The
current focus of the Company’s market is veterinary oncology, a branch of veterinary medicine whose emphasis is dealing
with cancer diagnosis and treatment in animals, especially companion animals. Over the years, the number of animals dying of cancer
has increased. For example, approximately 45% of dogs aged ten years and above die of cancer. Part of PetLife’s product
expansion will include an injectable version that will focus on decreasing and eliminating tumors that are accessible to direct
injection.
PetLife
realizes the parallels between animals and their owners regarding certain conditions, such as poor diet, and its consequences.
Overall, the companion animal segment is considered to be the health segment that parallels the human pharmaceutical sector. Many
of the innovations in human medicine, at least in terms of new medications, are subsequently adapted and tailored to suit companion
animals, like Escozine for Pets™. It is therefore no surprise that Escozine for Pets’ studies have paralleled the
Escozine™ for humans’ studies.
The
global animal health market is consolidated with the top ten players controlling the majority share of the market. For many years
the largest players in the companion animal market have been Merial and Zoetis (a subsidiary of Pfizer). However, the market has
seen considerable consolidation mainly through acquisitions and mergers. Proposed mergers and acquisitions are monitored very
carefully by governments around the world (such as the Directorate General for Economic and Financial Affairs in the European
Union and the United States Federal Trade Commission) to protect the consumer from reduced competition, price increases, and reduced
innovation.
While
the U.S. represents the biggest regional market for animal medication, Asia-Pacific region represents the fastest growing market
for pet medication with annual dollar sales in the region growing at an annual average of 10.5%. This can be attributed to factors
such as increasing income, improvement in per capita consumption per animal, improved living standards, as well as increased pet
populations in various regions all over the world. About half of Brazilian households have a dog, more than any other country,
according to data tracker Euromonitor, and pet food sales there rose 10 percent to $5.6 billion last year, trailing only the U.S.
and U.K. in the $71 billion worldwide market.
Despite
remaining challenges in the overall economy, the pet industry has remained unshaken and has been less affected by the recent global
recession than other retail sectors. Over the past years, the United States and the United Kingdom have been the leaders in the
pet industry. However, today there are a number of other countries that are coming out as players in the global pet industry.
The trend in world pet industry has increased tremendously, which is a clear indication of the growing market for veterinary oncology
services.
Market
in the United States
The
veterinary oncology market in the United States is expected to grow mainly due to an increase in the pet population in the United
States. People’s attitude towards companion animals has changed, as most pet owners now treat their pets as family members.
In a report by Global Industry Analysts, Animal Medication: A Global Strategic Business Report, the authors indicate that animal
care often mirrors the trends in human health care. Diagnosis and disease monitoring in veterinary medicine, particularly for
companion animals, has also followed trends in human medicine. Diagnostic imaging techniques, such as ultrasonography, computed
tomography (CT), and magnetic resonance imaging (MRI), have become commonplace and many veterinary practices have in-house analyzers
for clinical chemistry and hematology, as well as rapid patient-side tests, e.g., for infectious agents.
The
National Cancer Institute reported that nearly six million dogs in the United States alone are diagnosed with cancer annually,
with dogs being 35 times more likely to suffer from cancer than humans. Over six years of age, sixty percent of America’s
83.3 million dogs and 95.6 million cats will be diagnosed with cancer. Unfortunately, only an estimated 10% of those diagnosed
receive treatment. PetLife understands that 90% of dogs and cats with cancer go untreated because of unrealistically high costs
associated with treatments as well as the dramatic suffering typical of treating pets with toxic, conventional chemotherapy.
Marketing
Plan
PetLife
will deliver its nutraceutical and prescription strength medicines to the enormous population of untreated animals by making the
product affordable and convenient in both the home and veterinary setting. PetLife will also capitalize on the increased willingness
of pet owners to spend more on their animals’ preventative health, and of veterinarians to meet that demand as key drivers
of this market. PetLife will also dedicate their marketing efforts to include educating the public as to the risk their companion
animal has at developing cancer. The Company will emphasize the importance on pet owners being proactive and taking the necessary
steps to protect their animal. The Company will offer Escozine for Pets as the preventative solution.
Currently,
apart from surgery which carries its own risks for older pets, the preponderance of treatment for dogs and cats with cancer is
simply the same, drastically expensive (most pets do not have medical insurance), toxic chemotherapy drugs developed for humans
— scaled down in dosage by the pet’s weight — with many of the same side effects such as nausea, diarrhea, lost
appetite, cachexia (“wasting” loss of muscle and fat tissues) headache, hair loss, liver and kidney toxicity and susceptibility
to opportune infections.
Direct
Sales
PetLife
has lined up key team members to execute a traditional, multichannel marketing campaign. This campaign, composed of the traditional
five “P” marketing mix will include:
●
Product – PetLife will capitalize on the proprietary nature of their primary product offering, Escozine for Pets™.
PetLife will sell Escozine for Pets™ to veterinarians using multiple respected veterinarian centers of influence to promote
the product. PetLife will also sell directly to pet owners through online and brick and mortar establishments; this will be achieved
using a well-connected sales force to market to both distributors and direct to retail outlets.
●
Physical attributes – a newly redesigned bottle has been approved.• Place or distribution
– the Company will configure its corporate structure to accommodate the most efficient global supply chain, production and
distribution through regional sales centers. PetLife representatives will manage the supply chain, local final production (as
required) and distribution.
●
Promotion – direct to vet, direct to consumer, direct to wholesaler; there will also be trade
shows, direct vet outreach and online webinars. PetLife will also implement an aggressive online marketing campaign to drive sales
in the United States and key international markets using:
(i)
DOSEP Campaign: DOSEP (Direct Organic Search Engine Optimization) is a groundbreaking online awareness campaign and technology
that will push PetLife’s web presence to the top of Google organic search results for targeted search keywords determined
by PetLife management.
(ii)
Multi-Touch Facebook Campaign: The retained Marketing Company will execute a targeted multi-touch Facebook advertising campaign
that will generate major social awareness (and resulting online sales) of Vet Oncology products. The marketing messages will appear
directly in the Facebook newsfeeds of specific demographic groups who additionally have expressed personal or professional interest
in cancer treatment, based on research and targeting of the specific interests contained in their Facebook profiles.
(iii)
Scientifically-Optimized Banner Ad Campaign: The Marketing Company will conduct research for optimal placement, and perform continuous
split testing of ads to quickly determine “winners” and then further refine campaigns to determine the optimal combination
of performance metrics to justify increasing ad budget spend for top-performing banner campaigns.
●
Media – the Company will engage a top notch boutique public relations firm to generate media
coverage for its products in general news, family, pet and veterinarian publications, e-zines, videos and the blogosphere to communicate
Escozine for Pets™ features and benefits to consumers.
The
online strategic marketing campaigns will repeatedly “local-launch” PetLife into specific metro markets while concurrently
building PetLife’s overall brand and marketing presence globally. Repeating the online marketing campaign in each additional
targeted metro market will not only localize and establish the PetLife’s brand and products, each event will also catalyze
PetLife’s local and nationwide Google search rankings via DOSEP, Multi-Touch Facebook and Scientifically-Optimized Banner
Ads, producing both short- and long-term online sales growth, and firmly establish PetLife as a market leader.
Multi-level
Marketing
PetLife
has partnered with a multi-level marketing (MLM) platform in order to drive sales. This MLM partner will utilize a sales force,
who will be compensated not only for sales they personally generate, but also for the sales of the other salespeople that they
recruit. This recruited sales force is referred to as the participant’s “downline”, and can provide multiple
levels of compensation. Independent distributors develop their organizations by either building an active consumer network, who
buy direct from the company, or by recruiting a downline of independent distributors who also build a consumer network base, thereby
expanding the overall organization. Essentially, salesmen become independent distributors. They operate as contractors. PetLife
sees three major benefits from the use of multi-level marketing: (1) PetLife will develop a large sales force without the expense
of full time employees, (2) PetLife will experience a decrease in direct sale and marketing costs, and (3) PetLife’s distributors
generally work very hard to improve their income, which in turn generates more revenue for PetLife.
Direct
Response: Infomercials
This
year an estimated $150 billion dollars in product sales will be generated by infomercials. Specifically, over $1.5 billion will
be spent on pet-related purchases through infomercials. Research has shown that retail sales driven by infomercials all range
from two to 15 times the infomercial sales. More and more major brands are integrating infomercials into their marketing mix.
Infomercials share the product story and benefits with millions of additional prospects at a cost per lead or cost per order that
usually matches or beats other direct marketing channels such as direct mail or print ads.
PetLife
intends to partner with a well-known household name from Hollywood and a well-respected veterinarian to film differentiated 2
to 30 minute infomercials on Escozine for Pets™. PetLife anticipates the infomercial will:
| |
● |
Reach
millions of new customers and present a complete product story. |
| |
|
|
| |
● |
Enhance,
support, and fill in where other advertising avenues, particularly spot television, leaves off. |
| |
|
|
| |
● |
Generate
qualified leads. |
| |
|
|
| |
● |
Reduce
advertising costs per order and avoid print and mailing costs. |
| |
|
|
| |
● |
Rapidly
and cost-effectively introduce a new product or revive an under-marketed but potentially profitable product. |
PetLife
intends to capitalize on the lucrative channels of distribution available to an infomercial marketer. Only one out of every 100
consumers watching an infomercial actively calls an 800 number and purchases the product immediately. Therefore, the retail channel
provides a means for the other 99 individuals - who may have been primed by the infomercial - to buy the product.
One-step
infomercials are commonly utilized for products previously unable to get retail shelf space in order to gain retailers attention
and establish instant distribution. For example, PetLife can inform a retailer that more than a million consumers ordered their
product directly from their television ads and thousands of others called for more information. These kinds of figures are meant
to grab the attention of brick and mortar retailers, who then purchase and showcase infomercial products realizing that “as
seen on TV” are big sellers at retail.
Competition
For
most of the products the Company offers there are a number of competitors, several of which are publically traded where they not
only manufacture and produce their own products but also have established distribution and sales networks and participate in large
group purchasing organizations within the medical industry. As mature companies, they also have extensive legacy systems and expensive
administrative and sales commission cost structures. In addition, there are independent distributorships of pet medications primarily
focused on limited geographic markets and products located across the United States.
Key
among the Company’s competitors are:
●
Zoetis, Inc.: the world’s largest producer medicine and vaccinations for pets and livestock.
Zoetis is engaged in the discovery, development, manufacture and commercialization of animal health medicines and vaccines, with
a focus on both livestock and companion animals. It offers a diversified product portfolio, including vaccines, parasiticides,
anti-infectives, medicated feed additives and other pharmaceuticals, for both livestock and companion animal customers.
●
Merial, Inc.: Sanofi Pasteur is a worldwide leader in the vaccine industry. Its net sales amounted
to €3,716 million in 2013, with leading vaccines in five areas: pediatric vaccines, influenza vaccines, adult and adolescent
booster vaccines, meningitis vaccines, and travel and endemic vaccines. The company’s Animal Health activity is carried
out through Merial, one of the world’s leading animal healthcare companies, dedicated to the research, development, manufacture
and delivery of innovative pharmaceuticals and vaccines used by veterinarians, farmers and pet owners and providing a comprehensive
line of products to enhance the health, well-being and performance of a wide range of production and companion animals.
●
Virbac Sa: Virbac SA develops, manufactures, and sells vaccines and medicines to prevent and treat pathologies for companion and
food-producing animals in France, the rest of Europe, North America, Latin America, Africa, the Middle East, Asia, and the Pacific
region. The company’s products are comprised of a multitude of products in the animal health sector.
●
Dechra Pharmaceuticals Plc: Dechra Pharmaceuticals PLC is engaged in the development, manufacture, distribution, sale, and marketing
of veterinary pharmaceuticals worldwide. It markets and sells licensed branded pharmaceuticals and specialist pet foods to the
veterinary professionals in Europe, as well as manufactures products for third party customers. The company also markets and sells
a range of endocrine, ophthalmic, dermatological, and equine products in North America. In addition, it develops and licenses
its branded veterinary product portfolio of novel and generic pharmaceuticals, and specialist pet diets.
Intellectual
Property
PetLife’s
initial licensed patent addresses the activities associated with the “polarization” of the scorpion venom solution
in order to enhance the targeted delivery to the specific cancerous cells in the dogs and cats. Additional continuation patents
will be prepared that more aggressively address the efficacy of the Polarized Blue Scorpion venom for pets and livestock. The
next continuation of the patent is Escozine for Pets GNP-1™, which is the drug version and will be the polarization of gold
nanoparticles in combination with Blue Scorpion Venom. The sister company, Medolife has already engaged a group of intellectual
property professionals (Hankin Patent Law) specifically skilled in the bio-pharmaceutical areas associated with the Escozine for
Pets™ product.
Regulatory
Issues
There
are worldwide, national, state and local rules, regulations and statutes that may impact the Company’s ability to fully
implement our strategic plan. The sale of animal health products is governed by the laws and regulations specific to each country
in which we sell our products. To maintain compliance with these regulatory requirements, we have established processes, systems
and dedicated resources with involvement from product concept to launch and maintenance in the market. In the majority of our
markets, the relevant animal health authority is separate from those governing human medicinal products.
United
States
The
regulatory body that is responsible for the regulation of animal health pharmaceuticals in the United States is the Center for
Veterinary Medicine (CVM), housed within the United States Food and Drug Administration (FDA). All manufacturers of animal health
pharmaceuticals must show their products to be safe, effective and produced by a consistent method of manufacture as defined under
the Federal Food, Drug and Cosmetic Act. Post-approval monitoring of products is required by law, with reports being provided
to the CVM’s Surveillance and Compliance group. Reports of product quality defects, adverse events or unexpected results
are produced in accordance with the law. Additionally, we are required to submit all new information for a product, regardless
of the source.
The
regulatory body in the United States for veterinary vaccines is the United States Department of Agriculture (USDA). The USDA’s
Center for Veterinary Biologics is responsible for the regulation of animal health vaccines, including immunotherapeutics. All
manufacturers of animal health biologicals must show their products to be pure, safe, effective and produced by a consistent method
of manufacture as defined under the Virus Serum Toxin Act. Post-approval monitoring of products is required. Reports of product
quality defects, adverse events or unexpected results are produced in accordance with the agency requirements.
Outside
of the United States
Country-specific
regulatory laws have provisions that include requirements for certain labeling, safety, efficacy and manufacturers’ quality
control procedures (to assure the consistency of the products), as well as company records and reports. With the exception of
the European Union, most other countries’ regulatory agencies will generally refer to the FDA, USDA, EU and other international
animal health entities, including the World Organization for Animal Health, Codex Alimentarius, in establishing standards and
regulations for veterinary pharmaceuticals and vaccines.
Employees
As
of August 31, 2015, we had 11 employees, of which three were executive officers. We have good relationships with our employees
and do not anticipate issues relative to our employees.
Item
1A. Risk Factors
Our
business and an investment in our securities are subject to a variety of risks. The following risk factors describe some of the
most significant events, facts or circumstances that could have a material adverse effect upon our business, financial condition,
results of operations, ability to implement our business plan and the market price for our securities. Many of these events are
outside of our control. If any of these risks actually occurs, our business, financial condition or results of operations may
be materially adversely affected. In such case, the trading price of our common stock could decline and investors in our common
stock could lose all or part of their investment.
Risks
Related to our Business
We
were formed on December 12, 2012 and have a limited operating history and accordingly may not be able to effectively operate our
business.
We
are still in the early stages of company development and accordingly, there is only a limited basis upon which to evaluate our
prospects for achieving our intended business objectives. There can be no assurance that we will ever achieve positive cash flow
or profitability, or that if either is achieved, that it will be at the levels estimated by management.
Prior
to the date of this annual report, we have not yet generated any revenue. Our failure to generate significant revenues would seriously
harm our business. Even if we are able to access capital, we anticipate that we will experience operating losses and incur significant
and increasing losses in the future due to growth, expansion, development and marketing. In the event that we are able to raise
adequate capital, we expect to significantly increase our sales and marketing and general and administrative expenses. As a result
of these additional expenses, we would need to generate substantial revenues to become profitable. We expect to incur significant
operating losses for at least the next several years.
Our
independent auditors have expressed substantial doubt about our ability to continue as a going concern, which may hinder our ability
to obtain future financing.
The
report of our independent auditors dated January 4, 2016 on our consolidated financial statements for the year ended August
31, 2015 included an explanatory paragraph indicating that there is substantial doubt about our ability to continue as a going
concern. Our auditors’ doubts are based on our incurring significant losses from operations and our working capital
deficit position. Our ability to continue as a going concern will be determined by our ability to obtain additional funding in
the short term to enable us to realize the commercialization of our planned business operations. Our consolidated financial
statements do not include any adjustments that might result from the outcome of this uncertain.
Our
Escozine for Pets™ product has not been approved by the FDA.
U.S.
pet owners may be hesitant to consider Escozine for Pets™ for treating cancer without FDA approval. Alternative cancer treatments
are not well-accepted in the U.S. The Company will need to educate and change the mindset of those who normally would not seek
medicinal alternatives for themselves or for their pets for us to be successful. Obtaining approval for our products will be a
lengthy and very costly process, the success of which cannot be assured.
Our
Brand is not well known in the United States.
Marketing
Escozine for Pets’ products will require the Company to establish branding reputation of credibility. U.S. pet owners and
veterinarians must become familiar with the products. Due to lack of brand recognition, significant advertising budgets will be
required to market and promote products in the U.S. Additional costs are required for a team of experts to seek approval of the
PetLife products in foreign markets. The brand initially may not be able to position itself on price, within the natural remedies
markets. While the product is less costly than chemotherapy treatments, it may still not be reasonably priced for entry in some
markets.
We
will require approval for the Products in Numerous Countries.
Even
though Medolife products have been approved for sale in six countries, our PetLife products may not be approved. If it is not
approved in the countries selected to market and promote the products, PetLife will need to consider secondary markets, which
may have limited sales potential. In addition, many chemotherapy drugs used by veterinarians today are used as “off label”
treatments with no FDA approval for veterinary use, regardless of successful use in treating humans. Veterinarians who have had
success with these drugs may be hesitant to try something new or more natural in treating pet cancers.
We
are highly dependent on the continued availability of our scorpion farm facilities and would be harmed if they were unavailable
for any prolonged period of time.
Any
failure in the physical infrastructure of our scorpion farm facilities or services could lead to significant costs and disruptions
that could reduce our revenues and harm our business reputation and financial results. We are highly reliant on our Dominican
Republic facilities. Any natural or man-made event that impacts our ability to utilize these facilities could have a significant
impact on our operating results, reputation and ability to continue operations. Our ability to rebuild facilities would take a
considerable amount of time and expense and cause a significant disruption in service to our customers.
Though
PetLife sister company, Medolife, is working with venom experts from the Instituto Butantan, São Paulo, Brazil, in applying
their methods and experience and knowhow to manage and grow a large population of scorpions necessary to make sufficient amounts
of the product; demand for the venom may increase, particularly if nutraceutical or pharmaceutical companies decide they want
to enter the market. This could decrease the availability of scorpion venom which would result in price increases for PetLife.
We
are not currently profitable and we will need to raise additional funds in the future; however, additional funds may not be available
on acceptable terms, or at all.
We
have substantial operating expenses associated with the sales and marketing of our products. The sales and marketing expenses
are anticipated to be funded from operating cash flow and from potential financing transactions. There can be no assurance that
we will have sufficient access to liquidity or cash flow to meet our operating expenses and other obligations. If we do not increase
our revenue or reduce our expenses, we will need to raise additional capital, which would result in dilution to our stockholders,
or seek additional loans. The incurrence of indebtedness would result in increased debt service obligations and could require
us to agree to operating and financial covenants that would restrict our operations. Financing may not be available in amounts
or on terms acceptable to us, if at all. Any failure by us to raise additional funds on terms favorable to us, or at all, could
result in our inability to pay our expenses as they come due, limit our ability to expand our business operations, and harm our
overall business prospects.
We
may not be able to raise capital or, if we can, it may not be on favorable terms. We may seek to raise additional capital through
public or private equity financings, partnerships, joint ventures, dispositions of assets, debt financings or restructurings,
bank borrowings or other sources. To obtain additional funding, we may need to enter into arrangements that require us to relinquish
rights to certain technologies, products and/or potential markets. If adequate funds are not otherwise available, we would be
forced to curtail operations significantly, including reducing our sales and marketing expenses which could negatively impact
product sales and we could even be forced to cease operations, liquidate our assets and possibly even seek bankruptcy protection.
We
operate in a highly regulated environment, and any legal or regulatory action could be time-consuming and costly.
If
we fail to comply with all applicable laws, standards and regulations, action by the FDA or other regulatory agencies could result
in significant restrictions, including restrictions on the marketing or use of our products or the withdrawal of products from
the market. Any such restrictions or withdrawals could materially affect our business and operations. In addition, governmental
authorities could impose fines, seize our inventory of products, or force us to recall any product already in the market if we
fail to comply with governmental regulations.
Competitive
products exist and more will be developed, and we may not be able to successfully compete because we are smaller and have fewer
financial resources.
Our
business is in a very competitive and evolving field. Rapid new developments in this field have occurred over the past few years,
and are expected to continue to occur. Other companies already have competing products available or may develop products to compete
with ours. Many of these products have short regulatory timeframes and our competitors, many with more substantial development
resources, may be able to develop competing products that are equal to or better than ours. This may make our products obsolete
or undesirable by comparison and reduce our revenue. Our success will depend, in large part, on our ability to maintain a competitive
position concerning our intellectual property, and to develop new technologies and new applications for our technologies. Many
of our competitors have substantially greater financial and technical resources, as well as greater production and marketing capabilities,
and our ability to compete remains uncertain.
We
will need to continue to innovate and develop new products to be desirable to our customers.
The
markets for our products and services are characterized by rapid technological change, frequent new introductions, changes in
customers’ demands and evolving industry standards. Accordingly, we will need to continue to innovate and develop additional
products. These efforts can be costly, subject to long development and regulatory delays and may not result in products approved
for sale. These costs may hurt operating results and may require additional capital. If additional capital is not available, we
may be forced to curtail development activities. In addition, any failure on our behalf to react to changing market conditions
could create an opportunity for other market participants to capture a critical share of the market within a short period of time.
Our
success will depend on our ability to engage and retain qualified technical personnel who are difficult to attract.
Our
success will depend on our ability to attract and retain qualified technical personnel to assist in research and development,
testing, product implementation, low-scale production and technical support. The demand for such personnel is high and the supply
of qualified technical personnel is limited. A significant increase in the wages paid by competing employers could result in a
reduction of our technical work force and increases in the wage rates that we must pay or both. If either of these events were
to occur, our cost structure could increase and our growth potential could be impaired.
Loss
of key members of our management who we need to succeed could adversely affect our business.
We
are highly dependent on the services of key members of our management team, and the loss of any of their services could have an
adverse effect on our future operations. We do not currently maintain key-man life insurance policies insuring the life of any
member of our management team.
We
will be required to invest in facilities and equipment on a continuing basis, which will put pressure on us to finance these investments.
We
have invested, and intend to continue to invest, in facilities and state-of-the-art equipment in order to increase, expand or
update our capabilities and facilities. Changes in technology or sales growth beyond currently established production capabilities,
which we anticipate, will require further investment. However, there can be no assurance that we will generate sufficient funds
from operations to maintain our existing facilities and equipment or to finance any required capital investments or that other
sources of funding will be available. Additionally, there can be no guarantee that any future expansion will not negatively affect
earnings.
Future
revenue will depend on our ability to increase sales.
We
intend to sell our products through numerous means, including direct sales by our employees, through infomercials and through
a multi-level marketing program. We have and will continue to incur increased sales and marketing expenses in building and expanding
our sales force, and there can be no assurance that we will generate increased sales as a result of this effort.
Our
operating results will be harmed if we are unable to effectively manage and sustain our future growth.
We
might not be able to manage our future growth efficiently or profitably. Our business is unproven on a large scale and actual
revenue and operating margins, or revenue and margin growth, may be less than expected. If we are unable to scale our production
capabilities efficiently, we may fail to achieve expected operating margins, which would have a material and adverse effect on
our operating results. Growth may also stress our ability to adequately manage our operations, quality of products, safety and
regulatory compliance. In order to grow, we may be required to obtain additional financing, which may increase our indebtedness
or result in dilution to our stockholders. Further, there can be no assurance that we would be able to obtain any additional financing.
Our
success depends on our ability to avoid infringing on the intellectual property rights of third parties which could expose us
to litigation or commercially unfavorable licensing arrangements.
Our
commercial success depends in part on our ability to avoid infringing patents and proprietary rights of third parties. Third parties
may accuse us of employing their proprietary technology in our products, or in the materials or processes used to research or
develop our products, without authorization. Any legal action against us claiming damages and/or seeking to stop our commercial
activities relating to the affected products, materials and processes could, in addition to subjecting us to potential liability
for damages, require us to obtain a license to continue to utilize the affected materials or processes or to manufacture or market
the affected products. We cannot predict whether we would prevail in any of these actions or whether any license required under
any of these patents would be made available on commercially reasonable terms, if at all. If we are unable to obtain such a license,
we may be unable to continue to utilize the affected materials or processes or manufacture or market the affected products or
we may be obligated by a court to pay substantial royalties and/or other damages to the patent holder. Even if we are able to
obtain such a license, the terms of such a license could substantially reduce the commercial value of the affected product or
products and impair our prospects for profitability. Accordingly, we cannot predict whether or to what extent the commercial value
of the affected product or products or our prospects for profitability may be harmed as a result of any of the liabilities discussed
above. Furthermore, infringement and other intellectual property claims, with or without merit, can be expensive and time-consuming
to litigate and can divert management’s attention from our core business. We may be unable to obtain and enforce intellectual
property rights to adequately protect our products and related intellectual property.
Our
business will become subject to continuing regulatory compliance by the FDA and other authorities which is costly and could result
in delays in the commercialization of our products.
Upon
completion of the FDA process for our approved drug Escozine for Pets product, we will become subject to extensive regulation
by the FDA and potentially other federal governmental agencies and, in some jurisdictions, by state and foreign governmental authorities.
These regulations govern the introduction of new pharmaceuticals even for pets, the observance of certain standards with respect
to the design, manufacture, testing, labeling, promotion and sales of the pharmaceuticals, the maintenance of certain records,
the ability to track devices, the reporting of potential problems, and other matters.
Future
revenue will depend on our ability to develop new sales channels and there can be no assurance that these efforts will result
in significant revenues.
We
are heavily dependent on developing sales channels for our products but there can be no assurance that these channels can be developed
or that we will continue to be successful in selling our products. We are engaging in a major initiative to build and further
expand our direct sales force, as well as develop infomercial and multi-level marketing sales. This effort will have significant
costs that will be incurred prior to the generation of revenue sufficient to cover these costs. The costs incurred for these efforts
may impact our operating results and there can be no assurance of their effectiveness. Many of our competitors have well-developed
sales channels and it may be difficult for us to break through these competitors to take market share. If we are unable to develop
these sales channels, we may not be able to grow revenue or maintain our current level of revenue generation.
Risks
Related to our Stock
There
may be fluctuations in our operating results, which will impact our stock price.
Significant
annual and quarterly fluctuations in our results of operations may be caused by, among other factors, our volume of revenues,
the timing of new product or service announcements, releases by us and our competitors in the marketplace of new products or services,
seasonality and general economic conditions. There can be no assurance that the level of revenues achieved by us in any particular
fiscal period will not be significantly lower than in other comparable fiscal periods. Our expense levels are based, in part,
on our expectations as to future revenues. As a result, if future revenues are below expectations, net income or loss may be disproportionately
affected by a reduction in revenues, as any corresponding reduction in expenses may not be proportionate to the reduction in revenues.
Because
we became public through a reverse merger, we may not be able to attract the attention of major brokerage firms or certain investors.
There
are coverage risks associated with our becoming public through a reverse merger, including, among other things, security analysts
of major brokerage firms may not provide coverage of us since there is no incentive to brokerage firms to recommend the purchase
of our common stock. In addition, we may not attract the attention of major brokerage firms and certain investors due to the possibility
of a low stock price. We cannot assure you that brokerage firms would want to conduct any public offerings on our behalf in the
future.
The
market price of our common stock is extremely volatile, which may affect our ability to raise capital in the future and may subject
the value of your investment to sudden decreases.
The
market price for securities of biotechnology companies, including ours, historically has been highly volatile, and the market
from time to time has experienced significant price and volume fluctuations that are unrelated to the operating performance of
such companies. Fluctuations in the trading price or liquidity of our common stock may harm the value of your investment in our
common stock.
Our
stockholders may experience significant dilution if future equity offerings are used to fund operations or acquire complementary
businesses.
If
our future operations or acquisitions are financed through the issuance of equity securities, our stockholders could experience
significant dilution. In addition, securities issued in connection with future financing activities or potential acquisitions
may have rights and preferences senior to the rights and preferences of our common stock.
We
do not anticipate paying dividends in the foreseeable future; you should not buy our stock if you expect dividends.
We
currently intend to retain our future earnings to support operations and to finance expansion and, therefore, we do not anticipate
paying any cash dividends on our common stock in the foreseeable future.
Our
current management can exert significant influence over us and make decisions that are not in the best interests of all stockholders.
As
of August 31, 2015, our executive officers and directors beneficially owned as a group approximately 51.0% of our outstanding
shares of common stock. These officers continue to own a controlling interest in the stock. As a result, these stockholders will
be able to assert significant influence over all matters requiring stockholder approval, including the election and removal of
directors and any change in control. In particular, this concentration of ownership of our outstanding shares of common stock
could have the effect of delaying or preventing a change in control, or otherwise discouraging or preventing a potential acquirer
from attempting to obtain control. This, in turn, could have a negative effect on the market price of our common stock. It could
also prevent our stockholders from realizing a premium over the market prices for their shares of common stock. Moreover, the
interests of the owners of this concentration of ownership may not always coincide with our interests or the interests of other
stockholders and, accordingly, could cause us to enter into transactions or agreements that we would not otherwise consider.
Our
common stock is considered “penny stock” and may be difficult to sell.
The
SEC has adopted Rule 3a51-1, which establishes the definition of a “penny stock” for the purposes relevant to us,
as any equity security that has a market price of less than $5.00 per share or with an exercise price of less than $5.00 per share,
subject to certain exceptions. The market price of our common stock is less than $5.00 per share and therefore may be designated
as a “penny stock” according to SEC rules. For any transaction involving a penny stock, unless exempt, Rule 15g-9
requires:
●
that a broker or dealer approve a person’s account for transactions in penny stocks; and
●
that the broker or dealer receives from the investor a written agreement to the transaction, setting forth the identity and quantity
of the penny stock to be purchased.
In
order to approve a person’s account for transactions in penny stocks, the broker or dealer must:
●
obtain financial information and investment experience objectives of the person; and
●
make a reasonable determination that the transactions in penny stocks are suitable for that person and the person has sufficient
knowledge and experience in financial matters to be capable of evaluating the risks of transactions in penny stocks. The broker
or dealer must also deliver, prior to any transaction in a penny stock, a disclosure schedule prescribed by the SEC relating to
the penny stock market, which, in highlight form:
●
sets forth the basis on which the broker or dealer made the suitability determination; and
●
that the broker or dealer received a signed, written agreement from the investor prior to the transaction.
Disclosure
also has to be made about the risks of investing in penny stocks in both public offerings and in secondary trading and about the
commissions payable to both the broker-dealer and the registered representative, current quotations for the securities and the
rights and remedies available to an investor in cases of fraud in penny stock transactions. Finally, monthly statements have to
be sent disclosing recent price information for the penny stock held in the account and information on the limited market in penny
stocks.
Generally,
brokers may be less willing to execute transactions in securities subject to the “penny stock” rules. This may make
it more difficult for investors to dispose of our Common Stock and cause a decline in the market value of our stock. In addition,
since the Common Stock is currently traded on the OTC Bulletin Board, investors may find it difficult to obtain accurate quotations
of the Common Stock and may experience a lack of buyers to purchase such stock or a lack of market makers to support the stock
price.
We
could issue “blank check” preferred stock without stockholder approval with the effect of diluting then current stockholder
interests and impairing their voting rights, and provisions in our charter documents and under Nevada law could discourage a takeover
that stockholders may consider favorable.
Our
certificate of incorporation provides for the authorization to issue up to 50,000,000 shares of “blank check” preferred
stock with designations, rights and preferences as may be determined from time to time by our board of directors. Our board of
directors is empowered, without stockholder approval, to issue one or more series of preferred stock with dividend, liquidation,
conversion, voting or other rights which could dilute the interest of, or impair the voting power of, our common stockholders.
The issuance of a series of preferred stock could be used as a method of discouraging, delaying or preventing a change in control.
For example, it would be possible for our board of directors to issue preferred stock with voting or other rights or preferences
that could impede the success of any attempt to change control of our company.
Item
1B. Unresolved Staff Comments
In
connection with the acquisition of Petlife Corporation the Securities and Exchange Commission requested that we provide the historical
financial statements of the business acquired. Those financial statements are provided with this Annual Report on Form 10-K and
the 8-K filed in connection with the acquisition will be amended concurrently to reflect those financial statements.
Item
2. Properties
Properties
We
presently lease our principal executive offices, located at 433 N. Camden Dr., Beverly Hills, CA 90210. We believe that our present
business property is adequate and suitable to meet our needs until we consummate a business combination.
The
PetLife Scorpion Reservation, owned and maintained by Medolife, an affiliated company through common ownership, is located in
a remote hill area approximately 4 hours driving time from Santo Domingo, the capital of the Dominican Republic. The farm includes
approximately 50,000 square meters (12 acres) of land of which 60% is usable for scorpion habitat. Through an agreement with Medolife,
Petlife currently has access to 41,500 scorpions with the potential post funding capacity for up to one million scorpions and
can currently produce 500,000 units of Escozine for Pets per month.
Strategically,
Medolife is looking to increase the size of its Scorpion Reservation in order to allow for a reduction in the scorpion density
per population unit as well as build an onsite state of the art laboratory which will also house the scorpion milking center and
interior scorpion breeding and habitat to provide production insurance against unanticipated weather / environmental catastrophes.
The reservation will be managed by Medolife specialists including handlers, milkers, biologists and reservation managers.
Medolife,
has an alliance with Laboratório de Artrópodes, Instituto Butantan in Sao Paolo, Brazil. Instituto Butanan are experts
in the maintenance and breeding of Scorpions. They have 20 years of experience and are willing to share their experience and methodology
of Scorpion maintenance and breeding with Medolife. They have been using the Scorpion venom effectively for the production of
a Scorpion antidote medication. Medolife will implement their 20 years of experience to ensure an efficient scorpion maintenance
and breeding operation.
PetLife
also anticipates working with global venom research leaders such as Venomics (a French Government funded research group of companies
from five European Union countries) and Venomtech (Specialists in Venom technology) in France, to ensure the latest research is
utilized to ensure the most effective results from PetLife’s products. PetLife will also utilize the results and benefits
from other venom based research and approved products; for example, the FDA approved scorpion antidote, Anascorp.
Item
3. Legal Proceedings
We
are not currently a party in any legal proceeding or governmental regulatory proceeding nor are we currently aware of any pending
or potential legal proceeding or governmental regulatory proceeding proposed to be initiated against us.
Item
4. Mine Safety Disclosures
Not
applicable.
PART
II
Item
5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Trading
Information
Our
common stock trades in the over-the-counter market and is currently quoted on the OTCQB maintained by OTC Markets Group, Inc.
under the symbol PTLF.
Below
is a table indicating the range of high and low closing price information for the common stock as reported by the OTC Markets
Group for the periods listed. These prices do not necessarily reflect actual transactions.
| | | |
High | | |
Low | |
| Through
December 24, 2015 | | |
$ | 0.04 | | |
$ | 0.01 | |
| August 31, 2015 | | |
| 0.04 | | |
| 0.01 | |
| May 31, 2015 | | |
| 0.04 | | |
| 0.01 | |
| February 28, 2015 | | |
| 0.03 | | |
| 0.01 | |
| November 30, 2014 | | |
| 0.03 | | |
| 0.01 | |
| August 31, 2014 | | |
| 0.03 | | |
| 0.01 | |
| May 31, 2014 | | |
| 0.71 | | |
| 0.12 | |
| February 28, 2014 | | |
| 0.68 | | |
| 0.12 | |
| November 30, 2013 | | |
| 0.90 | | |
| 0.33 | |
| August 31, 2013 | | |
| 0.88 | | |
| 0.18 | |
Transfer
Agent
Our
current transfer agent and registrar for our common stock is Empire Stock Transfer, 1859 Whitney Mesa Drive, Henderson, NV 89014.
As
of August 31, 2015, there were 132 reported holders of record of our common stock and a total of 58,532,156 shares of Common
Stock issued and outstanding.
We
have never paid a cash dividend. Payment of future dividends, if any, will be at the discretion of our Board of Directors after
taking into account various factors, including the terms of any credit arrangements, our financial condition, operating results,
current and anticipated cash needs and plans for growth. Our initial earnings, if any, will likely be retained to finance our
growth. At the present time, we are not party to any agreement that would limit our ability to pay dividends.
The
Securities Enforcement and Penny Stock Reform Act of 1990
The
Securities and Exchange Commission has adopted rules that regulate broker-dealer practices in connection with transactions in
penny stocks. Penny stocks are generally equity securities with prices of less than $5.00 (other than securities registered on
certain national securities exchanges or quoted on the NASDAQ system, provided that current prices and volume information with
respect to transactions in such securities is provided by the exchange or system). Our shares are currently subject to the penny
stock rules.
A
purchaser is purchasing penny stock which limits the ability to sell the stock. The classification of penny stock makes it more
difficult for a broker-dealer to sell the stock into a secondary market, which makes it more difficult for a purchaser to liquidate
his/her investment. Any broker-dealer engaged by the purchaser for the purpose of selling his or her shares in us will be subject
to Rules 15g-1 through 15g-10 of the Securities and Exchange Act. Rather than creating a need to comply with those rules, some
broker-dealers will refuse to attempt to sell penny stock.
The
penny stock rules require a broker-dealer, prior to a transaction in a penny stock not otherwise exempt from those rules, to deliver
a standardized risk disclosure document prepared by the Commission, which:
| ● |
contains
a description of the nature and level of risk in the market for penny stocks in both public offerings and secondary trading; |
| |
|
| ● |
contains
a description of the broker’s or dealer’s duties to the customer and of the rights and remedies available to the
customer with respect to a violation to such duties or other requirements of the Securities Act of 1934, as amended; |
| |
|
| ● |
contains
a brief, clear, narrative description of a dealer market, including “bid” and “ask” prices for penny
stocks and the significance of the spread between the bid and ask prices; |
| |
|
| ● |
contains
a toll-free telephone number for inquiries on disciplinary actions; |
| |
|
| ● |
defines
significant terms in the disclosure document or in the conduct of trading penny stocks; and |
| |
|
| ● |
contains
such other information and is in such form (including language, type, size and format) as the Securities and Exchange Commission
shall require by rule or regulation. |
| |
|
| ● |
The
broker-dealer also must provide, prior to effecting any transaction in a penny stock, to the customer: |
| |
|
| ● |
the
bid and offer quotations for the penny stock; |
| |
|
| ● |
the
compensation of the broker-dealer and its salesperson in the transaction; |
| |
|
| ● |
the
number of shares to which such bid and ask prices apply, or other comparable information relating to the depth and liquidity
of the market for such stock; and |
| |
|
| ● |
monthly
account statements showing the market value of each penny stock held in the customer’s account. |
In
addition, the penny stock rules require that prior to a transaction in a penny stock not otherwise exempt from those rules; the
broker-dealer must make a special written determination that the penny stock is a suitable investment for the purchaser and receive
the purchaser’s written acknowledgment of the receipt of a risk disclosure statement, a written agreement to transactions
involving penny stocks, and a signed and dated copy of a written suitability statement. These disclosure requirements have the
effect of reducing the trading activity in the secondary market for our stock. Thus, stockholders may have difficulty selling
their securities.
Securities
Authorized for Issuance under Equity Compensation Plans
Equity
Compensation Plan Information
The
following sets forth information for the equity compensation plans outstanding as of August 31, 2015 (including individual compensation
arrangements) under which shares of our common stock are authorized for issuance:
Equity
Compensation Plan Information
| | |
Number of securities
| |
| | | |
| | |
| | |
to be issued upon
| |
| Weighted
average | | |
| Number
of securities | |
| | |
exercise of | |
| exercise
price of | | |
| remaining
available for | |
| | |
outstanding options,
| |
| outstanding
options, | | |
| future
issuance as of | |
| Plan
Category | |
warrants
and rights | |
| warrants
and rights | | |
| August
31, 2015 | |
| | |
| |
| | | |
| | |
| Equity compensation plans approved by
security holders: | |
- | |
| - | | |
| - | |
| | |
| |
| | | |
| | |
| Equity compensation plans not approved
by security holders: | |
- | |
| - | | |
| - | |
Item
6. Selected Financial Data.
Not
required by smaller reporting companies.
Item
7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
The
following discussion provides information that management believes is relevant to an assessment and understanding of the financial
condition and results of operations of Petlife Pharmaceuticals, Inc., formerly Clear TV Ventures, Inc. (the “Company”).
This
discussion addresses matters we consider important for an understanding of our financial condition and results of operations for
the years ended August 31, 2015 and 2014. It consists of the following subsections:
“Introduction
and Plan of Operation” which provides a brief summary of our consolidated results and financial position and the primary
factors affecting those results, as well as a summary of our expectations for fiscal 2016;
“Liquidity
and Capital Resources,” which contains a discussion of our cash flows and liquidity, investing activities and financing
activities, contractual obligations, and critical obligations;
“Results
of Operations and Comparison”,” which sets forth an analysis of the operating results for the last two fiscal years
“Critical
Accounting Policies,” which provides an analysis of the accounting policies we consider critical because of their effect
on the reported amounts of assets, liabilities, income and/or expenses in our consolidated financial statements and/or because
they require difficult, subjective or complex judgments by our management;
“Recent
Accounting Pronouncements and Developments,” which summarizes recently published authoritative accounting guidance, how
it might apply to us and how it might affect our future results.
This
item should be read in conjunction with our consolidated financial statements and the notes thereto included in this annual report.
Introduction
and Plan of Operation
The
following discussion updates our plan of operation for the 2016 Fiscal Year. The discussion also summarizes the results
of our operations for the years ended August 31, 2015 and 2014.
Merger
Agreements
On
or about April 18, 2014, the Company entered into a Share Exchange Agreement (the “Share Exchange”) with Petlife
Corporation, a Delaware corporation (“Petlife”) and the shareholders of Petlife Corporation (the “Shareholders”)
for the exchange of all of the issued and outstanding shares of Petlife. Effective at the Closing on August 12, 2014, these
shares of Petlife were exchanged for approximately 47,000,000 fully paid non-assessable shares of the Company reflecting approximately
80% of the issued and outstanding shares of the Company and acquisition of all assets and liabilities of Petlife Corporation and
was valued equated to the net assets of Petlife Corporation as of the date of the acquisition. For accounting purposes, Petlife
was the accounting acquirer and or the surviving entity. Accordingly, the historical financial statements are those of Petlife,
the accounting acquirer, immediately following the consummation of the reverse merger.
On
June 26, 2014, Clear TV Ventures, Inc. entered into an Agreement and Plan of Merger with its wholly-owned subsidiary, Petlife
Pharmaceuticals, Inc., a Nevada Corporation. As part of that merger, the name of the Company was changed to Petlife Pharmaceuticals,
Inc. and the Company exchanged one share for every 15 shares of Clear TV Ventures. Effective August 12, 2014 we completed the
closing of the Share Exchange Agreement and the acquisition of Petlife Corporation.
Plan
of Operations
For
the years ended August 31, 2015 and 2014, we experienced
the negative effects of the financial markets upheaval, which made capital acquisition extremely difficult.
We
will be developing neutraceuticals and FDA approved prescription drugs for veterinary use for retail sales and distribution throughout
the world.
About
Petlife Pharmaceuticals, Inc.
PetLife
Pharmaceuticals, Inc. (PetLife) has developed and is launching a new generation of potentiated veterinary cancer medications and
nutraceuticals, based on the same patented formula “Escozine” and production processes that have been scientifically
proven as an effective treatment for cancer in humans for years. Escozine (for humans) is currently sold as either a nutraceutical
or prescription drug in 40 countries including the United States.
Petlife’s
primary goal is to bring its scientifically proven, potentiated bioactive medication and Nutraceuticals to the world of veterinary
oncology, with the ultimate goal of extending the life of pets with cancer and improving their quality of life. In the process
of achieving these objectives, Petlife will transition into a world renowned, professionally respected veterinary pharmaceutical
company that will create new industry standards as well as being profitable and innovative.
Results
of Operations
Year
ended August 31, 2015 compared to period ended August 31, 2014
Net
loss
We
incurred a net loss of $1,935,599 for the year ended August 31, 2015 as compared to the net loss we incurred for the year ended
August 31, 2014 of $1,512,051. The increase in loss is attributable principally to increased administrative expenses as
well as an increase in stock-based compensation.
Operating
expenses
For
the year ended August 31, 2015 we incurred $1,935,599 in operating expenses as compared to $1,512,051 in operating expenses
we incurred for the prior year. Operating expenses for the year ended August 31, 2015 increased due to the commencement of
operations of our PetLife products business as well as an increase in stock-based compensation.
Loss
from Operations
For
the year ended August 31, 2015 we incurred a loss from operations of $1,935,599 as compared to the operating loss we incurred
for the year ended August 31, 2014 of $1,512,051.
Liquidity
and Capital Resources
The
Company has been in the development stage and received no revenue from business operations for the years ended August 31, 2015
and 2014. We have consequently relied on funds received in connection with our equity and debt offerings to finance our
ongoing operations. We have experienced net losses since inception, and we expect we will continue to incur losses for the
next year. As of the date of this filing, we do not have any available external source of funds. We require additional
capital in the near term to maintain our current operations. Although we are actively seeking additional equity and debt
financing, such financing may not be available on acceptable terms, if at all.
Our
consolidated financial statements have been prepared assuming that we will continue as a going concern. Since our inception on
December 12, 2012, we have not generated revenue and have incurred net loss. Accordingly, we have not generated cash flow from
operations and have primarily relied upon loans from officers, promissory notes and advances from related parties, and equity
financing to fund our operations. The report of our Independent Registered Public Accounting Firm include an explanatory paragraph
indicating that there is substantial doubt about our ability to continue as a going concern.
We
do not have sufficient cash resources or current assets to pay our obligations, and we have been meeting many of our obligations
through the issuance of our common stock to our employees, consultants and advisors as payment for goods and services. Considering
the foregoing, we are dependent on additional financing to continue our operations. Our significant capital requirements for the
foreseeable future include development and operational costs, and our corporate overhead expenses.
We
are actively seeking additional equity or debt financing. However, there can be no assurance that funds required during the next
twelve months or thereafter will be available from external sources. The lack of additional capital resulting from the inability
to generate cash flow from operations or to raise capital from external sources would force us to substantially curtail or cease
operations and would, therefore, have a material adverse effect on our business. Further, there can be no assurance that any such
required funds, if available, will be available on attractive terms or that they will not have a significantly dilutive effect
on our existing shareholders. All of these factors have been exacerbated by the extremely unsettled credit and capital markets
presently existing.
Net
Cash Used in Operating Activities
Net
cash used in operating activities was $22,034 for the year ended August 31, 2015, as compared to net cash used of $267,066
in prior year’s operating activities. The decrease was primarily due to the increase in current liabilities for our PetLife
business in 2015 as compared to the activity incurred in 2014.
Net
Cash Used in Investing Activities
The
Company used $600 for capital expenditures for the year end August 31, 2015. For the year end August 31, 2014 the Company received
$100,000 in connection with the reverse merger. The proceeds were primarily used for merger expenses.
Net
Cash Provided by Financing Activities
During
the year ended August 31, 2015, we received net cash provided by financing activities totaling $10,000 from a shareholder advance.
During the year ended August 31, 2014, we received net cash provided by financing activities totaling $173,918 primarily from
the sale of common stock.
Going
Concern
The
accompanying consolidated financial statements have been prepared on a going concern basis, which contemplates the realization
of assets and the satisfaction of liabilities in the normal course of business. The Company has not established any sources
of revenue to cover its operating expenses for the years ended August 31, 2015 and 2014. In addition, the Company
has incurred deficit accumulated during development stage of $3,447,688 and used $22,034 in cash for operating activities
for the year ended August 31, 2015. These factors raise substantial doubt about the Company’s ability to continue
as a going concern.
The
Company’s ability to continue existence is dependent upon commencing its planned operations, management’s ability
to develop and achieve profitable operations and/or upon obtaining additional financing to carry out its planned business. The
Company intends to fund its business development, acquisition endeavors and operations through equity and debt financing arrangements.
There
can be no assurance that any additional financings will be available to the Company on satisfactory terms and conditions, if at
all. In the event the Company is unable to continue as a going concern, it may elect or be required to seek protection from its
creditors by filing a voluntary petition in bankruptcy or may be subject to an involuntary petition in bankruptcy. To date, management
has not considered this alternative, nor does management view it as a likely occurrence.
The
accompanying consolidated financial statements do not include any adjustments related to the recoverability or classification
of asset-carrying amounts or the amounts and classification of liabilities that might be necessary should the Company be unable
to continue as a going concern.
Critical
Accounting Policies and Estimates
Estimates
The
preparation of the financial statements in conformity with generally accepted accounting principles requires management to make
estimates and assumptions that affect certain reported amounts and disclosures. Accordingly, actual results could differ from
those estimates.
Revenue
Recognition
The
Company will recognize revenue in accordance with Accounting Standards Codification subtopic 605-10, Revenue Recognition (“ASC
605-10”) which requires that four basic criteria must be met before revenue can be recognized: (1) persuasive evidence of
an arrangement exists; (2) delivery has occurred; (3) the selling price is fixed and determinable; and (4) collectability is reasonably
assured. Determination of criteria (3) and (4) are based on management’s judgments regarding the fixed nature of the selling
prices of the products delivered and the collectability of those amounts. Provisions for discounts and rebates to customers, estimated
returns and allowances, and other adjustments will be provided for in the same period the related sales will be recorded.
ASC
605-10 incorporates Accounting Standards Codification subtopic 605-25, Multiple-Element Arrangements (“ASC 605-25”).
ASC 605-25 addresses accounting for arrangements that may involve the delivery or performance of multiple products, services and/or
rights to use assets. There was no effect on implementing ASC 605-25 on the Company’s financial position and results of
operations, since the Company has not started generating revenue.
Cash
The
Company considers cash to consist of cash on hand and temporary investments having an original maturity of 90 days or less that
are readily convertible into cash.
Property
and Equipment
Property
and equipment are stated at cost. When retired or otherwise disposed, the related carrying value and accumulated depreciation
are removed from the respective accounts and the net difference less any amount realized from disposition, is reflected in earnings.
Long-Lived
Assets
The
Company follows FASB ASC 360-10-15-3, “Impairment or Disposal of Long-lived Assets,” which established a “primary
asset” approach to determine the cash flow estimation period for a group of assets and liabilities that represents the unit
of accounting for a long-lived asset to be held and used. Long-lived assets to be held and used are reviewed for impairment whenever
events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. The carrying amount of
a long-lived asset is not recoverable if it exceeds the sum of the undiscounted cash flows expected to result from the use and
eventual disposition of the asset. Long-lived assets to be disposed of are reported at the lower of carrying amount or fair value
less cost to sell.
Income
Taxes
The
Company has adopted Accounting Standards Codification subtopic 740-10, Income Taxes (“ASC 740-10”) which requires
the recognition of deferred tax liabilities and assets for the expected future tax consequences of events that have been included
in the financial statement or tax returns. Under this method, deferred tax liabilities and assets are determined based on the
difference between financial statements and tax basis of assets and liabilities using enacted tax rates in effect for the year
in which the differences are expected to reverse. Temporary differences between taxable income reported for financial reporting
purposes and income tax purposes consist primarily of timing differences such as deferred officers’ compensation and stock
based compensation accounting.
Net
Loss per Common Share, basic and diluted
The
Company has adopted Accounting Standards Codification subtopic 260-10, Earnings Per Share (“ASC 260-10”) specifying
the computation, presentation and disclosure requirements of earnings per share information. Basic loss per share has been calculated
based upon the weighted average number of common shares outstanding.
Stock
based compensation
The
Company follows Accounting Standards Codification subtopic 718-10, Compensation (“ASC 718-10”) which requires that
all share-based payments to both employees and non-employees be recognized in the income statement based on their fair values.
As
of August 31, 2015, the Company did not have any issued or outstanding stock options.
Concentrations
of Credit Risk
Financial
instruments and related items, which potentially subject the Company to concentrations of credit risk, consist primarily of cash,
cash equivalents and trade receivables. The Company places its cash and temporary cash investments with high credit quality institutions.
At times, such investments may be in excess of the FDIC insurance limit.
Research
and Development
The
Company accounts for research and development costs in accordance with Accounting Standards Codification subtopic 730-10, Research
and Development (“ASC 730-10”). Under ASC 730-10, all research and development costs must be charged to expense
as incurred. Accordingly, internal research and development costs are expensed as incurred. Third-party research and
development costs are expensed when the contracted work has been performed or as milestone results have been achieved as defined
under the applicable agreement. Company-sponsored research and development costs related to both present and future products
are expensed in the period incurred. The Company incurred costs of $0 and $73,198 for research and development for the years
ended August 31, 2015 and 2014.
Reliance
on Key Personnel and Consultants
The
Company has eleven full-time employees, three of which are executive officers. Additionally, the Company has consultants performing
various specialized services. The Company is heavily dependent on the continued active participation of these current executive
officers, employees and key consultants. The loss of any of the senior management or key consultants could significantly and negatively
impact the business until adequate replacements can be identified and put in place.
Fair
Value
Accounting
Standards Codification subtopic 825-10, Financial Instruments (“ASC 825-10”) requires disclosure of the fair value
of certain financial instruments. The carrying amount reported in the unaudited condensed consolidated balance sheet for accounts
payable and accrued expenses, advances and notes payable approximates fair value because of the immediate or short-term maturity
of these financial instruments.
Recent
Accounting Pronouncements
In
May 2014, the FASB issued ASU 2014-09, “Revenue from Contracts with Customers (Topic 606).” This guidance outlines
a single, comprehensive model for accounting for revenue from contracts with customers. We will adopt the standard in 2018. While
we are currently assessing the impact of the new standard, we do not expect this new guidance to have a material impact on our
Consolidated Financial Statements.
No
other new accounting pronouncement issued or effective during the fiscal year had or is expected to have a material impact on
the Consolidated Financial Statements.
Off-Balance
sheet Arrangements
We
do not maintain off-balance sheet arrangements nor do we participate in any non-exchange traded contracts requiring fair value
accounting treatment other than the operating leases as disclosed in Item 2, Properties and Notes to Consolidated Financial Statements
(Note 10).
Item
7A. Quantitative and Qualitative Disclosures About Market Risk
This
item is not applicable to smaller reporting companies.
Item
8. Financial Statements and Supplementary Data
Set
for below beginning with page F-1.
Item
9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
On
December 1, 2015 the Company engaged LBB & Associates Ltd., LLP (“LBB”) as its independent registered public accounting
firm for the Company’s fiscal years ended August 31, 2014 and 2013. LBB remains the Company’s accounting firm.
On
August 16, 2011 (the “Engagement Date”), the Company engaged RBSM LLP (“RBSM”) as its independent registered
public accounting firm for the Company’s fiscal year ended August 31, 2011. RBSM remained the Company’s independent
registered public accounting firm for the fiscal years ended August 31, 2012 and 2013. The decision to engage RBSM as the Company’s
independent registered public accounting firm was approved by the Company’s Board of Directors.
During
the two most recent fiscal years and through the Engagement Date, the Company has not consulted with LBB or RBSM regarding either:
1.
the application of accounting principles to any specified transaction, either completed or proposed, or the type of audit opinion
that might be rendered on the Company’s financial statements, and neither a written report was provided to the Company nor
oral advice was provided that RBSM concluded was an important factor considered by the Company in reaching a decision as to the
accounting, auditing or financial reporting issue; or
2.
any matter that was either the subject of a disagreement (as defined in paragraph (a)(1)(iv) of Item 304 of Regulation S-K and
the related instructions thereto) or a reportable event (as described in paragraph (a)(1)(v) of Item 304 of Regulation S-K).
Item
9A. Controls and Procedures
Evaluation
of Disclosure Controls and Procedures
As
of August 31, 2015, under the supervision and with the participation of our Chief Executive Officer (Principal Executive Officer),
and Chief Financial Officer (Principal Financial Officer), management has evaluated the effectiveness of the design and operations
of the Company’s disclosure controls and procedures. Based on that evaluation, the Chief Executive Officer and Chief Financial
Officer concluded that the Company’s disclosure controls and procedures were not effective as of August 31, 2015 as a result
of the material weakness in internal control over financial reporting discussed below.
Management’s
Report on Internal Control Over Financial Reporting
Our
management is responsible for establishing and maintaining adequate internal control over financial reporting, as such term is
defined in the Securities Exchange Act of 1934 Rule 13a-15(f). Our Chief Executive Officer and Chief Financial Officer conducted
an evaluation of the effectiveness of our internal control over financial reporting based on the framework in Internal Control
— Integrated Framework, issued by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO Framework”).
The
Company’s management assessed the effectiveness of the Company’s internal control over financial reporting as of August
31, 2015. Based on this evaluation, management concluded that our internal control over financial reporting was not effective
as of August 31, 2015. Our Chief Executive Officer and Chief Financial Officer concluded we have a material weakness due
to lack of segregation of duties and a limited corporate governance structure.
Our
size has prevented us from being able to employ sufficient resources to enable us to have an adequate level of supervision and
segregation of duties within our internal control system. Therefore while there are some compensating controls in place, it is
difficult to ensure effective segregation of accounting and financial reporting duties. Management reported a material weakness
resulting from the combination of the following significant deficiencies:· Lack of segregation of duties in certain accounting
and financial reporting processes including the approval and execution of disbursements;· The Company’s corporate
governance responsibilities are performed by the Board of Directors; we do not have independent Board of Directors, we do not
have an audit committee or compensation committee. Because our Board of Directors only meets periodically throughout the year,
several of our corporate governance functions are not performed concurrent (or timely) with the underlying transaction, evaluation,
or recordation of the transaction.
While
we strive to segregate duties as much as practicable, there is an insufficient volume of transactions at this point in time to
justify additional full time staff. We believe that this is typical in most exploration stage companies. We may not be able to
fully remediate the material weakness until we commence significant sales of our PetLife products at which time we would expect
to hire more staff. We will continue to monitor and assess the costs and benefits of additional staffing.
In
light of the above material weakness, we performed additional analyses and procedures in order to conclude that our consolidated
financial statements for the years ended August 31, 2014 and August 31, 2015 included in this Annual Report on Form 10-K were
fairly stated in accordance with US GAAP. Accordingly, management believes that despite our material weaknesses, our consolidated
financial statements for the years ended August 31, 2014 and August 31, 2015 are fairly stated, in all material respects, in accordance
with US GAAP.
This
report does not include an attestation report of the Company’s independent registered public accounting firm regarding internal
control over financial reporting. Management’s report was not subject to attestation by the Company’s independent
registered public accounting firm pursuant to temporary rules of the SEC that permit the Company to provide only management’s
report on internal control in this annual report.
There
were no significant changes in internal control over financial reporting that occurred during the last fiscal quarter covered
by this report that have materially affected, or are reasonably likely to materially affect, the Company’s internal control
over financial reporting.
PART
III
Item
10. Directors, Executive Officers and Corporate Governance Identify Directors and Executive Officers
Executive
Officers and Directors
The
following table sets forth information regarding the Company’s executive officers and directors at August 31, 2015. There
is no agreement or understanding between the Company and each current or proposed director or executive officer pursuant to which
he was selected as an officer or director.
| Name |
|
Age |
|
Position
(1) |
| |
|
|
|
|
| Arthur
G. Mikaelian |
|
53 |
|
Chief
Executive Officer and Director |
| Bruce
Niswander |
|
62 |
|
Chief
Financial Officer |
| Vivek
Ramana |
|
57 |
|
Chief
Medical Officer |
| Sebastian
Serrell-Watts |
|
50 |
|
Executive
Vice President, Secretary and Director |
| (1) | All
but Arthur G. Mikaelian have resigned as of the date hereof. |
Arthur
G. Mikaelian. For the past 20 years Dr. Mikaelian has been independently actively studying stress hormones (cortisol,
epinephrine), master gene (ATF3) and their indirect influences on cancer cell activity and tumor progression. His initial findings
quickly led him to the innovative creation of a new generation of nutraceuticals and pharmaceutical drugs, as well as expanding
his interests in cancer and oncology.
One
of Dr. Mikaelian’s greatest scientific contributions and ground-breaking inventions is his Polarization technology which
targets key elements in the main ingredients of medications, leading to the intensification of potency of liquid or solid compounds,
which substantially increases their therapeutic effectiveness. Dr. Mikaelian’s polarization technology has been awarded
U.S. Patent 8,097,284 B2 as it pertains to Polarized Scorpion Venom solution and the method for making it.
Dr.
Mikaelian’s technical education began at the 2nd Medical Institute of Moscow and continued at the Vernadsky University of
Biosphere Knowledge in Moscow, where he earned his doctorate in Biological Psychology; he then went on to complete his post-doctorate
work at Vernadsky University. He also earned an MBA from the University of Bologna, located in Italy.
Bruce
Niswander. Mr. Bruce Niswander has had over 30 years of experience in commercializing new, entrepreneurial ventures, and
was previously working as Director of Economic Development Initiatives with United Nations affiliate, South-South Corporation.
During that association he also created and managed the Global Business Incubator (GBI). Prior to Mr. Niswander’s work with
the United Nations and GBI he spent six years on the faculty of New York University teaching Technology Entrepreneurship, Entrepreneurial
Finance and Managing Intellectual Property. While at the University, Mr. Niswander also served as the director of Technology Transfer
and created and served as the on-going manager of 3 very successful business incubators in partnership with NYC Mayor Michael
Bloomberg. Prior to moving to New York City he personally created and commercialized six distinct entrepreneurial start-ups. He
also functioned as the Manager of Entrepreneurial Initiatives for Battelle Memorial Institute (the largest independent research
institute in the world).
Mr.
Niswander studied chemical engineering at the University of Cincinnati, holds a bachelor’s degree in finance (Summa Cum
Laude), a Master’s degree in Business Administration (MBA) and a juris doctor (JD) – from Ohio State University. He
has also successfully passed all of the requirements for the designation of Certified Financial Analyst (CFA).
Vivek
Ramana. Prior to joining PetLife, Dr. Ramana served as Senior Safety Medical Officer/Senior Medical Director for Ambit
Biosciences. In 2008 - 2009, Dr. Ramana served as Senior Medical Director/Therapeutic Area Lead for US Astellas. From 2006 to
2008 he was Chief Medical Officer and President for AdPharma. From 2003 to 2006, he served as Vice President, Medical Services
at Ovation Pharmaceuticals. During the early part of Dr. Ramana’s career, from 1984 to 2003, Dr. Ramana served as Medical
Director of R&D for Pharmaceutical Clinical Research, Inc., which involved joint ventures with BMS, Pfizer, Schering Plough,
and Astra Zeneca.
Dr.
Ramana received his medical degree from the University of Belgrade, Serbia and specialized in Radiation Oncology and Clinical
Pathology. Post Graduate studies in Molecular Immunology, Genetics and Molecular Biology from University of New Haven, CT; Post-Doctoral
Studies in Clinical Pharmacology, Drug Research & Development and Regulation from TUFTS University in Boston.
Sebastian
Serrell-Watts. Mr. Serrell-Watts has diverse experience in operations, healthcare, and the financial industry, with tenures
at firms such as Merrill Lynch and Kovack Securities. He is also experienced in corporate operations, management, sales and distribution.
He was educated in England and at the University of Southern California.
Family
Relationships
There
are no family relationships among the Company’s existing or incoming directors or officers.
Board
and Director Independence
The
Company utilizes the definition of “independent” set forth in the listing standards of The NASDAQ Stock Market, LLC
(“Nasdaq”). Currently, the Company believes that none of its directors would be considered independent. As discussed
below, the Company currently does not have a standing audit committee, compensation committee or nominating committee or any other
standing committees.
Corporate
Governance
The
entire board of directors serves as the audit committee. The board presently does not have an “audit committee financial
expert,” as such term is defined under the securities laws. The board does not believe it is necessary to have a financial
expert, given the early stage of the Company’s commercial operations and limited financial resources and activities. The
Company believes that none of its directors would be considered “independent,” applying the Nasdaq listing standards
for independence for members of an audit committee.
The
Company is not required to have and does not have a compensation committee. The Company does not believe it is necessary for the
board of directors to appoint a compensation committee because the volume of compensation matters that will come before the board
for consideration permits the entire board to give sufficient time and attention to such matters to be involved in all decision
making. The Company has not paid any compensation to its executive officers or directors in the last two fiscal years. The entire
board participates in consideration of executive officer and director compensation. The Company expects that the board will make
all decisions regarding executive officer compensation. The board will consider the recommendations of the Chief Executive Officer
when determining compensation for the other executive officers. The Chief Executive Officer will have no role in determining his
own compensation. The Company has not paid any fee to or otherwise engaged any compensation consultants.
The
Company also is not required to have and does not have a nominating committee. Given the limited scope of the Company’s
operations, the board believes appointing a nominating committee would be premature and of little assistance until the Company’s
business operations are at a more advanced level.
The
Company does not have a written policy or formal procedural requirements for stockholders to submit recommendations for director
nominations. However, the board will consider recommendations from stockholders. Stockholders should communicate nominee suggestions
directly to the board and accompany the recommendation with biographical details and a statement of support for the nominee. The
suggested nominee must also provide a statement of consent to being considered for nomination.
The
entire board of directors decides on nominees. The board reviews any written information provided with respect to the candidates
and interviews the candidates. Although there are no formal criteria for nominees, the board believes that persons should be actively
engaged in business endeavors, have a financial background, be familiar with acquisition strategies and money management and be
able to promote a diversity of views based on the person’s education, experience and professional employment. Based on the
information gathered, the board then makes a decision on whether to recommend the candidates as nominees for director. The committee
does not distinguish among nominees recommended by stockholders and other persons. The Company does not pay any fee to or otherwise
engage any third party or parties to identify or evaluate or assist in identifying or evaluating potential nominees. Though the
committee does not have specific guidelines on diversity, it is one of many criteria considered by the board when evaluating candidates.
Significant
Employees
We
have no significant employees other than our chief executive officer and chief financial officer.
Board
Meetings
During
the fiscal year ended August 31, 2015, we had two directors. During the year fiscal year ended August 31, 2015, the Board held
one meeting and has taken numerous actions by unanimous written consent.
Shareholder
Communications
We
do not have a formal shareholder communications process. Shareholders are welcome to communicate with the Company by forwarding
correspondence to Petlife Pharmaceuticals, Inc., 433 N Camden Drive, Beverly Hills, CA 90210, Attn.: Shareholder Relations.
Section
16(a) Beneficial Ownership Reporting Compliance
Section
16(a) of the Exchange Act, requires the Company’s officers and directors, and persons who own more than 10% of the Company’s
Common Stock, to file reports of ownership and changes in ownership of the Company’s Common Stock with the SEC. To our knowledge,
during the fiscal year ended August 31, 2015, based solely on a review of such materials as are required by the SEC, all required
reporting is current and accurate.
Code
of Business Conduct and Ethics
We
have adopted a code of business conduct and ethics that applies to all of our executive officers and employees. The Code addresses
conflicts of interest, compliance with all laws and other legal requirements, conduct of business in an honest and ethical manner,
integrity and actions in the Company’s best interest. Directors, officers and employees are required to report any conduct
that they believe in good faith to be an actual or apparent violation of the Code. The Sarbanes-Oxley Act of 2002 requires companies
to have procedures to receive, retain and treat complaints received regarding accounting, internal accounting controls or auditing
matters and to allow for the confidential and anonymous submission by employees of concerns regarding questionable accounting
or auditing matters. The Company currently has such procedures in place.
Item
11. Executive Compensation
Summary
Compensation Table
The
following table summarizes all compensation recorded by us in the period for the years ended August 31, 2015 and 2014 for our
named executive officers.
Name
and
Principal
Position | |
Year | | |
Salary | | |
Bonus | | |
Stock
Awards | | |
Option
Awards | | |
Non-Equity
Incentive Plan
Compensation | | |
Nonqualified
Deferred Compensation Earnings | | |
All
other Compensation | | |
Total | |
| | |
| | |
| | |
| | |
| | |
| | |
| | |
| | |
| | |
| |
| Arthur G. Mikaelian,
CEO | |
| 2014 | | |
| - | | |
| - | | |
| 40,000 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 40,000 | |
| | |
| 2015 | | |
| 110,400 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 110,400 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Sebastian Serrell-Watts, CFO | |
| 2014 | | |
| - | | |
| - | | |
| 32,000 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 32,000 | |
| | |
| 2015 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Bruce Niswander, CFO | |
| 2014 | | |
| - | | |
| - | | |
| 10,400 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| 10,400 | |
| | |
| 2015 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | |
| Vivek Ramana, CMO | |
| 2014 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| (2 | ) |
| | |
| 2015 | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | | |
| - | |
Executive
Employment Agreements
We
currently have no employment agreements with any officers or directors or significant employees.
Equity
Compensation Plans
We
have no current equity compensation plans.
Outstanding
Equity Awards at Fiscal Year-end
There
we no outstanding equity awards for our Executive officers for the years ended August 31, 2014 and August 31, 2015.
Stock
Option Exercised
There
were no stock options exercised on common shares during the years ended August 31, 2014 and August 31, 2015.
Expense
Reimbursement
We
will reimburse our officers and directors for reasonable expenses incurred during the course of their performance.
Retirement
Plans and Benefits.
None.
Director
Compensation
The
following table summarizes all director compensation for our Executive officers in the most recent fiscal year ended August 31,
2015 and for the previous fiscal year ended August 31, 2014. No compensation was paid in either fiscal year due to lack of operating
funds. There are no other standard compensation arrangements in place and all directors are treated equally with respect to any
compensation.
| | |
| | |
Fees
earned or | | |
Stock
| | |
Option | | |
Total | |
| Name | |
Year | | |
paid
in cash ($) | | |
awards
($) | | |
awards
($) | | |
($) | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Arthur
G. Mikaelian | |
| 2014 | | |
| — | | |
| 40,000 | | |
| — | | |
| 40,000 | |
| | |
| 2015 | | |
| 110,400 | | |
| - | | |
| - | | |
| 110,00 | |
| Sebastian Serrell-Watts | |
| 2014 | | |
| — | | |
| 32,000 | | |
| — | | |
| 32,000 | |
| Randall Lanham | |
| 2013 | | |
| — | | |
| — | | |
| — | | |
| — | |
Standard
Director Compensation Arrangement
We
do not have a standard compensation arrangement for directors.
Indemnification
and Limitation on Liability of Directors
Our
Articles of Incorporation and Bylaws provide that we must indemnify, to the fullest extent permitted by the laws of the State
of Nevada, any of our directors, officers, employees or agents made or threatened to be made a party to a proceeding, by reason
of the person serving or having served in a capacity as such, against judgments, penalties, fines, settlements and reasonable
expenses incurred by the person in connection with the proceeding if certain standards are met.
The
Nevada Revised Statutes allows indemnification of directors, officers, employees and agents of a company against liabilities incurred
in any proceeding in which an individual is made a party because he or she was a director, officer, employee or agent of the company
if such person conducted himself in good faith and reasonably believed his actions were in, or not opposed to, the best interests
of the company, and with respect to any criminal action or proceeding, had no reasonable cause to believe his conduct was unlawful.
A person must be found to be entitled to indemnification under this statutory standard by procedures designed to assure that disinterested
members of the board of directors have approved indemnification or that, absent the ability to obtain sufficient numbers of disinterested
directors, independent counsel or shareholders have approved the indemnification based on a finding that the person has met the
standard. Indemnification is limited to reasonable expenses.
At
present, there is no pending litigation or proceeding involving any of our directors, officers, employees or agents where indemnification
will be required or permitted. Insofar as indemnification for liabilities arising under the 1933 Act may be permitted to our directors,
officers and controlling persons pursuant to the foregoing provisions, or otherwise, we have been advised that in the opinion
of the SEC, such indemnification is against public policy as expressed in the 1933 Act and is, therefore, unenforceable.
Our
Articles of Incorporation limit the liability of our directors to the fullest extent permitted by law. Specifically, our directors
will not be personally liable for monetary damages for breach of fiduciary duty as directors, except for:
●
any breach of the duty of loyalty to us or our stockholders;
●
acts or omissions not in good faith or that involved intentional misconduct or a knowing violation of law;
●
dividends or other distributions of corporate assets that are in contravention of certain statutory or contractual restrictions;
●
violations of certain laws; or
●
any transaction from which the director derives an improper personal benefit.
Item
12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The
following table sets forth certain information regarding the beneficial ownership of the Company’s Common Stock as of December
24, 2015, by (i) each person known by the Company to beneficially own more than five percent of the outstanding shares of
Common Stock, (ii) each current director and named executive officer of the Company and (iii) all executive officers and directors
as a group. Except as indicated, the persons named in the table have sole voting and investment power with respect to all shares
beneficially owned. The shares of capital stock listed reflect the exchange of 1 share for every 15 shares of common stock effective
August 12, 2014, and reflect a total of 58,532,156 shares issued and outstanding as of August 31, 2015.
The
business address of each individual or entity is c/o PetLife Pharmaceuticals, Inc., 433 N. Camden Dr., Beverly Hills, CA 90210.
| Title
of Class | |
Name
and Address of Beneficial Owner | |
Amount
and Nature of Beneficial Ownership(1) | | |
Percentage
of
Common Stock | |
| Common
Stock | |
Dr. Arthur
Grant Mikaelian (2) | |
| 26,754,940 | | |
| 44.2
| % |
| Common Stock | |
Sebastian Serrell-Watts
(2) | |
| 3,000,000 | | |
| 5.0 | % |
| Common Stock | |
Bruce Niswander (2) | |
| 150,000 | | |
| 0.2 | % |
| Common Stock | |
Vivek Ramana (2) | |
| 150,000 | | |
| 0.2 | % |
| Common
Stock | |
Elite International
Partners, Inc., 8233 Roxbury, Los Angeles, CA 90069 | |
| 5,491,944 | | |
| 9.2 | % |
| Common
Stock | |
All officers and directors
(4 persons) (2) | |
| 30,054,940 | | |
| 49.7
| % |
| |
(1) |
All
shares are owned directly. |
| |
|
|
| |
(2) |
Officer
and director. |
Item
13. Certain Relationships and Related Transactions, and Director Independence
Dr.
Arthur Mikaelian, our President and Chief Executive Officer, is also the President and CEO of Medolife Corp., a company with the
rights to the non-veterinary uses of Polarized Blue Scorpion venom in their product Escozine. Dr. Mikaelian is also President
and Chief Executive Officer of our wholly-owned operating subsidiary Petlife Corporation.
The
scorpion reservation that we use is owned by Medolife. We have entered into a definitive agreement with Medolife for our utilization
of that reservation in the manufacture and preparation of our products.
Most
of our current employees, including without limitation all employees who are engaged in research and development, are also employed
or otherwise work for Medolife Corporation.
Item
14. Principal Accountant Fees and Services
On
December 1, 2015 the Company engaged LBB & Associates Ltd., LLP (“LBB”) as its independent registered public accounting
firm for the Company’s fiscal year ended August 31, 2014 and 2013. LBB remains the Company’s accounting firm. LBB
has been paid a retainer of $10,000 but has not yet billed the Company for audit fees.
RBSM
LLP (“RBSM”) served as our independent registered public accounting firm from August 16, 2011 through and for the
period ended August 31, 2013. RBSM initially was engaged for the fiscal year ended August 31, 2014 but that relationship has terminated.
The
following table sets forth fees billed to us by RBSM during the fiscal years ended August 31, 2014 and 2013 for: (i) services
rendered for the audit of our annual consolidated financial statements and the review of our quarterly consolidated financial
statements, (ii) services by our auditor that are reasonably related to the performance of the audit or review of our consolidated
financial statements and that are not reported as Audit Fees, (iii) services rendered in connection with tax compliance, tax advice
and tax planning, and (iv) all other fees for services rendered.
| | |
Fiscal
Years Ended | |
| | |
August
31, 2015 | | |
August
31, 2014 | |
| Audit Fees
(1) | |
$ | 22,000 | | |
$ | 21,500 | |
| Audit Related Fees
(2) | |
| - | | |
| - | |
| Tax Fess (3) | |
| - | | |
| - | |
| All
Other Fees (4) | |
| - | | |
| - | |
| | |
$ | 22,000 | | |
$ | 21,500 | |
(1)
Audit Fees
Audit
fees include fees incurred for professional services rendered in connection with the audit of the Company’s annual consolidated
financial statements for the two years ended August 31, 2015 and for the reviews of the quarterly interim consolidated financial
statements included in the Company’s Forms 10-Q for the fiscal years ended August 31, 2015.
(2)
Audit-Related Fees
We
have not yet been billed by LBB for audit related fees for August 31, 2015.
(3)
Tax Fees
No
fees of this sort were billed for the years ended August 31, 2014 and 2015.
(4)
All Other Fees
No
fees of this sort were billed for the years ended August 31, 2014 and 2015.
Policy
on Pre-Approval of Audit and Permissible Non-audit Services of Independent Auditors
Consistent
with the SEC policies regarding auditor independence, our Board of Directors has responsibility for appointing, setting compensation
and overseeing the work of the independent auditor. In recognition of this responsibility, our Board of Directors has established
a policy to pre-approve all audit and permissible non-audit services provided by the independent auditor.
Prior
to engagement of the independent auditor for the next year’s audit, management will submit an aggregate of services expected
to be rendered during that year for each of the following four categories of services to the Board of Directors for approval.
PART
IV
Item
15. Exhibits and Financial Statement Schedules
See
the Exhibit Index and the Index to Financial Statements immediately following the signature page of this report.
SIGNATURES
Pursuant
to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this Annual
Report on Form 10-K for the fiscal year ended August 31, 2015 to be signed on its behalf by the undersigned, thereunto duly authorized.
| |
Petlife
Pharmaceuticals, Inc. |
| January
4, 2016 |
|
|
| |
By: |
/s/
Arthur G. Mikaelian |
| |
|
Arthur
G. Mikaelian |
| |
|
Chief
Executive Officer, Director |
| |
|
|
| |
By: |
/s/
Arthur G. Mikaelian |
| |
|
Arthur
G. Mikaelian |
| |
|
Chief
Financial & Principal Accounting Officer, |
EXHIBIT
INDEX
Exhibits
| Exhibit
No. |
|
Description |
| |
|
|
| 2.1 |
|
Agreement
and Plan of Merger dated as of June 26, 2014 between Eco Ventures Group, Inc. and PetLife Pharmaceuticals, Inc. (i) |
| |
|
|
| 3.2 |
|
Bylaws
(ii) |
| |
|
|
| 10.1 |
|
Reorganization
Agreement dated as of April 28, 2014 by and between Eco Ventures Group, Inc. and PetLife Corporation (i) |
| |
|
|
| 10.2 |
|
Patent
License Agreement dated as of August 1, 2014 between Arthur Grant Mikaelian and Petlife Pharmaceuticals, Inc. (i) |
| |
|
|
| 10.3 |
|
Manufacturing
Agreement dated as of May 8, 2014 between Petlife Corporation and Samson Pharmaceuticals, Inc. (i) |
| |
|
|
| 31.1 |
|
Certification
Pursuant to Rule 13A-14 or 15D-14 of the Securities Exchange Act of 1934, as adopted pursuant to Section 302 of the Sarbanes-Oxley
Act of 2002 signed by the Principal Executive Officer |
| |
|
|
| 31.2 |
|
Certification
Pursuant to Rule 13A-14 or 15D-14 of the Securities Exchange Act of 1934, as adopted pursuant to Section 302 of the Sarbanes-Oxley
Act of 2002 signed by the Principal Financial Officer |
| |
|
|
| 32.1 |
|
Certification
Required by 18 U.S.C. Section 1350, as Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 signed by Principal
Executive Officer and Chief Financial Officer |
| |
|
|
| 101 |
|
Interactive
data files |
(i)
Incorporated herein by reference to the Company’s Periodic Report on Form 8-K filed on April 8, 2014.
(ii)
Incorporated herein by reference to the Company’s Annual Report on Form 10-K filed on December 16, 2014.
PETLIFE
PHARMACEUTICALS, INC.
INDEX
TO CONSOLIDATED FINANCIAL STATEMENTS
PETLIFE
PHARMACEUTICALS, INC.
Consolidated
Balance Sheets
| | |
August 31, | |
| | |
2015 | | |
2014 | |
| ASSETS | |
| | | |
| | |
| Current assets | |
| | | |
| | |
| Cash | |
$ | - | | |
$ | 6,852 | |
| Due from affiliate | |
| 4,800 | | |
| 10,582 | |
| Total current assets | |
| 4,800 | | |
| 17,434 | |
| | |
| | | |
| | |
| Website development | |
| 600 | | |
| - | |
| | |
| | | |
| | |
| Total asssets | |
$ | 5,400 | | |
$ | 17,434 | |
| | |
| | | |
| | |
| LIABILITIES AND STOCKHOLDERS’ EQUITY (DEFICIT) | |
| | | |
| | |
| Current liabilities | |
| | | |
| | |
| Accounts payable and accrued expense | |
$ | 360,527 | | |
$ | - | |
| Due to shareholder | |
| 10,000 | | |
| - | |
| Total current liabilities | |
| 370,527 | | |
| - | |
| | |
| | | |
| | |
| Total liabilities | |
| 370,527 | | |
| - | |
| | |
| | | |
| | |
| Stockholders’ equity (deficit) | |
| | | |
| | |
| Preferred stock, $0.001 par value, 50,000,000 authorized, none issued
and outstanding | |
| - | | |
| - | |
| Common stock, $0.001 par value, 750,000,000 authorized, 58,532,156 and 54,634,056 shares
issued and outstanding, respectively | |
| 58,532 | | |
| 54,634 | |
| Additional paid-in capital | |
| 3,024,029 | | |
| 1,474,889 | |
| Accumulated deficit | |
| (3,447,688 | ) | |
| (1,512,089 | ) |
| Total stockholders’ equity (deficit) | |
| (365,127 | ) | |
| 17,434 | |
| | |
| | | |
| | |
| Total liabilities and stockholders ‘ equity (deficit) | |
$ | 5,400 | | |
$ | 17,434 | |
The
accompanying notes are an integral part of these financial statements.
PETLIFE
PHARMACEUTICALS, INC.
Consolidated
Statement of Operations
| | |
Year ended | | |
Year ended | |
| | |
August
31, 2015 | | |
August
31, 2014 | |
| | |
| | |
| |
| Revenue | |
$ | - | | |
$ | - | |
| | |
| | | |
| | |
| Operating Expenses | |
| | | |
| | |
| Research and development-related party | |
| - | | |
| 73,198 | |
| Manufacturing and production | |
| 220,733 | | |
| 60,079 | |
| General and administrative | |
| 1,714,866 | | |
| 1,378,774 | |
| Total operating expenses | |
| 1,935,599 | | |
| 1,512,051 | |
| | |
| | | |
| | |
| Operating loss | |
| (1,935,599 | ) | |
| (1,512,051 | ) |
| | |
| | | |
| | |
| Net loss | |
$ | (1,935,599 | ) | |
$ | (1,512,051 | ) |
| | |
| | | |
| | |
| Net loss per share - basic and diluted | |
$ | (0.03 | ) | |
$ | (0.04 | ) |
| | |
| | | |
| | |
| Weighted average shares outstanding | |
| 57,297,917 | | |
| 40,113,465 | |
The
accompanying notes are an integral part of these financial statements.
PETLIFE
PHARMACEUTICALS, INC.
Statements
of Stockholders’ Equity (Deficit)
Year
Ended August 31, 2015 and 2014
| | |
| | |
| | |
Additional | | |
| | |
| |
| | |
Common Stock | | |
Paid-in | | |
Accumulated | | |
| |
| | |
Shares | | |
Amount | | |
Capital | | |
Deficit | | |
Total | |
| Balance: August 31, 2013 | |
| 37,990,000 | | |
| 37,990 | | |
| (37,952 | ) | |
| (38 | ) | |
| - | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Common stock issued in connection with reverse merger | |
| 10,988,116 | | |
| 10,988 | | |
| 89,012 | | |
| - | | |
| 100,000 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Issuance of common stock for cash | |
| 676,000 | | |
| 676 | | |
| 183,824 | | |
| - | | |
| 184,500 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Stock-based compensation | |
| 4,979,940 | | |
| 4,980 | | |
| 1,240,005 | | |
| - | | |
| 1,244,985 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Net loss | |
| - | | |
| - | | |
| - | | |
| (1,512,051 | ) | |
| (1,512,051 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Balance August 31, 2014 | |
| 54,634,056 | | |
| 54,634 | | |
| 1,474,889 | | |
| (1,512,089 | ) | |
| 17,434 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Stock-based compensation | |
| 3,898,100 | | |
| 3,898 | | |
| 1,549,140 | | |
| - | | |
| 1,553,038 | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Net loss | |
| - | | |
| - | | |
| - | | |
| (1,935,599 | ) | |
| (1,935,599 | ) |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Balance August 31, 2015 | |
| 58,532,156 | | |
$ | 58,532 | | |
$ | 3,024,029 | | |
$ | (3,447,688 | ) | |
$ | (365,127 | ) |
The
accompanying notes are an integral part of these financial statements.
PETLIFE
PHARMACEUTICALS, INC.
Consolidated
Statements of Cash Flow
| | |
For the year | | |
For the year | |
| | |
ended | | |
ended | |
| | |
August 31, 2015 | | |
August 31, 2014 | |
| Net Cash from (used in) operating activities | |
| | | |
| | |
| Net loss | |
$ | (1,935,599 | ) | |
$ | (1,512,051 | ) |
| Stock-based compensation | |
| 1,553,038 | | |
| 1,244,985 | |
| Accounts payable and accrued expenses | |
| 360,527 | | |
| - | |
| Net cash used in operating activities | |
| (22,034 | ) | |
| (267,066 | ) |
| | |
| | | |
| | |
| Net cash from (used in) investing activities | |
| | | |
| | |
| Capital expenditure | |
| (600 | ) | |
| - | |
| Reverse acquisition | |
| - | | |
| 100,000 | |
| Net cash provided by (used in) investing activities | |
| (600 | ) | |
| 100,000 | |
| | |
| | | |
| | |
| Net cash provided by financing activities | |
| | | |
| | |
| Proveeds from the sale of common stock | |
| - | | |
| 184,500 | |
| Due from affiliate, net | |
| 5,782 | | |
| (10,582 | ) |
| Proceeds from sharholder advance | |
| 10,000 | | |
| - | |
| Net cash provided by financing activities | |
| 15,782 | | |
| 173,918 | |
| | |
| | | |
| | |
| Increase (decrease) in cash | |
| (6,852 | ) | |
| 6,852 | |
| | |
| | | |
| | |
| Cash, beginning of period | |
| 6,852 | | |
| - | |
| | |
| | | |
| | |
| Cash, end of period | |
$ | - | | |
$ | 6,852 | |
The
accompanying notes are an integral part of these financial statements.
PETLIFE
PHARMACEUTICALS, INC.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
AUGUST
31, 2015 and from
Inception
(December 12, 2012) to August 31, 2015
NOTE
1 – SIGNIFICANT ACCOUNTING POLICIES
Nature
of Operations
PetLife
Pharmaceuticals, Inc. (“Company”) is a registered U.S. Veterinary Pharmaceutical company whose mission is to bring
its scientifically proven, potentiated bioactive medication and nutraceuticals — “Escozine for Pets™”
— to the world of veterinary oncology. The Company specializes in the research, development, sales and support of advanced
drugs and nutraceuticals for pet cancer and autoimmune related diseases such as arthritis.
A
summary of the significant accounting policies applied in the presentation of the accompanying consolidated financial statements
follows:
Basis
and business presentation
The
Company was incorporated on April 5, 2002 under the laws of the State of Nevada as “Aztek Ventures Inc.” Effective
November 13, 2007, we filed a Certificate of Amendment to our Articles of Incorporation to change our name from “Aztek Ventures
Inc.” to “Genesis Uranium Corp.” Effective April 21, 2008, we amended our Articles of Incorporation to change
our name from “Genesis Uranium Corp.” to “Vault Technology Inc.” to reflect the change in our business
focus beyond solely that of uranium exploration. Effective July 10, 2009, we filed a Certificate of Amendment to our Articles
of Incorporation to change our name from “Vault Technology, Inc.” to “Modern Renewable Technologies, Inc.”
(“Modern”). On May 27, 2011, Modern, merged with Eco Ventures Group, Inc., and the name of the Company was changed
to Eco Ventures Group, Inc. On July 15, 2013, the Company entered into an Agreement and Plan of Merger with Clear TV Ventures,
Inc. Under the terms of the merger, Clear TV became the surviving corporation. On June 26, 2014, Eco Ventures Group, Inc. entered
into an Agreement and Plan of Merger with its subsidiary, PetLife Pharmaceuticals, Inc., a Nevada Corporation, with PetLife Pharmaceuticals,
Inc. being the surviving entity. As part of that merger, the name of the Company was changed to PetLife Pharmaceuticals, Inc.
and each 15 shares of our common stock were exchanged for one share in the surviving company. Effective August 12, 2014 we completed
the closing of the Share Exchange Agreement and the acquisition of Petlife and changed our name to Petlife Pharmaceuticals, Inc.
All references herein to the number of shares outstanding and per-share amounts have been retroactively restated to reflect the
exchange ratio in the merger with PetLife Pharmaceuticals, Inc.
All
references that refer to (the “Company” or “PetLife Pharmaceuticals, Inc.” or "Petlife" or “we”
or “us” or “our”) are PetLife Pharmaceuticals, Inc., the Registrant and its wholly and or majority owned
subsidiaries. We are in the development stage, as defined by Accounting Standards Codification subtopic 915-10, Development Stage
Entities ("ASC 915-10") and have developed and is launching a new generation of potentiated veterinary cancer medications
and nutraceuticals, based on the same patented formula “Escozine” and production processes that have been scientifically
proven as an effective treatment for cancer in humans for years. We have not generated any revenues to date, have incurred expenses
and has sustained losses since December 12, 2012 (date of inception). Consequently, our operations are subject to all the risks
inherent in the establishment of a new business enterprise. For the period from December 12, 2012 (date of inception) through
August 31, 2015, we have accumulated a deficit through its development stage of approximately $3,447,688.
PETLIFE
PHARMACEUTICALS, INC.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
AUGUST
31, 2015 and from
Inception
(December 12, 2012) to August 31, 2015
The
consolidated financial statements include the accounts of the Company and its majority owned subsidiaries. All significant intercompany
balances and transactions have been eliminated in consolidation.
Merger
and Corporate Restructure
On
or about April 18, 2014, the Company entered into a Share Exchange Agreement (the “Share Exchange”) with Petlife
Corporation, a Delaware corporation (“Petlife”) and the shareholders of Petlife Corporation (the “Shareholders”)
for the exchange of all of the issued and outstanding shares of Petlife. Effective at the Closing on August 12, 2014, these
shares of Petlife were exchanged for approximately 47,000,000 fully paid non-assessable shares of the Company reflecting approximately
80% of the issued and outstanding shares of the Company and acquisition of all assets and liabilities of the Company and was valued
equated to the net assets of the Company as of the date of the acquisition of $100,000. No liabilities were assumed. In
connection with the share exchange, Petlife Corporation became our wholly owned subsidiary.
Accordingly,
the historical financial statements are those of Petlife, the accounting acquirer, immediately following the consummation of the
reverse acquisition. The Company did not recognize goodwill or any intangible assets in connection with this transaction.
Estimates
The
preparation of the financial statements in conformity with generally accepted accounting principles requires management to make
estimates and assumptions that affect certain reported amounts and disclosures. Accordingly, actual results could differ from
those estimates.
Revenue
Recognition
The
Company will recognize revenue in accordance with Accounting Standards Codification subtopic 605-10, Revenue Recognition (“ASC
605-10”) which requires that four basic criteria must be met before revenue can be recognized: (1) persuasive evidence of
an arrangement exists; (2) delivery has occurred; (3) the selling price is fixed and determinable; and (4) collectability is reasonably
assured. Determination of criteria (3) and (4) are based on management’s judgments regarding the fixed nature of the selling
prices of the products delivered and the collectability of those amounts. Provisions for discounts and rebates to customers, estimated
returns and allowances, and other adjustments will be provided for in the same period the related sales will be recorded.
ASC
605-10 incorporates Accounting Standards Codification subtopic 605-25, Multiple-Element Arraignments (“ASC 605-25”).
ASC 605-25 addresses accounting for arrangements that may involve the delivery or performance of multiple products, services and/or
rights to use assets. There was no effect on implementing ASC 605-25 on the Company’s financial position and results of
operations, since the Company has not started generating revenue.
PETLIFE
PHARMACEUTICALS, INC.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
AUGUST
31, 2015 and from
Inception
(December 12, 2012) to August 31, 2015
Cash
The
Company considers cash to consist of cash on hand and temporary investments having an original maturity of 90 days or less that
are readily convertible into cash.
Property
and Equipment
Property
and equipment are stated at cost. When retired or otherwise disposed, the related carrying value and accumulated depreciation
are removed from the respective accounts and the net difference less any amount realized from disposition, is reflected in earnings.
Long-Lived
Assets
The
Company follows FASB ASC 360-10-15-3, “Impairment or Disposal of Long-lived Assets,” which established a “primary
asset” approach to determine the cash flow estimation period for a group of assets and liabilities that represents the unit
of accounting for a long-lived asset to be held and used. Long-lived assets to be held and used are reviewed for impairment whenever
events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. The carrying amount of
a long-lived asset is not recoverable if it exceeds the sum of the undiscounted cash flows expected to result from the use and
eventual disposition of the asset. Long-lived assets to be disposed of are reported at the lower of carrying amount or fair value
less cost to sell.
Income
Taxes
The
Company has adopted Accounting Standards Codification subtopic 740-10, Income Taxes (“ASC 740-10”) which requires
the recognition of deferred tax liabilities and assets for the expected future tax consequences of events that have been included
in the financial statement or tax returns. Under this method, deferred tax liabilities and assets are determined based on the
difference between financial statements and tax basis of assets and liabilities using enacted tax rates in effect for the year
in which the differences are expected to reverse. Temporary differences between taxable income reported for financial reporting
purposes and income tax purposes consist primarily of timing differences such as deferred officers’ compensation and stock
based compensation accounting.
Net
Loss per Common Share, basic and diluted
The
Company has adopted Accounting Standards Codification subtopic 260-10, Earnings Per Share (“ASC 260-10”) specifying
the computation, presentation and disclosure requirements of earnings per share information. Basic loss per share has been calculated
based upon the weighted average number of common shares outstanding.
PETLIFE
PHARMACEUTICALS, INC.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
AUGUST
31, 2015 and from
Inception
(December 12, 2012) to August 31, 2015
Stock
based compensation
The
Company follows Accounting Standards Codification subtopic 718-10, Compensation (“ASC 718-10”) which requires that
all share-based payments to both employees and non-employees be recognized in the income statement based on their fair values.
As
of August 31, 2015, the Company did not have any issued or outstanding stock options.
Concentrations
of Credit Risk
Financial
instruments and related items, which potentially subject the Company to concentrations of credit risk, consist primarily of cash,
cash equivalents and trade receivables. The Company places its cash and temporary cash investments with high credit quality institutions.
At times, such investments may be in excess of the FDIC insurance limit.
Research
and Development
The
Company accounts for research and development costs in accordance with Accounting Standards Codification subtopic 730-10, Research
and Development (“ASC 730-10”). Under ASC 730-10, all research and development costs must be charged to expense as
incurred. Accordingly, internal research and development costs are expensed as incurred. Third-party research and development
costs are expensed when the contracted work has been performed or as milestone results have been achieved as defined under the
applicable agreement. Company-sponsored research and development costs related to both present and future products are expensed
in the period incurred. The Company incurred costs of $0 and $73,198 for research and development expenses for the years
ended August 31, 2015 and 2014.
Reliance
on Key Personnel and Consultants
The
Company has eleven full-time employees, three of which are executive officers. Additionally, the Company has consultants performing
various specialized services. The Company is heavily dependent on the continued active participation of these current executive
officers, employees and key consultants. The loss of any of the senior management or key consultants could significantly and negatively
impact the business until adequate replacements can be identified and put in place.
PETLIFE
PHARMACEUTICALS, INC.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
AUGUST
31, 2015 and from
Inception
(December 12, 2012) to August 31, 2015
Fair
Value
Accounting
Standards Codification subtopic 825-10, Financial Instruments (“ASC 825-10”) requires disclosure of the fair value
of certain financial instruments. The carrying amount reported in the unaudited condensed consolidated balance sheet for accounts
payable and accrued expenses, advances and notes payable approximates fair value because of the immediate or short-term maturity
of these financial instruments.
Recent
Accounting Pronouncements
In
May 2014, the FASB issued ASU 2014-09, “Revenue from Contracts with Customers (Topic 606).” This guidance outlines
a single, comprehensive model for accounting for revenue from contracts with customers. We will adopt the standard in 2018. While
we are currently assessing the impact of the new standard, we do not expect this new guidance to have a material impact on our
Consolidated Financial Statements.
No
other new accounting pronouncement issued or effective during the fiscal year had or is expected to have a material impact on
the Consolidated Financial Statements.
NOTE
2 – GOING CONCERN
The
accompanying consolidated financial statements have been prepared on a going concern basis, which contemplates the realization
of assets and the satisfaction of liabilities in the normal course of business. As shown in the accompanying consolidated financial
statements as of August 31, 2015, the Company has an accumulated deficit of $3,447,688. As of August 31, 2015, the Company’s
absence of revenues, recurring losses and its need for additional financing to operate and fund its projected loss in 2016 raise
substantial doubt about its ability to continue as a going concern.
The
Company’s existence is currently dependent upon management’s ability to develop profitable operations and or upon
obtaining additional financing to carry out its planned business. Management is devoting substantially all of its efforts to the
commercialization of its planned products, as well as raising additional debt or equity financing in order to accelerate the development
and commercialization of additional products. There can be no assurance that the Company’s commercialization or financing
efforts will result in profitable operations or the resolution of the Company’s liquidity problems.
There
can be no assurance that any additional financings will be available to the Company on satisfactory terms and conditions, if at
all. In the event the Company is unable to continue as a going concern, it may elect or required to seek protection from its creditors
by filing a voluntary petition in bankruptcy or may be subject to an involuntary petition in bankruptcy. To date, management has
not considered this alternative, nor does management view it as a likely occurrence.
The
accompanying consolidated statements do not include any adjustments relating to the recoverability of assets and classification
of assets and liabilities that might be necessary should the Company be unable to continue as a going concern.
PETLIFE
PHARMACEUTICALS, INC.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
AUGUST
31, 2015 and from
Inception
(December 12, 2012) to August 31, 2015
NOTE
3 – STOCKHOLDERS’ EQUITY (DEFICIT)
Common
Stock
During
the year ended August 31, 2014, the Company issued 4,979,940 shares of its $.001 par value common stock for
services valued at $1,244,985.
In
August, 2014, the Company sold 676,000 shares of its $.001 par value common stock for $184,500.
In
September 2014, the Company issued 10,000 shares of its $.001 par value common stock for cash of $10,000. As of May
31, 2015, the common stock has not been issued and is included in accrued expenses.
During
the year ended August 31, 2015, the Company issued 3,898,100 shares of its $.001 par value common stock for services valued at
$1,553,038.
NOTE
4 – RELATED PARTY TRANSACTIONS
On
December 30, 2013, the Company entered into an agreement with Medolife Corporation
("Medolife), an affiliated company, for the performance of certain ongoing
services and maintenance related to a scorpion reservation , together with the providing of other services such
as product operations, research and development to support the Company's product marketing. During the year ended August
31, 2014 Medolife invoiced the Company for services and maintenance of $165,800. During
year ended August 31, 2014, Medolife received proceeds from the sale of the Company’s common stock totaling $184,500. The
Company was owed $4,800 and $10,582 from
Medolife as of August
31, 2015 and 2014.
On
August 1, 2014, the Company entered into a patent license agreement with a shareholder for a polarized scorpion venom solution
and a method for making polarized scorpion venom solution for veterinary use. The licensor has provided Medolife, an affiliated
company by common shareholders, with an exclusive right to the polarized scorpion venom patent with the exception of the rights
for veterinary use. The Company will manufacture, use, and sell polarized scorpion venom containing such patented improvements
for veterinary use.
NOTE
5 – INCOME TAXES
As
of August 31, 2015 the Company has accumulated losses of approximately $3,447,000. A deferred tax asset has been provided
and has a 100% valuation
allowance.
NOTE
6 – SHARE EXCHANGE AGREEMENT
On April
18, 2014, Petlife Corporation entered into a Share Exchange agreement with PetLife Pharmaceuticals, Inc. (formerly Clear
TV Ventures, Inc.), a publicly traded company. The shares of Petlife Corporation were exchanged for 47,000,000, or 80%
of the issued and outstanding shares of PetLife Pharmaceuticals, Inc.
The
closing of the Share Exchange Agreement was conditioned upon certain, limited customary representations and warranties as well
as conditions to close such as the total issued and outstanding shares of the Company being limited to 58,000,000 issued and outstanding
post-closing.
The
Share Exchange Agreement closed on August 11, 2014 with the issuance of 47,000,000 shares to shareholders or designees of Petlife
Corporation. The shares of common stock of PetLife Pharmaceuticals, Inc. issued in the exchange to the Petlife Corporation shareholders
were not registered under the Securities Act of 1933, as amended (the “Securities Act”), or the securities laws of
any state, and were in each case offered, sold and issued in reliance upon the exemption from registration provided by Section
4(2) of the Securities Act, as a transaction by an issuer not involving a public offering, and Rule 506 of Regulation D promulgated
thereunder. The Company relied on such exemptions based in part on written representations made by the Company shareholders, including
representations with respect to each member’s status as an accredited investor and investment intent with respect to the
acquired securities. The shares of common stock issued in the Exchange to the Company shareholders may not be offered or sold
absent registration or an applicable exemption from the registration requirements of the Securities Act, and each of the certificates
or instruments evidencing such shares bears a legend to that effect.
PETLIFE
PHARMACEUTICALS, INC.
NOTES
TO THE CONSOLIDATED FINANCIAL STATEMENTS
AUGUST
31, 2015 and from
Inception
(December 12, 2012) to August 31, 2015
The
transaction was recorded as a reverse acquisition whereby Petlife Corporation was considered to be the accounting acquirer as
its shareholders retained control of Petlife Pharmaceuticals, Inc., after the exchange, although Petlife Pharmaceuticals, Inc.,
is the legal parent company. The share exchange was treated as a recapitalization of Petlife Corporation (and its historical financial
statements) is the continuing entity for financial reporting purposes.
NOTE
7 – SUBSEQUENT EVENTS
Pursuant
to agreement, effective November 13, 2015 Bruce Niswander and Sebastian Serrell-Watts resigned as officers and directors of the
Company.
On
November 21, 2015 the Company entered into a Reorganization and Stock Purchase Agreement with Alexian Scientific, Inc. Closing
of the agreement is conditioned on the Company obtaining funding of a minimum of $10,000,000. At closing, the Company will issue
38,777,630 shares of common stock to designees of Alexian in consideration for 100% of the equity of Alexian, the Company will
appoint designees of Alexian as its officers and directors, and all of the current officers and directors of the Company will
resign. As part of the agreement with Alexian, shareholders of the Company cancelled certain shares of common stock of the Company
such that subsequent to closing there will be a total of 80,786,730 shares of common stock outstanding.
Effective
November 20, 2015 in connection with the proposed Alexian transaction, principals of the Company cancelled and returned to treasury
a total of 21,583,000 shares of common stock held by them.
Also
effective November 20, 2015, the Company issued a total of 626,000 shares of common stock to investors and issued 530,200 shares
of common stock to officers and employees of the Company in lieu of salary.
v3.3.1.900
Document and Entity Information - USD ($)
|
12 Months Ended |
|
|
Aug. 31, 2015 |
Dec. 24, 2015 |
Nov. 28, 2014 |
| Document And Entity Information |
|
|
|
| Entity Registrant Name |
PetLife Pharmaceuticals, Inc.
|
|
|
| Entity Central Index Key |
0001354591
|
|
|
| Document Type |
10-K
|
|
|
| Document Period End Date |
Aug. 31, 2015
|
|
|
| Amendment Flag |
false
|
|
|
| Current Fiscal Year End Date |
--08-31
|
|
|
| Entity a Well-known Seasoned Issuer |
No
|
|
|
| Entity a Voluntary Filer |
No
|
|
|
| Entity's Reporting Status Current |
Yes
|
|
|
| Entity Filer Category |
Smaller Reporting Company
|
|
|
| Entity Public Float |
|
|
$ 15,338,480
|
| Entity Common Stock, Shares Outstanding |
|
38,115,356
|
|
| Trading Symbol |
PTLF
|
|
|
| Document Fiscal Period Focus |
FY
|
|
|
| Document Fiscal Year Focus |
2015
|
|
|
| X |
- DefinitionIf the value is true, then the document is an amendment to previously-filed/accepted document.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionEnd date of current fiscal year in the format --MM-DD.
| Name: |
dei_CurrentFiscalYearEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:gMonthDayItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThis is focus fiscal period of the document report. For a first quarter 2006 quarterly report, which may also provide financial information from prior periods, the first fiscal quarter should be given as the fiscal period focus. Values: FY, Q1, Q2, Q3, Q4, H1, H2, M9, T1, T2, T3, M8, CY.
| Name: |
dei_DocumentFiscalPeriodFocus |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fiscalPeriodItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThis is focus fiscal year of the document report in CCYY format. For a 2006 annual report, which may also provide financial information from prior periods, fiscal 2006 should be given as the fiscal year focus. Example: 2006.
| Name: |
dei_DocumentFiscalYearFocus |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:gYearItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe end date of the period reflected on the cover page if a periodic report. For all other reports and registration statements containing historical data, it is the date up through which that historical data is presented. If there is no historical data in the report, use the filing date. The format of the date is CCYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word "Other".
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation 12B
-Number 240
-Section 12b
-Subsection 1
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate number of shares or other units outstanding of each of registrant's classes of capital or common stock or other ownership interests, if and as stated on cover of related periodic report. Where multiple classes or units exist define each class/interest by adding class of stock items such as Common Class A [Member], Common Class B [Member] or Partnership Interest [Member] onto the Instrument [Domain] of the Entity Listings, Instrument.
| Name: |
dei_EntityCommonStockSharesOutstanding |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionIndicate "Yes" or "No" whether registrants (1) have filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that registrants were required to file such reports), and (2) have been subject to such filing requirements for the past 90 days. This information should be based on the registrant's current or most recent filing containing the related disclosure.
| Name: |
dei_EntityCurrentReportingStatus |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:yesNoItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate whether the registrant is one of the following: (1) Large Accelerated Filer, (2) Accelerated Filer, (3) Non-accelerated Filer, (4) Smaller Reporting Company (Non-accelerated) or (5) Smaller Reporting Accelerated Filer. Definitions of these categories are stated in Rule 12b-2 of the Exchange Act. This information should be based on the registrant's current or most recent filing containing the related disclosure.
| Name: |
dei_EntityFilerCategory |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:filerCategoryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionState aggregate market value of voting and non-voting common equity held by non-affiliates computed by reference to price at which the common equity was last sold, or average bid and asked price of such common equity, as of the last business day of registrant's most recently completed second fiscal quarter. The public float should be reported on the cover page of the registrants form 10K.
| Name: |
dei_EntityPublicFloat |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation 12B
-Number 240
-Section 12b
-Subsection 1
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate "Yes" or "No" if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act.
| Name: |
dei_EntityVoluntaryFilers |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:yesNoItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate "Yes" or "No" if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Is used on Form Type: 10-K, 10-Q, 8-K, 20-F, 6-K, 10-K/A, 10-Q/A, 20-F/A, 6-K/A, N-CSR, N-Q, N-1A.
| Name: |
dei_EntityWellKnownSeasonedIssuer |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:yesNoItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.3.1.900
Consolidated Balance Sheets - USD ($)
|
Aug. 31, 2015 |
Aug. 31, 2014 |
| Current assets |
|
|
| Cash |
|
$ 6,852
|
| Due from affiliate |
$ 4,800
|
10,582
|
| Total current assets |
4,800
|
$ 17,434
|
| Website development |
600
|
|
| Total asssets |
5,400
|
$ 17,434
|
| Current liabilities |
|
|
| Accounts payable and accrued expense |
360,527
|
|
| Due to shareholder |
10,000
|
|
| Total current liabilities |
370,527
|
|
| Total liabilities |
$ 370,527
|
|
| Stockholders' equity (deficit) |
|
|
| Preferred stock, $0.001 par value, 50,000,000 authorized, none issued and outstanding |
|
|
| Common stock, $0.001 par value, 750,000,000 authorized, 58,532,156 and 54,634,056 shares issued and outstanding, respectively |
$ 58,532
|
$ 54,634
|
| Additional paid-in capital |
3,024,029
|
1,474,889
|
| Accumulated deficit |
(3,447,688)
|
(1,512,089)
|
| Total stockholders' equity (deficit) |
(365,127)
|
17,434
|
| Total liabilities and stockholders' equity (deficit) |
$ 5,400
|
$ 17,434
|
| X |
- DefinitionSum of the carrying values as of the balance sheet date of obligations incurred through that date and due within one year (or the operating cycle, if longer), including liabilities incurred (and for which invoices have typically been received) and payable to vendors for goods and services received, taxes, interest, rent and utilities, accrued salaries and bonuses, payroll taxes and fringe benefits. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.19,20)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
| Name: |
us-gaap_AccountsPayableAndAccruedLiabilitiesCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionExcess of issue price over par or stated value of the entity's capital stock and amounts received from other transactions involving the entity's stock or stockholders. Includes adjustments to additional paid in capital. Some examples of such adjustments include recording the issuance of debt with a beneficial conversion feature and certain tax consequences of equity instruments awarded to employees. Use this element for the aggregate amount of additional paid-in capital associated with common and preferred stock. For additional paid-in capital associated with only common stock, use the element additional paid in capital, common stock. For additional paid-in capital associated with only preferred stock, use the element additional paid in capital, preferred stock. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.30(a)(1))
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 31
-Article 5
| Name: |
us-gaap_AdditionalPaidInCapital |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionSum of the carrying amounts as of the balance sheet date of all assets that are recognized. Assets are probable future economic benefits obtained or controlled by an entity as a result of past transactions or events. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.18)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 03
-Paragraph 12
-Article 7
| Name: |
us-gaap_Assets |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionSum of the carrying amounts as of the balance sheet date of all assets that are expected to be realized in cash, sold, or consumed within one year (or the normal operating cycle, if longer). Assets are probable future economic benefits obtained or controlled by an entity as a result of past transactions or events. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.9)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section 45
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=28358313&loc=d3e6801-107765
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section 45
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=28358313&loc=d3e6676-107765
| Name: |
us-gaap_AssetsCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_AssetsCurrentAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of currency on hand as well as demand deposits with banks or financial institutions. Includes other kinds of accounts that have the general characteristics of demand deposits. Also includes short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Excludes cash and cash equivalents within disposal group and discontinued operation. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Cash
-URI http://asc.fasb.org/extlink&oid=6506951
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section 45
-Paragraph 1
-Subparagraph (a)
-URI http://asc.fasb.org/extlink&oid=28358313&loc=d3e6676-107765
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 4
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3044-108585
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Cash Equivalents
-URI http://asc.fasb.org/extlink&oid=6507016
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.1)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
| Name: |
us-gaap_CashAndCashEquivalentsAtCarryingValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAggregate par or stated value of issued nonredeemable common stock (or common stock redeemable solely at the option of the issuer). This item includes treasury stock repurchased by the entity. Note: elements for number of nonredeemable common shares, par value and other disclosure concepts are in another section within stockholders' equity. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 30
-Article 5
| Name: |
us-gaap_CommonStockValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of receivables due from an entity that is affiliated with the reporting entity by means of direct or indirect ownership, due within 1 year (or 1 business cycle). Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 850
-SubTopic 10
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=6457730&loc=d3e39599-107864
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08.(k)(1))
-URI http://asc.fasb.org/extlink&oid=26873400&loc=d3e23780-122690
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 850
-SubTopic 10
-Section 50
-Paragraph 4
-URI http://asc.fasb.org/extlink&oid=6457730&loc=d3e39622-107864
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.3(a)(2))
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 850
-SubTopic 10
-Section 50
-Paragraph 1
-Subparagraph (d)
-URI http://asc.fasb.org/extlink&oid=6457730&loc=d3e39549-107864
| Name: |
us-gaap_DueFromAffiliateCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionSum of the carrying amounts as of the balance sheet date of all liabilities that are recognized. Liabilities are probable future sacrifices of economic benefits arising from present obligations of an entity to transfer assets or provide services to other entities in the future. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.19-26)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
| Name: |
us-gaap_Liabilities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of liabilities and equity items, including the portion of equity attributable to noncontrolling interests, if any. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.32)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 03
-Paragraph 25
-Article 7
| Name: |
us-gaap_LiabilitiesAndStockholdersEquity |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionTotal obligations incurred as part of normal operations that are expected to be paid during the following twelve months or within one business cycle, if longer. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.21)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
| Name: |
us-gaap_LiabilitiesCurrent |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_LiabilitiesCurrentAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe net carrying amount as of the balance sheet date of other indefinite-lived and finite-lived intangible assets that are not separately presented on the statement of financial position.
| Name: |
us-gaap_OtherIntangibleAssetsNet |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAggregate par or stated value of issued nonredeemable preferred stock (or preferred stock redeemable solely at the option of the issuer). This item includes treasury stock repurchased by the entity. Note: elements for number of nonredeemable preferred shares, par value and other disclosure concepts are in another section within stockholders' equity. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-URI http://asc.fasb.org/extlink&oid=27012166&loc=d3e187085-122770
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.28)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 29
-Article 5
| Name: |
us-gaap_PreferredStockValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe cumulative amount of the reporting entity's undistributed earnings or deficit. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.31(a)(3))
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 04
-Article 3
| Name: |
us-gaap_RetainedEarningsAccumulatedDeficit |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionTotal of all stockholders' equity (deficit) items, net of receivables from officers, directors, owners, and affiliates of the entity which are attributable to the parent. The amount of the economic entity's stockholders' equity attributable to the parent excludes the amount of stockholders' equity which is allocable to that ownership interest in subsidiary equity which is not attributable to the parent (noncontrolling interest, minority interest). This excludes temporary equity and is sometimes called permanent equity. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 310
-SubTopic 10
-Section S99
-Paragraph 2
-Subparagraph (SAB TOPIC 4.E)
-URI http://asc.fasb.org/extlink&oid=27010918&loc=d3e74512-122707
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29-31)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Staff Accounting Bulletin (SAB)
-Number Topic 4
-Section E
| Name: |
us-gaap_StockholdersEquity |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_StockholdersEquityAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.3.1.900
Consolidated Balance Sheets (Parenthetical) - $ / shares
|
Aug. 31, 2015 |
Aug. 31, 2014 |
| Statement of Financial Position [Abstract] |
|
|
| Preferred stock, par value |
$ 0.001
|
$ 0.001
|
| Preferred stock, shares authorized |
50,000,000
|
50,000,000
|
| Preferred stock, shares issued |
|
|
| Preferred stock, shares outstanding |
|
|
| Common stock, par value |
$ 0.001
|
$ 0.001
|
| Common stock, shares authorized |
750,000,000
|
750,000,000
|
| Common stock, shares issued |
58,532,156
|
54,634,056
|
| Common stock, shares outstanding |
58,532,156
|
54,634,056
|
| X |
- DefinitionFace amount or stated value per share of common stock. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 30
-Article 5
| Name: |
us-gaap_CommonStockParOrStatedValuePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
num:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe maximum number of common shares permitted to be issued by an entity's charter and bylaws. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 30
-Article 5
| Name: |
us-gaap_CommonStockSharesAuthorized |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionTotal number of common shares of an entity that have been sold or granted to shareholders (includes common shares that were issued, repurchased and remain in the treasury). These shares represent capital invested by the firm's shareholders and owners, and may be all or only a portion of the number of shares authorized. Shares issued include shares outstanding and shares held in the treasury. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 30
-Article 5
| Name: |
us-gaap_CommonStockSharesIssued |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of shares of common stock outstanding. Common stock represent the ownership interest in a corporation. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21463-112644
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-URI http://asc.fasb.org/extlink&oid=27012166&loc=d3e187085-122770
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 30
-Article 5
| Name: |
us-gaap_CommonStockSharesOutstanding |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionFace amount or stated value per share of preferred stock nonredeemable or redeemable solely at the option of the issuer. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.28)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 29
-Article 5
| Name: |
us-gaap_PreferredStockParOrStatedValuePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
num:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe maximum number of nonredeemable preferred shares (or preferred stock redeemable solely at the option of the issuer) permitted to be issued by an entity's charter and bylaws. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.28)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 29
-Article 5
| Name: |
us-gaap_PreferredStockSharesAuthorized |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionTotal number of nonredeemable preferred shares (or preferred stock redeemable solely at the option of the issuer) issued to shareholders (includes related preferred shares that were issued, repurchased, and remain in the treasury). May be all or portion of the number of preferred shares authorized. Excludes preferred shares that are classified as debt. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.28)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 29
-Article 5
| Name: |
us-gaap_PreferredStockSharesIssued |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionAggregate share number for all nonredeemable preferred stock (or preferred stock redeemable solely at the option of the issuer) held by stockholders. Does not include preferred shares that have been repurchased. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.28)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 29
-Article 5
| Name: |
us-gaap_PreferredStockSharesOutstanding |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
us-gaap_StatementOfFinancialPositionAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.3.1.900
Consolidated Statement of Operations - USD ($)
|
12 Months Ended |
Aug. 31, 2015 |
Aug. 31, 2014 |
| Income Statement [Abstract] |
|
|
| Revenue |
|
|
| Operating Expenses |
|
|
| Research and devlopment-related party |
|
$ 73,198
|
| Manufacturing and production |
$ 220,733
|
60,079
|
| General and administrative |
1,714,866
|
1,378,774
|
| Total operating expenses |
1,935,599
|
1,512,051
|
| Operating loss |
(1,935,599)
|
(1,512,051)
|
| Net loss |
$ (1,935,599)
|
$ (1,512,051)
|
| Net loss per share - basic and diluted |
$ (0.03)
|
$ (0.04)
|
| Weighted average shares outstanding |
57,297,917
|
40,112,465
|
| X |
- DefinitionThe amount of net income or loss for the period per each share in instances when basic and diluted earnings per share are the same amount and reported as a single line item on the face of the financial statements. Basic earnings per share is the amount of net income or loss for the period per each share of common stock or unit outstanding during the reporting period. Diluted earnings per share includes the amount of net income or loss for the period available to each share of common stock or common unit outstanding during the reporting period and to each share or unit that would have been outstanding assuming the issuance of common shares or units for all dilutive potential common shares or units outstanding during the reporting period.
| Name: |
us-gaap_EarningsPerShareBasicAndDiluted |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
num:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe aggregate total of expenses of managing and administering the affairs of an entity, including affiliates of the reporting entity, which are not directly or indirectly associated with the manufacture, sale or creation of a product or product line. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 225
-SubTopic 10
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.4)
-URI http://asc.fasb.org/extlink&oid=26872669&loc=d3e20235-122688
| Name: |
us-gaap_GeneralAndAdministrativeExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_IncomeStatementAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe aggregate costs incurred in the production of goods for sale. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 225
-SubTopic 10
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.2(a))
-URI http://asc.fasb.org/extlink&oid=26872669&loc=d3e20235-122688
| Name: |
us-gaap_ManufacturingCosts |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe portion of profit or loss for the period, net of income taxes, which is attributable to the parent. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Net Income
-URI http://asc.fasb.org/extlink&oid=51831255
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 28
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3602-108585
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 944
-SubTopic 225
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04.19)
-URI http://asc.fasb.org/extlink&oid=6879464&loc=d3e573970-122913
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 260
-SubTopic 10
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6371337&loc=d3e3550-109257
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 225
-SubTopic 10
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.18)
-URI http://asc.fasb.org/extlink&oid=26872669&loc=d3e20235-122688
Reference 6: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 944
-SubTopic 225
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04.22)
-URI http://asc.fasb.org/extlink&oid=6879464&loc=d3e573970-122913
Reference 7: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Other Comprehensive Income
-URI http://asc.fasb.org/extlink&oid=51831270
Reference 8: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 03
-Paragraph 19
-Article 5
Reference 9: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 04
-Paragraph 20
-Article 9
| Name: |
us-gaap_NetIncomeLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionGenerally recurring costs associated with normal operations except for the portion of these expenses which can be clearly related to production and included in cost of sales or services. Includes selling, general and administrative expense.
| Name: |
us-gaap_OperatingExpenses |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_OperatingExpensesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe net result for the period of deducting operating expenses from operating revenues.
| Name: |
us-gaap_OperatingIncomeLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe aggregate costs incurred (1) in a planned search or critical investigation aimed at discovery of new knowledge with the hope that such knowledge will be useful in developing a new product or service, a new process or technique, or in bringing about a significant improvement to an existing product or process; or (2) to translate research findings or other knowledge into a plan or design for a new product or process or for a significant improvement to an existing product or process whether intended for sale or the entity's use, during the reporting period charged to research and development projects, including the costs of developing computer software up to the point in time of achieving technological feasibility, and costs allocated in accounting for a business combination to in-process projects deemed to have no alternative future use. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 730
-SubTopic 10
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6420194&loc=d3e21568-108373
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 985
-SubTopic 20
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6501960&loc=d3e128462-111756
| Name: |
us-gaap_ResearchAndDevelopmentExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of revenue recognized from goods sold, services rendered, insurance premiums, or other activities that constitute an earning process. Includes, but is not limited to, investment and interest income before deduction of interest expense when recognized as a component of revenue, and sales and trading gain (loss). Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 225
-SubTopic 10
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.1)
-URI http://asc.fasb.org/extlink&oid=26872669&loc=d3e20235-122688
| Name: |
us-gaap_Revenues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAverage number of shares or units issued and outstanding that are used in calculating basic and diluted earnings per share (EPS).
| Name: |
us-gaap_WeightedAverageNumberOfShareOutstandingBasicAndDiluted |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.3.1.900
Statements of Stockholders' Equity (Deficit) - USD ($)
|
Common Stock [Member] |
Additional Paid-In Capital [Member] |
Accumulated Deficit [Member] |
Total |
| Balance at Aug. 31, 2013 |
$ 37,990
|
$ (37,952)
|
$ (38)
|
$ 0
|
| Balance, shares at Aug. 31, 2013 |
3,799,000
|
|
|
|
| Common stock issued in connection with reverse merger |
$ 10,988
|
89,012
|
|
100,000
|
| Common stock issued in connection with reverse merger, shares |
10,988,116
|
|
|
|
| Issuance of common stock for cash |
$ 676
|
183,824
|
|
184,500
|
| Issuance of common stock for cash, shares |
676,000
|
|
|
|
| Stock-based compensation |
$ 4,980
|
$ 1,240,005
|
|
1,244,985
|
| Stock-based compensation, shares |
4,979,940
|
|
|
|
| Net loss |
|
|
$ (1,512,051)
|
(1,512,051)
|
| Balance at Aug. 31, 2014 |
$ 54,634
|
$ 1,474,889
|
$ (1,549,389)
|
17,434
|
| Balance, shares at Aug. 31, 2014 |
54,634,056
|
|
|
|
| Stock-based compensation |
$ 3,898
|
$ 1,549,140
|
|
1,553,038
|
| Stock-based compensation, shares |
3,898,100
|
|
|
|
| Net loss |
|
|
$ (1,935,599)
|
(1,935,599)
|
| Balance at Aug. 31, 2015 |
$ 58,532
|
$ 3,024,029
|
$ (3,447,688)
|
$ (365,127)
|
| Balance, shares at Aug. 31, 2015 |
58,532,156
|
|
|
|
| X |
- DefinitionCommon stock issued in connection with reverse merger.
| Name: |
PTLF_CommonStockIssuedInConnectionWithReverseMerger |
| Namespace Prefix: |
PTLF_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionCommon stock issued in connection with reverse merger, shares.
| Name: |
PTLF_CommonStockIssuedInConnectionWithReverseMergerShares |
| Namespace Prefix: |
PTLF_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe portion of profit or loss for the period, net of income taxes, which is attributable to the parent. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Net Income
-URI http://asc.fasb.org/extlink&oid=51831255
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 28
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3602-108585
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 944
-SubTopic 225
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04.19)
-URI http://asc.fasb.org/extlink&oid=6879464&loc=d3e573970-122913
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 260
-SubTopic 10
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6371337&loc=d3e3550-109257
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 225
-SubTopic 10
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.18)
-URI http://asc.fasb.org/extlink&oid=26872669&loc=d3e20235-122688
Reference 6: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 944
-SubTopic 225
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04.22)
-URI http://asc.fasb.org/extlink&oid=6879464&loc=d3e573970-122913
Reference 7: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Other Comprehensive Income
-URI http://asc.fasb.org/extlink&oid=51831270
Reference 8: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 03
-Paragraph 19
-Article 5
Reference 9: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 04
-Paragraph 20
-Article 9
| Name: |
us-gaap_NetIncomeLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of shares issued which are neither cancelled nor held in the treasury.
| Name: |
us-gaap_SharesOutstanding |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of shares of stock issued during the period that is attributable to transactions involving issuance of stock not separately disclosed.
| Name: |
us-gaap_StockIssuedDuringPeriodSharesOther |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of shares (or other type of equity) issued during the period as a result of any equity-based compensation plan other than an employee stock ownership plan (ESOP), net of any shares forfeited. Shares issued could result from the issuance of restricted stock, the exercise of stock options, stock issued under employee stock purchase plans, and/or other employee benefit plans. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21463-112644
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-URI http://asc.fasb.org/extlink&oid=27012166&loc=d3e187085-122770
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 29, 30
-Article 5
| Name: |
us-gaap_StockIssuedDuringPeriodSharesShareBasedCompensation |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionValue of shares of stock issued during the period that is attributable to transactions involving issuance of stock not separately disclosed.
| Name: |
us-gaap_StockIssuedDuringPeriodValueOther |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionValue of stock (or other type of equity) issued during the period as a result of any equity-based compensation plan other than an employee stock ownership plan (ESOP), net of stock value of such awards forfeited. Stock issued could result from the issuance of restricted stock, the exercise of stock options, stock issued under employee stock purchase plans, and/or other employee benefit plans. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 1
-Subparagraph (c)
-URI http://asc.fasb.org/extlink&oid=6415400&loc=d3e5047-113901
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 2
-Subparagraph (d)(1)
-URI http://asc.fasb.org/extlink&oid=6415400&loc=d3e5070-113901
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-URI http://asc.fasb.org/extlink&oid=27012166&loc=d3e187085-122770
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 29, 30, 31
-Article 5
| Name: |
us-gaap_StockIssuedDuringPeriodValueShareBasedCompensation |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionTotal of all stockholders' equity (deficit) items, net of receivables from officers, directors, owners, and affiliates of the entity which are attributable to the parent. The amount of the economic entity's stockholders' equity attributable to the parent excludes the amount of stockholders' equity which is allocable to that ownership interest in subsidiary equity which is not attributable to the parent (noncontrolling interest, minority interest). This excludes temporary equity and is sometimes called permanent equity. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 310
-SubTopic 10
-Section S99
-Paragraph 2
-Subparagraph (SAB TOPIC 4.E)
-URI http://asc.fasb.org/extlink&oid=27010918&loc=d3e74512-122707
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29-31)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Staff Accounting Bulletin (SAB)
-Number Topic 4
-Section E
| Name: |
us-gaap_StockholdersEquity |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- Details
| Name: |
us-gaap_StatementEquityComponentsAxis=us-gaap_CommonStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementEquityComponentsAxis=us-gaap_AdditionalPaidInCapitalMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementEquityComponentsAxis=us-gaap_RetainedEarningsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.3.1.900
Consolidated Statements of Cash Flow - USD ($)
|
12 Months Ended |
Aug. 31, 2015 |
Aug. 31, 2014 |
| Net Cash from (used in) operating activities |
|
|
| Net loss |
$ (1,935,599)
|
$ (1,512,051)
|
| Stock-based compensation |
1,553,038
|
$ 1,244,985
|
| Accounts payable and accrued expenses |
360,527
|
|
| Net cash used in operating activities |
(22,034)
|
$ (267,066)
|
| Net cash from (used in) investing activities |
|
|
| Capital expenditure |
$ (600)
|
|
| Reverse acquisition |
|
$ 100,000
|
| Net cash provided by (used in) investing activities |
$ (600)
|
100,000
|
| Net cash provided by financiang activities |
|
|
| Proceeds from sale of common stock |
|
184,500
|
| Due from affiliate, net |
$ 5,782
|
$ (10,582)
|
| Proceeds from sharholder advance |
10,000
|
|
| Net cash provided by financiang activities |
15,782
|
$ 173,918
|
| Increase (decrease) in cash |
(6,852)
|
$ 6,852
|
| Cash, beginning of period |
$ 6,852
|
|
| Cash, end of period |
|
$ 6,852
|
| X |
- DefinitionAmount of currency on hand as well as demand deposits with banks or financial institutions. Includes other kinds of accounts that have the general characteristics of demand deposits. Also includes short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Excludes cash and cash equivalents within disposal group and discontinued operation. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Cash
-URI http://asc.fasb.org/extlink&oid=6506951
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section 45
-Paragraph 1
-Subparagraph (a)
-URI http://asc.fasb.org/extlink&oid=28358313&loc=d3e6676-107765
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 4
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3044-108585
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Cash Equivalents
-URI http://asc.fasb.org/extlink&oid=6507016
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.1)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
| Name: |
us-gaap_CashAndCashEquivalentsAtCarryingValue |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionAmount of increase (decrease) in cash and cash equivalents. Cash and cash equivalents are the amount of currency on hand as well as demand deposits with banks or financial institutions. Includes other kinds of accounts that have the general characteristics of demand deposits. Also includes short-term, highly liquid investments that are both readily convertible to known amounts of cash and so near their maturity that they present insignificant risk of changes in value because of changes in interest rates. Includes effect from exchange rate changes. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 24
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3521-108585
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 830
-SubTopic 230
-Section 45
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=49171198&loc=d3e33268-110906
| Name: |
us-gaap_CashAndCashEquivalentsPeriodIncreaseDecrease |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe increase (decrease) during the reporting period in the amounts payable to vendors for goods and services received and the amount of obligations and expenses incurred but not paid. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 28
-Subparagraph (a)
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3602-108585
| Name: |
us-gaap_IncreaseDecreaseInAccountsPayableAndAccruedLiabilities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash inflow (outflow) from financing activities, including discontinued operations. Financing activity cash flows include obtaining resources from owners and providing them with a return on, and a return of, their investment; borrowing money and repaying amounts borrowed, or settling the obligation; and obtaining and paying for other resources obtained from creditors on long-term credit. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 24
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3521-108585
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 26
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3574-108585
| Name: |
us-gaap_NetCashProvidedByUsedInFinancingActivities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_NetCashProvidedByUsedInFinancingActivitiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash inflow (outflow) from investing activities, including discontinued operations. Investing activity cash flows include making and collecting loans and acquiring and disposing of debt or equity instruments and property, plant, and equipment and other productive assets. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 24
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3521-108585
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 26
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3574-108585
| Name: |
us-gaap_NetCashProvidedByUsedInInvestingActivities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_NetCashProvidedByUsedInInvestingActivitiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAmount of cash inflow (outflow) from operating activities, including discontinued operations. Operating activity cash flows include transactions, adjustments, and changes in value not defined as investing or financing activities. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 24
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3521-108585
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 28
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3602-108585
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 25
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3536-108585
| Name: |
us-gaap_NetCashProvidedByUsedInOperatingActivities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_NetCashProvidedByUsedInOperatingActivitiesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe portion of profit or loss for the period, net of income taxes, which is attributable to the parent. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Net Income
-URI http://asc.fasb.org/extlink&oid=51831255
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 28
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3602-108585
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 944
-SubTopic 225
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04.19)
-URI http://asc.fasb.org/extlink&oid=6879464&loc=d3e573970-122913
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 260
-SubTopic 10
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6371337&loc=d3e3550-109257
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 225
-SubTopic 10
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.18)
-URI http://asc.fasb.org/extlink&oid=26872669&loc=d3e20235-122688
Reference 6: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 944
-SubTopic 225
-Section S99
-Paragraph 1
-Subparagraph (SX 210.7-04.22)
-URI http://asc.fasb.org/extlink&oid=6879464&loc=d3e573970-122913
Reference 7: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Other Comprehensive Income
-URI http://asc.fasb.org/extlink&oid=51831270
Reference 8: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 03
-Paragraph 19
-Article 5
Reference 9: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 04
-Paragraph 20
-Article 9
| Name: |
us-gaap_NetIncomeLoss |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash outflow associated with the acquisition of business during the period. The cash portion only of the acquisition price. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Investing Activities
-URI http://asc.fasb.org/extlink&oid=6516133
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 13
-Subparagraph (b)
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3213-108585
| Name: |
us-gaap_PaymentsToAcquireBusinessesGross |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash outflow for purchases of and capital improvements on property, plant and equipment (capital expenditures), software, and other intangible assets. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Investing Activities
-URI http://asc.fasb.org/extlink&oid=6516133
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 13
-Subparagraph (c)
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3213-108585
| Name: |
us-gaap_PaymentsToAcquireProductiveAssets |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash inflow from the collection of money previously advanced to an entity that is related to it but not strictly controlled. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 12
-Subparagraph (a)
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3179-108585
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Investing Activities
-URI http://asc.fasb.org/extlink&oid=6516133
| Name: |
us-gaap_ProceedsFromCollectionOfAdvanceToAffiliate |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash inflow from the additional capital contribution to the entity. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Financing Activities
-URI http://asc.fasb.org/extlink&oid=6513228
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 14
-Subparagraph (a)
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3255-108585
| Name: |
us-gaap_ProceedsFromIssuanceOfCommonStock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe aggregate amount of noncash, equity-based employee remuneration. This may include the value of stock or unit options, amortization of restricted stock or units, and adjustment for officers' compensation. As noncash, this element is an add back when calculating net cash generated by operating activities using the indirect method. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 28
-Subparagraph (a)
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3602-108585
| Name: |
us-gaap_ShareBasedCompensation |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
v3.3.1.900
Significant Accounting Policies
|
12 Months Ended |
Aug. 31, 2015 |
|---|
| Accounting Policies [Abstract] |
|
| Significant Accounting Policies |
NOTE 1 – SIGNIFICANT ACCOUNTING
POLICIES
Nature of Operations
PetLife Pharmaceuticals, Inc. (“Company”)
is a registered U.S. Veterinary Pharmaceutical company whose mission is to bring its scientifically proven, potentiated bioactive
medication and nutraceuticals — “Escozine for Pets™” — to the world of veterinary oncology. The Company
specializes in the research, development, sales and support of advanced drugs and nutraceuticals for pet cancer and autoimmune
related diseases such as arthritis.
A summary of the significant accounting
policies applied in the presentation of the accompanying consolidated financial statements follows:
Basis and business presentation
The Company was incorporated on April
5, 2002 under the laws of the State of Nevada as “Aztek Ventures Inc.” Effective November 13, 2007, we filed a Certificate
of Amendment to our Articles of Incorporation to change our name from “Aztek Ventures Inc.” to “Genesis Uranium
Corp.” Effective April 21, 2008, we amended our Articles of Incorporation to change our name from “Genesis Uranium
Corp.” to “Vault Technology Inc.” to reflect the change in our business focus beyond solely that of uranium exploration.
Effective July 10, 2009, we filed a Certificate of Amendment to our Articles of Incorporation to change our name from “Vault
Technology, Inc.” to “Modern Renewable Technologies, Inc.” (“Modern”). On May 27, 2011, Modern, merged
with Eco Ventures Group, Inc., and the name of the Company was changed to Eco Ventures Group, Inc. On July 15, 2013, the Company
entered into an Agreement and Plan of Merger with Clear TV Ventures, Inc. Under the terms of the merger, Clear TV became the surviving
corporation. On June 26, 2014, Eco Ventures Group, Inc. entered into an Agreement and Plan of Merger with its subsidiary, PetLife
Pharmaceuticals, Inc., a Nevada Corporation, with PetLife Pharmaceuticals, Inc. being the surviving entity. As part of that merger,
the name of the Company was changed to PetLife Pharmaceuticals, Inc. and each 15 shares of our common stock were exchanged for
one share in the surviving company. Effective August 12, 2014 we completed the closing of the Share Exchange Agreement and the
acquisition of Petlife and changed our name to Petlife Pharmaceuticals, Inc. All references herein to the number of shares outstanding
and per-share amounts have been retroactively restated to reflect the exchange ratio in the merger with PetLife Pharmaceuticals,
Inc.
All references that refer to (the “Company”
or “PetLife Pharmaceuticals, Inc.” or "Petlife" or “we” or “us” or “our”)
are PetLife Pharmaceuticals, Inc., the Registrant and its wholly and or majority owned subsidiaries. We are in the development
stage, as defined by Accounting Standards Codification subtopic 915-10, Development Stage Entities ("ASC 915-10") and
have developed and is launching a new generation of potentiated veterinary cancer medications and nutraceuticals, based on the
same patented formula “Escozine” and production processes that have been scientifically proven as an effective treatment
for cancer in humans for years. We have not generated any revenues to date, have incurred expenses and has sustained losses since
December 12, 2012 (date of inception). Consequently, our operations are subject to all the risks inherent in the establishment
of a new business enterprise. For the period from December 12, 2012 (date of inception) through August 31, 2015, we have accumulated
a deficit through its development stage of approximately $3,447,688.
The consolidated financial statements
include the accounts of the Company and its majority owned subsidiaries. All significant intercompany balances and transactions
have been eliminated in consolidation.
Merger and Corporate Restructure
On or about April 18, 2014, the Company
entered into a Share Exchange Agreement (the “Share Exchange”) with Petlife Corporation, a Delaware corporation (“Petlife”)
and the shareholders of Petlife Corporation (the “Shareholders”) for the exchange of all of the issued and outstanding
shares of Petlife. Effective at the Closing on August 12, 2014, these shares of Petlife were exchanged for approximately 47,000,000
fully paid non-assessable shares of the Company reflecting approximately 80% of the issued and outstanding shares of the Company
and acquisition of all assets and liabilities of the Company and was valued equated to the net assets of the Company as of the
date of the acquisition of $100,000. No liabilities were assumed. In connection with the share exchange, Petlife Corporation became
our wholly owned subsidiary.
Accordingly, the historical financial
statements are those of Petlife, the accounting acquirer, immediately following the consummation of the reverse acquisition. The
Company did not recognize goodwill or any intangible assets in connection with this transaction.
Estimates
The preparation of the financial statements
in conformity with generally accepted accounting principles requires management to make estimates and assumptions that affect certain
reported amounts and disclosures. Accordingly, actual results could differ from those estimates.
Revenue Recognition
The Company will recognize revenue in
accordance with Accounting Standards Codification subtopic 605-10, Revenue Recognition (“ASC 605-10”) which requires
that four basic criteria must be met before revenue can be recognized: (1) persuasive evidence of an arrangement exists; (2) delivery
has occurred; (3) the selling price is fixed and determinable; and (4) collectability is reasonably assured. Determination of criteria
(3) and (4) are based on management’s judgments regarding the fixed nature of the selling prices of the products delivered
and the collectability of those amounts. Provisions for discounts and rebates to customers, estimated returns and allowances, and
other adjustments will be provided for in the same period the related sales will be recorded.
ASC 605-10 incorporates Accounting Standards
Codification subtopic 605-25, Multiple-Element Arraignments (“ASC 605-25”). ASC 605-25 addresses accounting for arrangements
that may involve the delivery or performance of multiple products, services and/or rights to use assets. There was no effect on
implementing ASC 605-25 on the Company’s financial position and results of operations, since the Company has not started
generating revenue.
Cash
The Company considers cash to consist
of cash on hand and temporary investments having an original maturity of 90 days or less that are readily convertible into cash.
Property and Equipment
Property and equipment are stated at
cost. When retired or otherwise disposed, the related carrying value and accumulated depreciation are removed from the respective
accounts and the net difference less any amount realized from disposition, is reflected in earnings.
Long-Lived Assets
The Company follows FASB ASC 360-10-15-3,
“Impairment or Disposal of Long-lived Assets,” which established a “primary asset” approach to determine
the cash flow estimation period for a group of assets and liabilities that represents the unit of accounting for a long-lived asset
to be held and used. Long-lived assets to be held and used are reviewed for impairment whenever events or changes in circumstances
indicate that the carrying amount of an asset may not be recoverable. The carrying amount of a long-lived asset is not recoverable
if it exceeds the sum of the undiscounted cash flows expected to result from the use and eventual disposition of the asset. Long-lived
assets to be disposed of are reported at the lower of carrying amount or fair value less cost to sell.
Income Taxes
The Company has adopted Accounting Standards
Codification subtopic 740-10, Income Taxes (“ASC 740-10”) which requires the recognition of deferred tax liabilities
and assets for the expected future tax consequences of events that have been included in the financial statement or tax returns.
Under this method, deferred tax liabilities and assets are determined based on the difference between financial statements and
tax basis of assets and liabilities using enacted tax rates in effect for the year in which the differences are expected to reverse.
Temporary differences between taxable income reported for financial reporting purposes and income tax purposes consist primarily
of timing differences such as deferred officers’ compensation and stock based compensation accounting.
Net Loss per Common Share, basic
and diluted
The Company has adopted Accounting Standards
Codification subtopic 260-10, Earnings Per Share (“ASC 260-10”) specifying the computation, presentation and disclosure
requirements of earnings per share information. Basic loss per share has been calculated based upon the weighted average number
of common shares outstanding.
Stock based compensation
The Company follows Accounting Standards
Codification subtopic 718-10, Compensation (“ASC 718-10”) which requires that all share-based payments to both employees
and non-employees be recognized in the income statement based on their fair values.
As of August 31, 2015, the Company did
not have any issued or outstanding stock options.
Concentrations of Credit Risk
Financial instruments and related items,
which potentially subject the Company to concentrations of credit risk, consist primarily of cash, cash equivalents and trade receivables.
The Company places its cash and temporary cash investments with high credit quality institutions. At times, such investments may
be in excess of the FDIC insurance limit.
Research and Development
The Company accounts for research and
development costs in accordance with Accounting Standards Codification subtopic 730-10, Research and Development (“ASC 730-10”).
Under ASC 730-10, all research and development costs must be charged to expense as incurred. Accordingly, internal research and
development costs are expensed as incurred. Third-party research and development costs are expensed when the contracted work has
been performed or as milestone results have been achieved as defined under the applicable agreement. Company-sponsored research
and development costs related to both present and future products are expensed in the period incurred. The Company incurred costs
of $0 and $73,198 for research and development expenses for the years ended August 31, 2015 and 2014.
Reliance on Key Personnel and Consultants
The Company has eleven full-time employees,
three of which are executive officers. Additionally, the Company has consultants performing various specialized services. The Company
is heavily dependent on the continued active participation of these current executive officers, employees and key consultants.
The loss of any of the senior management or key consultants could significantly and negatively impact the business until adequate
replacements can be identified and put in place.
Fair Value
Accounting Standards Codification subtopic
825-10, Financial Instruments (“ASC 825-10”) requires disclosure of the fair value of certain financial instruments.
The carrying amount reported in the unaudited condensed consolidated balance sheet for accounts payable and accrued expenses, advances
and notes payable approximates fair value because of the immediate or short-term maturity of these financial instruments.
Recent Accounting Pronouncements
In May 2014, the FASB issued ASU 2014-09,
“Revenue from Contracts with Customers (Topic 606).” This guidance outlines a single, comprehensive model for accounting
for revenue from contracts with customers. We will adopt the standard in 2018. While we are currently assessing the impact of the
new standard, we do not expect this new guidance to have a material impact on our Consolidated Financial Statements.
No other new accounting pronouncement
issued or effective during the fiscal year had or is expected to have a material impact on the Consolidated Financial Statements.
|
| X |
- References
+ Details
| Name: |
us-gaap_AccountingPoliciesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for all significant accounting policies of the reporting entity. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 6
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18861-107790
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18780-107790
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18726-107790
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18743-107790
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 5
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18854-107790
| Name: |
us-gaap_SignificantAccountingPoliciesTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.3.1.900
Going Concern
|
12 Months Ended |
Aug. 31, 2015 |
|---|
| Organization, Consolidation and Presentation of Financial Statements [Abstract] |
|
| Going Concern |
NOTE 2 – GOING CONCERN
The accompanying
consolidated financial statements have been prepared on a going concern basis, which contemplates the realization of assets and
the satisfaction of liabilities in the normal course of business. As shown in the accompanying consolidated financial statements
as of August 31, 2015, the Company has an accumulated deficit of $3,447,688. As of August 31, 2015, the Company’s absence
of revenues, recurring losses and its need for additional financing to operate and fund its projected loss in 2016 raise
substantial doubt about its ability to continue as a going concern.
The Company’s existence is currently
dependent upon management’s ability to develop profitable operations and or upon obtaining additional financing to carry
out its planned business. Management is devoting substantially all of its efforts to the commercialization of its planned products,
as well as raising additional debt or equity financing in order to accelerate the development and commercialization of additional
products. There can be no assurance that the Company’s commercialization or financing efforts will result in profitable operations
or the resolution of the Company’s liquidity problems.
There can be no assurance that any additional
financings will be available to the Company on satisfactory terms and conditions, if at all. In the event the Company is unable
to continue as a going concern, it may elect or required to seek protection from its creditors by filing a voluntary petition in
bankruptcy or may be subject to an involuntary petition in bankruptcy. To date, management has not considered this alternative,
nor does management view it as a likely occurrence.
The accompanying consolidated statements
do not include any adjustments relating to the recoverability of assets and classification of assets and liabilities that might
be necessary should the Company be unable to continue as a going concern.
|
| X |
- References
+ Details
| Name: |
us-gaap_OrganizationConsolidationAndPresentationOfFinancialStatementsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure when substantial doubt is raised about the ability to continue as a going concern. Includes, but is not limited to, principal conditions or events that raised substantial doubt about the ability to continue as a going concern, management's evaluation of the significance of those conditions or events in relation to the ability to meet its obligations, and management's plans that alleviated or are intended to mitigate the conditions or events that raise substantial doubt about the ability to continue as a going concern. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 205
-SubTopic 40
-Section 50
-Paragraph 13
-URI http://asc.fasb.org/extlink&oid=51888302&loc=SL51888449-203568
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 205
-SubTopic 40
-Section 50
-Paragraph 12
-URI http://asc.fasb.org/extlink&oid=51888302&loc=SL51888443-203568
| Name: |
us-gaap_SubstantialDoubtAboutGoingConcernTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.3.1.900
Stockholders' Equity (Deficit)
|
12 Months Ended |
Aug. 31, 2015 |
|---|
| Equity [Abstract] |
|
| Stockholders' Equity (Deficit) |
NOTE 3 – STOCKHOLDERS’
EQUITY (DEFICIT)
Common Stock
During the year ended August 31, 2014,
the Company issued 4,979,940 shares of its $.001 par value common stock for services valued at $1,244,985.
In August, 2014, the Company sold 676,000
shares of its $.001 par value common stock for $184,500.
In September 2014, the Company issued
10,000 shares of its $.001 par value common stock for cash of $10,000. As of May 31, 2015, the common stock has not been issued
and is included in accrued expenses.
During the year ended August 31, 2015,
the Company issued 3,898,100 shares of its $.001 par value common stock for services valued at $1,553,038.
|
| X |
- References
+ Details
| Name: |
us-gaap_EquityAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for shareholders' equity comprised of portions attributable to the parent entity and noncontrolling interest, including other comprehensive income. Includes, but is not limited to, balances of common stock, preferred stock, additional paid-in capital, other capital and retained earnings, accumulated balance for each classification of other comprehensive income and amount of comprehensive income. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08.(d),(e))
-URI http://asc.fasb.org/extlink&oid=26873400&loc=d3e23780-122690
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 4
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21484-112644
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 310
-SubTopic 10
-Section S99
-Paragraph 2
-Subparagraph (SAB TOPIC 4.E)
-URI http://asc.fasb.org/extlink&oid=27010918&loc=d3e74512-122707
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 5
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21488-112644
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29-31)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 6: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section S99
-Paragraph 4
-Subparagraph (SAB TOPIC 4.C)
-URI http://asc.fasb.org/extlink&oid=27012166&loc=d3e187143-122770
Reference 7: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 6
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21506-112644
Reference 8: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-URI http://asc.fasb.org/extlink&oid=27012166&loc=d3e187085-122770
Reference 9: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 30
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=6405834&loc=d3e23285-112656
Reference 10: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21475-112644
Reference 11: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Preferred Stock
-URI http://asc.fasb.org/extlink&oid=6521494
Reference 12: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21463-112644
Reference 13: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 11
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21564-112644
Reference 14: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Staff Accounting Bulletin (SAB)
-Number Topic 4
-Section E
Reference 15: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Staff Accounting Bulletin (SAB)
-Number Topic 4
-Section C
| Name: |
us-gaap_StockholdersEquityNoteDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.3.1.900
Related Party Transactions
|
12 Months Ended |
Aug. 31, 2015 |
|---|
| Related Party Transactions [Abstract] |
|
| Related Party Transactions |
NOTE 4 – RELATED PARTY TRANSACTIONS
On December 30, 2013, the Company entered
into an agreement with Medolife Corporation ("Medolife), an affiliated company,
for the performance of certain ongoing services and maintenance related to
a scorpion reservation , together with the providing of other services such as product
operations, research and development to support the Company's product marketing. During the year ended August 31, 2014
Medolife invoiced the Company for services and maintenance of $165,800. During year ended August 31, 2014, Medolife received
proceeds from the sale of the Company’s common stock totaling $184,500. The Company was owed $4,800 and $10,582
from Medolife as of
August 31, 2015 and 2014.
On August 1, 2014, the Company entered
into a patent license agreement with a shareholder for a polarized scorpion venom solution and a method for making polarized scorpion
venom solution for veterinary use. The licensor has provided Medolife, an affiliated company by common shareholders, with an exclusive
right to the polarized scorpion venom patent with the exception of the rights for veterinary use. The Company will manufacture,
use, and sell polarized scorpion venom containing such patented improvements for veterinary use.
|
| X |
- DefinitionThe entire disclosure for related party transactions. Examples of related party transactions include transactions between (a) a parent company and its subsidiary; (b) subsidiaries of a common parent; (c) and entity and its principal owners; and (d) affiliates. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 850
-SubTopic 10
-Section 50
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=6457730&loc=d3e39603-107864
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 850
-SubTopic 10
-Section 50
-Paragraph 4
-URI http://asc.fasb.org/extlink&oid=6457730&loc=d3e39622-107864
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 850
-SubTopic 10
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6457730&loc=d3e39549-107864
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 850
-SubTopic 10
-Section 50
-Paragraph 5
-URI http://asc.fasb.org/extlink&oid=6457730&loc=d3e39678-107864
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 850
-SubTopic 10
-Section 50
-Paragraph 6
-URI http://asc.fasb.org/extlink&oid=6457730&loc=d3e39691-107864
Reference 6: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08.(k))
-URI http://asc.fasb.org/extlink&oid=26873400&loc=d3e23780-122690
Reference 7: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 04
-Paragraph b
-Article 3A
| Name: |
us-gaap_RelatedPartyTransactionsDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.3.1.900
Income Taxes
|
12 Months Ended |
Aug. 31, 2015 |
|---|
| Income Tax Disclosure [Abstract] |
|
| Income Taxes |
NOTE 5 – INCOME TAXES
As of August 31, 2015 the
Company has accumulated losses of approximately $3,447,000. A deferred tax asset has been provided and
has a 100% valuation allowance.
|
| X |
- DefinitionThe entire disclosure for income taxes. Disclosures may include net deferred tax liability or asset recognized in an enterprise's statement of financial position, net change during the year in the total valuation allowance, approximate tax effect of each type of temporary difference and carryforward that gives rise to a significant portion of deferred tax liabilities and deferred tax assets, utilization of a tax carryback, and tax uncertainties information. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.4-08.(h))
-URI http://asc.fasb.org/extlink&oid=26873400&loc=d3e23780-122690
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 740
-SubTopic 10
-Section 50
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=6907707&loc=d3e32559-109319
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 740
-SubTopic 10
-Section 50
-Paragraph 15
-URI http://asc.fasb.org/extlink&oid=6907707&loc=d3e32718-109319
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 740
-SubTopic 10
-Section 50
-Paragraph 9
-URI http://asc.fasb.org/extlink&oid=6907707&loc=d3e32639-109319
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 740
-SubTopic 10
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=6907707&loc=d3e32537-109319
| Name: |
us-gaap_IncomeTaxDisclosureTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.3.1.900
Share Exchange Agreement
|
12 Months Ended |
Aug. 31, 2015 |
|---|
| Share Exchange Agreement |
|
| Share Exchange Agreement |
NOTE 6 – SHARE EXCHANGE AGREEMENT
On April 18, 2014, Petlife Corporation entered into a Share
Exchange agreement with PetLife Pharmaceuticals, Inc. (formerly Clear TV Ventures, Inc.), a publicly traded company. The shares
of Petlife Corporation were exchanged for 47,000,000, or 80% of the issued and outstanding shares of PetLife Pharmaceuticals, Inc.
The closing of the Share Exchange Agreement
was conditioned upon certain, limited customary representations and warranties as well as conditions to close such as the total
issued and outstanding shares of the Company being limited to 58,000,000 issued and outstanding post-closing.
The Share Exchange Agreement closed
on August 11, 2014 with the issuance of 47,000,000 shares to shareholders or designees of Petlife Corporation. The shares of common
stock of PetLife Pharmaceuticals, Inc. issued in the exchange to the Petlife Corporation shareholders were not registered under
the Securities Act of 1933, as amended (the “Securities Act”), or the securities laws of any state, and were in each
case offered, sold and issued in reliance upon the exemption from registration provided by Section 4(2) of the Securities Act,
as a transaction by an issuer not involving a public offering, and Rule 506 of Regulation D promulgated thereunder. The Company
relied on such exemptions based in part on written representations made by the Company shareholders, including representations
with respect to each member’s status as an accredited investor and investment intent with respect to the acquired securities.
The shares of common stock issued in the Exchange to the Company shareholders may not be offered or sold absent registration or
an applicable exemption from the registration requirements of the Securities Act, and each of the certificates or instruments evidencing
such shares bears a legend to that effect.
The transaction was recorded as a reverse
acquisition whereby Petlife Corporation was considered to be the accounting acquirer as its shareholders retained control of Petlife
Pharmaceuticals, Inc., after the exchange, although Petlife Pharmaceuticals, Inc., is the legal parent company. The share exchange
was treated as a recapitalization of Petlife Corporation (and its historical financial statements) is the continuing entity for
financial reporting purposes.
|
| X |
- References
+ Details
| Name: |
PTLF_ShareExchangeAgreementAbstract |
| Namespace Prefix: |
PTLF_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionShare Exchange Agreement Disclosure [Text Block]
| Name: |
PTLF_ShareExchangeAgreementDisclosureTextBlock |
| Namespace Prefix: |
PTLF_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.3.1.900
Subsequent Events
|
12 Months Ended |
Aug. 31, 2015 |
|---|
| Subsequent Events [Abstract] |
|
| Subsequent Events |
NOTE 7 – SUBSEQUENT EVENTS
Pursuant to agreement, effective November
13, 2015 Bruce Niswander and Sebastian Serrell-Watts resigned as officers and directors of the Company.
On November 21, 2015 the Company entered
into a Reorganization and Stock Purchase Agreement with Alexian Scientific, Inc. Closing of the agreement is conditioned on the
Company obtaining funding of a minimum of $10,000,000. At closing, the Company will issue 38,777,630 shares of common stock to
designees of Alexian in consideration for 100% of the equity of Alexian, the Company will appoint designees of Alexian as its officers
and directors, and all of the current officers and directors of the Company will resign. As part of the agreement with Alexian,
shareholders of the Company cancelled certain shares of common stock of the Company such that subsequent to closing there will
be a total of 80,786,730 shares of common stock outstanding.
Effective November 20, 2015 in connection
with the proposed Alexian transaction, principals of the Company cancelled and returned to treasury a total of 21,583,000 shares
of common stock held by them.
Also effective November 20, 2015, the
Company issued a total of 626,000 shares of common stock to investors and issued 530,200 shares of common stock to officers and
employees of the Company in lieu of salary.
|
| X |
- References
+ Details
| Name: |
us-gaap_SubsequentEventsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe entire disclosure for significant events or transactions that occurred after the balance sheet date through the date the financial statements were issued or the date the financial statements were available to be issued. Examples include: the sale of a capital stock issue, purchase of a business, settlement of litigation, catastrophic loss, significant foreign exchange rate changes, loans to insiders or affiliates, and transactions not in the ordinary course of business.
| Name: |
us-gaap_SubsequentEventsTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.3.1.900
Significant Accounting Policies (Policies)
|
12 Months Ended |
Aug. 31, 2015 |
|---|
| Accounting Policies [Abstract] |
|
| Nature of Operations |
Nature of Operations
PetLife Pharmaceuticals, Inc. (“Company”)
is a registered U.S. Veterinary Pharmaceutical company whose mission is to bring its scientifically proven, potentiated bioactive
medication and nutraceuticals — “Escozine for Pets™” — to the world of veterinary oncology. The
Company specializes in the research, development, sales and support of advanced drugs and nutraceuticals for pet cancer and autoimmune
related diseases such as arthritis.
|
| Basis and Business Presentation |
Basis and business presentation
The Company was incorporated on April
5, 2002 under the laws of the State of Nevada as “Aztek Ventures Inc.” Effective November 13, 2007, we filed a Certificate
of Amendment to our Articles of Incorporation to change our name from “Aztek Ventures Inc.” to “Genesis Uranium
Corp.” Effective April 21, 2008, we amended our Articles of Incorporation to change our name from “Genesis Uranium
Corp.” to “Vault Technology Inc.” to reflect the change in our business focus beyond solely that of uranium exploration.
Effective July 10, 2009, we filed a Certificate of Amendment to our Articles of Incorporation to change our name from “Vault
Technology, Inc.” to “Modern Renewable Technologies, Inc.” (“Modern”). On May 27, 2011, Modern, merged
with Eco Ventures Group, Inc., and the name of the Company was changed to Eco Ventures Group, Inc. On July 15, 2013, the Company
entered into an Agreement and Plan of Merger with Clear TV Ventures, Inc. Under the terms of the merger, Clear TV became the surviving
corporation. On June 26, 2014, Eco Ventures Group, Inc. entered into an Agreement and Plan of Merger with its subsidiary, PetLife
Pharmaceuticals, Inc., a Nevada Corporation, with PetLife Pharmaceuticals, Inc. being the surviving entity. As part of that merger,
the name of the Company was changed to PetLife Pharmaceuticals, Inc. and each 15 shares of our common stock were exchanged for
one share in the surviving company. Effective August 12, 2014 we completed the closing of the Share Exchange Agreement and the
acquisition of Petlife and changed our name to Petlife Pharmaceuticals, Inc. All references herein to the number of shares outstanding
and per-share amounts have been retroactively restated to reflect the exchange ratio in the merger with PetLife Pharmaceuticals,
Inc.
All references that refer to (the “Company”
or “PetLife Pharmaceuticals, Inc.” or "Petlife" or “we” or “us” or “our”)
are PetLife Pharmaceuticals, Inc., the Registrant and its wholly and or majority owned subsidiaries. We are in the development
stage, as defined by Accounting Standards Codification subtopic 915-10, Development Stage Entities ("ASC 915-10") and
have developed and is launching a new generation of potentiated veterinary cancer medications and nutraceuticals, based on the
same patented formula “Escozine” and production processes that have been scientifically proven as an effective treatment
for cancer in humans for years. We have not generated any revenues to date, have incurred expenses and has sustained losses since
December 12, 2012 (date of inception). Consequently, our operations are subject to all the risks inherent in the establishment
of a new business enterprise. For the period from December 12, 2012 (date of inception) through August 31, 2015, we have accumulated
a deficit through its development stage of approximately $3,447,688.
The consolidated financial statements
include the accounts of the Company and its majority owned subsidiaries. All significant intercompany balances and transactions
have been eliminated in consolidation.
|
| Merger and Corporate Restructure |
Merger and Corporate Restructure
On or about April 18, 2014, the Company
entered into a Share Exchange Agreement (the “Share Exchange”) with Petlife Corporation, a Delaware corporation (“Petlife”)
and the shareholders of Petlife Corporation (the “Shareholders”) for the exchange of all of the issued and outstanding
shares of Petlife. Effective at the Closing on August 12, 2014, these shares of Petlife were exchanged for approximately 47,000,000
fully paid non-assessable shares of the Company reflecting approximately 80% of the issued and outstanding shares of the Company
and acquisition of all assets and liabilities of the Company and was valued equated to the net assets of the Company as of the
date of the acquisition of $100,000. No liabilities were assumed. In connection with the share exchange, Petlife Corporation became
our wholly owned subsidiary.
Accordingly, the historical financial
statements are those of Petlife, the accounting acquirer, immediately following the consummation of the reverse acquisition. The
Company did not recognize goodwill or any intangible assets in connection with this transaction.
|
| Estimates |
Estimates
The preparation of the financial statements
in conformity with generally accepted accounting principles requires management to make estimates and assumptions that affect
certain reported amounts and disclosures. Accordingly, actual results could differ from those estimates.
|
| Revenue Recognition |
Revenue Recognition
The Company will recognize revenue in
accordance with Accounting Standards Codification subtopic 605-10, Revenue Recognition (“ASC 605-10”) which requires
that four basic criteria must be met before revenue can be recognized: (1) persuasive evidence of an arrangement exists; (2) delivery
has occurred; (3) the selling price is fixed and determinable; and (4) collectability is reasonably assured. Determination of criteria
(3) and (4) are based on management’s judgments regarding the fixed nature of the selling prices of the products delivered
and the collectability of those amounts. Provisions for discounts and rebates to customers, estimated returns and allowances, and
other adjustments will be provided for in the same period the related sales will be recorded.
ASC 605-10 incorporates Accounting
Standards Codification subtopic 605-25, Multiple-Element Arraignments (“ASC 605-25”). ASC 605-25 addresses accounting
for arrangements that may involve the delivery or performance of multiple products, services and/or rights to use assets. There
was no effect on implementing ASC 605-25 on the Company’s financial position and results of operations, since the Company
has not started generating revenue.
|
| Cash |
Cash
The Company considers cash to consist
of cash on hand and temporary investments having an original maturity of 90 days or less that are readily convertible into cash.
|
| Property and Equipment |
Property and Equipment
Property and equipment are stated at
cost. When retired or otherwise disposed, the related carrying value and accumulated depreciation are removed from the respective
accounts and the net difference less any amount realized from disposition, is reflected in earnings.
|
| Long-Lived Assets |
Long-Lived Assets
The Company follows FASB ASC 360-10-15-3,
“Impairment or Disposal of Long-lived Assets,” which established a “primary asset” approach to determine
the cash flow estimation period for a group of assets and liabilities that represents the unit of accounting for a long-lived
asset to be held and used. Long-lived assets to be held and used are reviewed for impairment whenever events or changes in circumstances
indicate that the carrying amount of an asset may not be recoverable. The carrying amount of a long-lived asset is not recoverable
if it exceeds the sum of the undiscounted cash flows expected to result from the use and eventual disposition of the asset. Long-lived
assets to be disposed of are reported at the lower of carrying amount or fair value less cost to sell.
|
| Income Taxes |
Income Taxes
The Company has adopted Accounting
Standards Codification subtopic 740-10, Income Taxes (“ASC 740-10”) which requires the recognition of deferred tax
liabilities and assets for the expected future tax consequences of events that have been included in the financial statement or
tax returns. Under this method, deferred tax liabilities and assets are determined based on the difference between financial statements
and tax basis of assets and liabilities using enacted tax rates in effect for the year in which the differences are expected to
reverse. Temporary differences between taxable income reported for financial reporting purposes and income tax purposes consist
primarily of timing differences such as deferred officers’ compensation and stock based compensation accounting.
|
| Net Loss Per Common Share, Basic and Diluted |
Net Loss per Common Share, basic
and diluted
The Company has adopted Accounting
Standards Codification subtopic 260-10, Earnings Per Share (“ASC 260-10”) specifying the computation, presentation
and disclosure requirements of earnings per share information. Basic loss per share has been calculated based upon the weighted
average number of common shares outstanding.
|
| Stock Based Compensation |
Stock based compensation
The Company follows Accounting Standards
Codification subtopic 718-10, Compensation (“ASC 718-10”) which requires that all share-based payments to both employees
and non-employees be recognized in the income statement based on their fair values.
As of August 31, 2015, the Company
did not have any issued or outstanding stock options.
|
| Concentrations of Credit Risk |
Concentrations of Credit Risk
Financial instruments and related items,
which potentially subject the Company to concentrations of credit risk, consist primarily of cash, cash equivalents and trade
receivables. The Company places its cash and temporary cash investments with high credit quality institutions. At times, such
investments may be in excess of the FDIC insurance limit.
|
| Research and Development |
Research and Development
The Company accounts for research and
development costs in accordance with Accounting Standards Codification subtopic 730-10, Research and Development (“ASC 730-10”).
Under ASC 730-10, all research and development costs must be charged to expense as incurred. Accordingly, internal research and
development costs are expensed as incurred. Third-party research and development costs are expensed when the contracted work has
been performed or as milestone results have been achieved as defined under the applicable agreement. Company-sponsored research
and development costs related to both present and future products are expensed in the period incurred. The Company incurred costs
of $0 and $73,198 for research and development expenses for the years ended August 31, 2015 and 2014.
|
| Reliance on Key Personnel and Consultants |
Reliance on Key Personnel and Consultants
The Company has eleven full-time employees,
three of which are executive officers. Additionally, the Company has consultants performing various specialized services. The
Company is heavily dependent on the continued active participation of these current executive officers, employees and key consultants.
The loss of any of the senior management or key consultants could significantly and negatively impact the business until adequate
replacements can be identified and put in place.
|
| Fair Value |
Fair Value
Accounting Standards Codification subtopic
825-10, Financial Instruments (“ASC 825-10”) requires disclosure of the fair value of certain financial instruments.
The carrying amount reported in the unaudited condensed consolidated balance sheet for accounts payable and accrued expenses,
advances and notes payable approximates fair value because of the immediate or short-term maturity of these financial instruments.
|
| Recent Accounting Pronouncements |
Recent Accounting Pronouncements
In May 2014, the FASB issued ASU 2014-09,
“Revenue from Contracts with Customers (Topic 606).” This guidance outlines a single, comprehensive model for accounting
for revenue from contracts with customers. We will adopt the standard in 2018. While we are currently assessing the impact of the
new standard, we do not expect this new guidance to have a material impact on our Consolidated Financial Statements.
No other new accounting pronouncement
issued or effective during the fiscal year had or is expected to have a material impact on the Consolidated Financial Statements.
|
| X |
- DefinitionNature of Operations [Policy Text Block]
| Name: |
PTLF_NatureOfOperationsPolicyTextBlock |
| Namespace Prefix: |
PTLF_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionReliance on Key Personnel and Consultants [Policy Text Block]
| Name: |
PTLF_RelianceOnKeyPersonnelAndConsultantsPolicyTextBlock |
| Namespace Prefix: |
PTLF_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
us-gaap_AccountingPoliciesAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for basis of accounting, or basis of presentation, used to prepare the financial statements (for example, US Generally Accepted Accounting Principles, Other Comprehensive Basis of Accounting, IFRS).
| Name: |
us-gaap_BasisOfAccountingPolicyPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for completed business combinations (purchase method, acquisition method or combination of entities under common control). This accounting policy may include a general discussion of the purchase method or acquisition method of accounting (including for example, the treatment accorded contingent consideration, the identification of assets and liabilities, the purchase price allocation process, how the fair values of acquired assets and liabilities are determined) and the entity's specific application thereof. An entity that acquires another entity in a leveraged buyout transaction generally discloses the accounting policy followed by the acquiring entity in determining the basis used to value its interest in the acquired entity, and the rationale for that accounting policy. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18780-107790
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 805
-SubTopic 10
-Section 05
-Paragraph 4
-Subparagraph (a)-(d)
-URI http://asc.fasb.org/extlink&oid=6909625&loc=d3e227-128457
| Name: |
us-gaap_BusinessCombinationsPolicy |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for cash and cash equivalents, including the policy for determining which items are treated as cash equivalents. Other information that may be disclosed includes (1) the nature of any restrictions on the entity's use of its cash and cash equivalents, (2) whether the entity's cash and cash equivalents are insured or expose the entity to credit risk, (3) the classification of any negative balance accounts (overdrafts), and (4) the carrying basis of cash equivalents (for example, at cost) and whether the carrying amount of cash equivalents approximates fair value. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 305
-SubTopic 10
-Section 05
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6375392&loc=d3e26790-107797
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6367179&loc=d3e4273-108586
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Cash
-URI http://asc.fasb.org/extlink&oid=6506951
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18780-107790
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Cash Equivalents
-URI http://asc.fasb.org/extlink&oid=6507016
Reference 6: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Financial Reporting Release (FRR)
-Number 203
-Paragraph 02-03
Reference 7: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 1
-Article 5
| Name: |
us-gaap_CashAndCashEquivalentsPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for credit risk. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 825
-SubTopic 10
-Section 55
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6875567&loc=d3e14489-108613
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 942
-SubTopic 825
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=51676700&loc=d3e61082-112788
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 944
-SubTopic 825
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=28088331&loc=SL29635902-196195
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 825
-SubTopic 10
-Section 50
-Paragraph 21
-URI http://asc.fasb.org/extlink&oid=49121117&loc=d3e13537-108611
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 825
-SubTopic 10
-Section 50
-Paragraph 20
-URI http://asc.fasb.org/extlink&oid=49121117&loc=d3e13531-108611
Reference 6: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 942
-SubTopic 825
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=51676700&loc=d3e61044-112788
| Name: |
us-gaap_ConcentrationRiskCreditRisk |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for computing basic and diluted earnings or loss per share for each class of common stock and participating security. Addresses all significant policy factors, including any antidilutive items that have been excluded from the computation and takes into account stock dividends, splits and reverse splits that occur after the balance sheet date of the latest reporting period but before the issuance of the financial statements. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 260
-SubTopic 10
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6371337&loc=d3e3550-109257
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 260
-SubTopic 10
-Section 50
-Paragraph 1
-Subparagraph (c)
-URI http://asc.fasb.org/extlink&oid=6371337&loc=d3e3550-109257
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 260
-SubTopic 10
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=6371337&loc=d3e3630-109257
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18780-107790
| Name: |
us-gaap_EarningsPerSharePolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for fair value measurements of financial and non-financial assets, liabilities and instruments classified in shareholders' equity. Disclosures include, but are not limited to, how an entity that manages a group of financial assets and liabilities on the basis of its net exposure measures the fair value of those assets and liabilities.
| Name: |
us-gaap_FairValueMeasurementPolicyPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for recognizing and measuring the impairment of long-lived assets. An entity also may disclose its accounting policy for long-lived assets to be sold. This policy excludes goodwill and intangible assets. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18780-107790
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 360
-SubTopic 10
-Section 05
-Paragraph 4
-URI http://asc.fasb.org/extlink&oid=51717228&loc=d3e202-110218
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Staff Accounting Bulletin (SAB)
-Number Topic 5
-Section CC
-Subsection 3
| Name: |
us-gaap_ImpairmentOrDisposalOfLongLivedAssetsPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for income taxes, which may include its accounting policies for recognizing and measuring deferred tax assets and liabilities and related valuation allowances, recognizing investment tax credits, operating loss carryforwards, tax credit carryforwards, and other carryforwards, methodologies for determining its effective income tax rate and the characterization of interest and penalties in the financial statements. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18780-107790
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 740
-SubTopic 10
-Section 50
-Paragraph 19
-URI http://asc.fasb.org/extlink&oid=6907707&loc=d3e32840-109319
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 740
-SubTopic 30
-Section 05
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6423966&loc=d3e40913-109327
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 954
-SubTopic 740
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6491622&loc=d3e9504-115650
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 740
-SubTopic 10
-Section 50
-Paragraph 17
-Subparagraph (b)
-URI http://asc.fasb.org/extlink&oid=6907707&loc=d3e32809-109319
Reference 6: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 740
-SubTopic 10
-Section 45
-Paragraph 25
-URI http://asc.fasb.org/extlink&oid=37586315&loc=d3e32247-109318
Reference 7: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 740
-SubTopic 10
-Section 45
-Paragraph 28
-URI http://asc.fasb.org/extlink&oid=37586315&loc=d3e32280-109318
| Name: |
us-gaap_IncomeTaxPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy pertaining to new accounting pronouncements that may impact the entity's financial reporting. Includes, but is not limited to, quantification of the expected or actual impact.
| Name: |
us-gaap_NewAccountingPronouncementsPolicyPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for long-lived, physical assets used in the normal conduct of business and not intended for resale. Includes, but is not limited to, basis of assets, depreciation and depletion methods used, including composite deprecation, estimated useful lives, capitalization policy, accounting treatment for costs incurred for repairs and maintenance, capitalized interest and the method it is calculated, disposals and impairments. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 360
-SubTopic 10
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6391035&loc=d3e2868-110229
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18780-107790
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.13(a))
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
| Name: |
us-gaap_PropertyPlantAndEquipmentPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for costs it has incurred (1) in a planned search or critical investigation aimed at discovery of new knowledge with the hope that such knowledge will be useful in developing a new product or service, a new process or technique, or in bringing about a significant improvement to an existing product or process; or (2) to translate research findings or other knowledge into a plan or design for a new product or process or for a significant improvement to an existing product or process. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 730
-SubTopic 10
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6420194&loc=d3e21568-108373
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18780-107790
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Research and Development
-URI http://asc.fasb.org/extlink&oid=6523717
| Name: |
us-gaap_ResearchAndDevelopmentExpensePolicy |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for revenue recognition. If the entity has different policies for different types of revenue transactions, the policy for each material type of transaction is generally disclosed. If a sales transaction has multiple element arrangements (for example, delivery of multiple products, services or the rights to use assets) the disclosure may indicate the accounting policy for each unit of accounting as well as how units of accounting are determined and valued. The disclosure may encompass important judgment as to appropriateness of principles related to recognition of revenue. The disclosure also may indicate the entity's treatment of any unearned or deferred revenue that arises from the transaction. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 4
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18823-107790
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18780-107790
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18726-107790
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 605
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SAB TOPIC 13.B.Q1)
-URI http://asc.fasb.org/extlink&oid=27012821&loc=d3e214044-122780
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Staff Accounting Bulletin (SAB)
-Number Topic 13
-Section B
-Paragraph Question 1
| Name: |
us-gaap_RevenueRecognitionPolicyTextBlock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for stock option and stock incentive plans. This disclosure may include (1) the types of stock option or incentive plans sponsored by the entity (2) the groups that participate in (or are covered by) each plan (3) significant plan provisions and (4) how stock compensation is measured, and the methodologies and significant assumptions used to determine that measurement. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6415400&loc=d3e5047-113901
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 235
-SubTopic 10
-Section 50
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=51655414&loc=d3e18780-107790
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 718
-SubTopic 10
-Section 50
-Paragraph 2
-Subparagraph (b),(f)
-URI http://asc.fasb.org/extlink&oid=6415400&loc=d3e5070-113901
| Name: |
us-gaap_ShareBasedCompensationOptionAndIncentivePlansPolicy |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of accounting policy for the use of estimates in the preparation of financial statements in conformity with generally accepted accounting principles. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 275
-SubTopic 10
-Section 50
-Paragraph 4
-URI http://asc.fasb.org/extlink&oid=51801978&loc=d3e6061-108592
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 275
-SubTopic 10
-Section 50
-Paragraph 9
-URI http://asc.fasb.org/extlink&oid=51801978&loc=d3e6143-108592
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 275
-SubTopic 10
-Section 50
-Paragraph 8
-URI http://asc.fasb.org/extlink&oid=51801978&loc=d3e6132-108592
| Name: |
us-gaap_UseOfEstimates |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
nonnum:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.3.1.900
Significant Accounting Policies (Details Narrative) - USD ($)
|
|
|
12 Months Ended |
Jun. 26, 2014 |
Apr. 18, 2014 |
Aug. 31, 2015 |
Aug. 31, 2014 |
| Accumulated deficit |
|
|
$ (3,447,688)
|
$ (1,512,089)
|
| Shares exchanged for approximately fully paid non-assessable shaes approximately |
|
47,000,000
|
|
|
| Percentage of issued and outstanding shares for acquisition of assets and liabilities |
|
80.00%
|
|
|
| Assets acquisition value |
|
$ 100,000
|
|
|
| Research and development expenses |
|
|
|
$ 73,198
|
| Eco Ventures Group, Inc [Member] |
|
|
|
|
| Number of common stock shares exchanges for shares |
15
|
|
|
|
| X |
- DefinitionPercentage of issued and outstanding shares for acquisition of assets and liabilities.
| Name: |
PTLF_PercentageOfIssuedAndOutstandingSharesForAcquisitionOfAssetsAndLiabilities |
| Namespace Prefix: |
PTLF_ |
| Data Type: |
num:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe aggregate costs incurred (1) in a planned search or critical investigation aimed at discovery of new knowledge with the hope that such knowledge will be useful in developing a new product or service, a new process or technique, or in bringing about a significant improvement to an existing product or process; or (2) to translate research findings or other knowledge into a plan or design for a new product or process or for a significant improvement to an existing product or process whether intended for sale or the entity's use, during the reporting period charged to research and development projects, including the costs of developing computer software up to the point in time of achieving technological feasibility, and costs allocated in accounting for a business combination to in-process projects deemed to have no alternative future use. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 730
-SubTopic 10
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6420194&loc=d3e21568-108373
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 985
-SubTopic 20
-Section 50
-Paragraph 1
-URI http://asc.fasb.org/extlink&oid=6501960&loc=d3e128462-111756
| Name: |
us-gaap_ResearchAndDevelopmentExpense |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cumulative amount of the reporting entity's undistributed earnings or deficit. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.31(a)(3))
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 04
-Article 3
| Name: |
us-gaap_RetainedEarningsAccumulatedDeficit |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of shares of stock issued during the period pursuant to acquisitions. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-URI http://asc.fasb.org/extlink&oid=27012166&loc=d3e187085-122770
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.28,29)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 29, 30
-Article 5
| Name: |
us-gaap_StockIssuedDuringPeriodSharesAcquisitions |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of shares issued during the period as a result of the conversion of convertible securities. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-URI http://asc.fasb.org/extlink&oid=27012166&loc=d3e187085-122770
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29-30)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21463-112644
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 3
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21475-112644
| Name: |
us-gaap_StockIssuedDuringPeriodSharesConversionOfConvertibleSecurities |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionValue of stock issued pursuant to acquisitions during the period. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21463-112644
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-URI http://asc.fasb.org/extlink&oid=27012166&loc=d3e187085-122770
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29-31)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
| Name: |
us-gaap_StockIssuedDuringPeriodValueAcquisitions |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
v3.3.1.900
| X |
- References
+ Details
| Name: |
us-gaap_OrganizationConsolidationAndPresentationOfFinancialStatementsAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe cumulative amount of the reporting entity's undistributed earnings or deficit. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.31(a)(3))
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 04
-Article 3
| Name: |
us-gaap_RetainedEarningsAccumulatedDeficit |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
v3.3.1.900
Stockholders' Equity (Deficit) (Details Narrative) - USD ($)
|
|
1 Months Ended |
12 Months Ended |
Aug. 11, 2014 |
Sep. 30, 2014 |
Aug. 31, 2015 |
Aug. 31, 2014 |
| Equity [Abstract] |
|
|
|
|
| Number of common stock issued during period |
|
10,000
|
|
|
| Issued shares par value |
|
$ 0.001
|
$ 0.001
|
$ 0.001
|
| Number of shares issued for services |
|
|
3,898,100
|
4,979,940
|
| Shares issued for services, value |
|
|
$ 1,553,038
|
$ 1,244,985
|
| Number of shares sold during period |
47,000,000
|
|
|
676,000
|
| Sold stock per share value |
|
|
|
$ 0.001
|
| Number of shares sold, value |
|
|
|
$ 184,500
|
| Shares issued for cash, value |
|
$ 10,000
|
|
|
| X |
- References
+ Details
| Name: |
us-gaap_EquityAbstract |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCash received on stock transaction after deduction of issuance costs.
| Name: |
us-gaap_SaleOfStockConsiderationReceivedOnTransaction |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of shares issued or sold by the subsidiary or equity method investee per stock transaction.
| Name: |
us-gaap_SaleOfStockNumberOfSharesIssuedInTransaction |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionPer share amount received by subsidiary or equity investee for each share of common stock issued or sold in the stock transaction.
| Name: |
us-gaap_SaleOfStockPricePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
num:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionPer share or per unit amount of equity securities issued.
| Name: |
us-gaap_SharesIssuedPricePerShare |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
num:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of shares issued in lieu of cash for services contributed to the entity. Number of shares includes, but is not limited to, shares issued for services contributed by vendors and founders.
| Name: |
us-gaap_StockIssuedDuringPeriodSharesIssuedForServices |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of new stock issued during the period. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21463-112644
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-URI http://asc.fasb.org/extlink&oid=27012166&loc=d3e187085-122770
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 29, 30
-Article 5
| Name: |
us-gaap_StockIssuedDuringPeriodSharesNewIssues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionValue of stock issued in lieu of cash for services contributed to the entity. Value of the stock issued includes, but is not limited to, services contributed by vendors and founders.
| Name: |
us-gaap_StockIssuedDuringPeriodValueIssuedForServices |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionEquity impact of the value of new stock issued during the period. Includes shares issued in an initial public offering or a secondary public offering. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21463-112644
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-URI http://asc.fasb.org/extlink&oid=27012166&loc=d3e187085-122770
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 29, 30, 31
-Article 5
| Name: |
us-gaap_StockIssuedDuringPeriodValueNewIssues |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
v3.3.1.900
| X |
- DefinitionMaintenance costs incurred and directly related to services rendered by an entity during the reporting period. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 225
-SubTopic 10
-Section S99
-Paragraph 2
-Subparagraph (SX 210.5-03.2(d))
-URI http://asc.fasb.org/extlink&oid=26872669&loc=d3e20235-122688
| Name: |
us-gaap_CostOfServicesMaintenanceCosts |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash inflow from the additional capital contribution to the entity. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Glossary Financing Activities
-URI http://asc.fasb.org/extlink&oid=6513228
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 230
-SubTopic 10
-Section 45
-Paragraph 14
-Subparagraph (a)
-URI http://asc.fasb.org/extlink&oid=56944662&loc=d3e3255-108585
| Name: |
us-gaap_ProceedsFromIssuanceOfCommonStock |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
v3.3.1.900
| X |
- DefinitionPercentage of valuation allowance.
| Name: |
PTLF_PercentageOfValuationAllowance |
| Namespace Prefix: |
PTLF_ |
| Data Type: |
num:percentItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionAccumulated change in equity from transactions and other events and circumstances from non-owner sources, net of tax effect, at period end. Excludes Net Income (Loss), and accumulated changes in equity from transactions resulting from investments by owners and distributions to owners. Includes foreign currency translation items, certain pension adjustments, unrealized gains and losses on certain investments in debt and equity securities, other than temporary impairment (OTTI) losses related to factors other than credit losses on available-for-sale and held-to-maturity debt securities that an entity does not intend to sell and it is not more likely than not that the entity will be required to sell before recovery of the amortized cost basis, as well as changes in the fair value of derivatives related to the effective portion of a designated cash flow hedge. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 220
-SubTopic 10
-Section 45
-Paragraph 11
-URI http://asc.fasb.org/extlink&oid=36458714&loc=d3e637-108580
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 220
-SubTopic 10
-Section 45
-Paragraph 14
-URI http://asc.fasb.org/extlink&oid=36458714&loc=d3e681-108580
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 220
-SubTopic 10
-Section 45
-Paragraph 14A
-URI http://asc.fasb.org/extlink&oid=36458714&loc=SL7669686-108580
Reference 4: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 31
-Article 5
Reference 5: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 04
-Article 3
| Name: |
us-gaap_AccumulatedOtherComprehensiveIncomeLossNetOfTax |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
v3.3.1.900
Share Exchange Agreement (Details Narrative) - shares
|
|
|
12 Months Ended |
|
Aug. 11, 2014 |
Apr. 18, 2014 |
Aug. 31, 2014 |
Aug. 31, 2015 |
| Share Exchange Agreement |
|
|
|
|
| Shares exchanged for approximately fully paid non-assessable shaes approximately |
|
47,000,000
|
|
|
| Percentage of issued and outstanding shares for acquisition of assets and liabilities |
|
80.00%
|
|
|
| Common stock, shares authorized |
|
|
750,000,000
|
750,000,000
|
| Number of shares sold during period |
47,000,000
|
|
676,000
|
|
| X |
- DefinitionPercentage of issued and outstanding shares for acquisition of assets and liabilities.
| Name: |
PTLF_PercentageOfIssuedAndOutstandingSharesForAcquisitionOfAssetsAndLiabilities |
| Namespace Prefix: |
PTLF_ |
| Data Type: |
num:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
PTLF_ShareExchangeAgreementAbstract |
| Namespace Prefix: |
PTLF_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe maximum number of common shares permitted to be issued by an entity's charter and bylaws. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 30
-Article 5
| Name: |
us-gaap_CommonStockSharesAuthorized |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe number of shares issued or sold by the subsidiary or equity method investee per stock transaction.
| Name: |
us-gaap_SaleOfStockNumberOfSharesIssuedInTransaction |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of shares of stock issued during the period pursuant to acquisitions. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-URI http://asc.fasb.org/extlink&oid=27012166&loc=d3e187085-122770
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.28,29)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 29, 30
-Article 5
| Name: |
us-gaap_StockIssuedDuringPeriodSharesAcquisitions |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.3.1.900
Subsequent Events (Details Narrative) - Subsequent Event [Member] - USD ($)
|
Nov. 21, 2015 |
Nov. 20, 2015 |
| Alexian Scientific, Inc. [Member] |
|
|
| Minimum obtaining fund |
$ 10,000,000
|
|
| Number of issue of shares of common stock |
38,777,630
|
|
| Percentage of equity of designees |
100.00%
|
|
| Number of common stock shares outstanding upon the cancellation |
80,786,730
|
21,583,000
|
| Imvestors [Member] |
|
|
| Number of shares issued during period |
|
626,000
|
| Officers And Employees [Member] |
|
|
| Number of shares issued during period |
|
530,200
|
| X |
- Definition
+ References
+ Details
| Name: |
PTLF_MinimumObtainingFund |
| Namespace Prefix: |
PTLF_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAggregate number of common shares reserved for future issuance. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 210
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.5-02.29)
-URI http://asc.fasb.org/extlink&oid=6877327&loc=d3e13212-122682
| Name: |
us-gaap_CommonStockCapitalSharesReservedForFutureIssuance |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe percentage of ownership of common stock or equity participation in the investee accounted for under the equity method of accounting. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 323
-SubTopic 10
-Section 50
-Paragraph 3
-Subparagraph (a)(1)
-URI http://asc.fasb.org/extlink&oid=6382943&loc=d3e33918-111571
| Name: |
us-gaap_EquityMethodInvestmentOwnershipPercentage |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
num:percentItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionNumber of shares (or other type of equity) issued during the period as a result of any equity-based compensation plan other than an employee stock ownership plan (ESOP), net of any shares forfeited. Shares issued could result from the issuance of restricted stock, the exercise of stock options, stock issued under employee stock purchase plans, and/or other employee benefit plans. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21463-112644
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section S99
-Paragraph 1
-Subparagraph (SX 210.3-04)
-URI http://asc.fasb.org/extlink&oid=27012166&loc=d3e187085-122770
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 29, 30
-Article 5
| Name: |
us-gaap_StockIssuedDuringPeriodSharesShareBasedCompensation |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionNumber of shares that have been repurchased during the period and have not been retired and are not held in treasury. Some state laws may govern the circumstances under which an entity may acquire its own stock and prescribe the accounting treatment therefore. This element is used when state law does not recognize treasury stock. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher FASB
-Name Accounting Standards Codification
-Topic 505
-SubTopic 10
-Section 50
-Paragraph 2
-URI http://asc.fasb.org/extlink&oid=6928386&loc=d3e21463-112644
Reference 2: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 04
-Article 3
Reference 3: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Regulation S-X (SX)
-Number 210
-Section 02
-Paragraph 29, 30
-Article 5
| Name: |
us-gaap_StockRepurchasedDuringPeriodShares |
| Namespace Prefix: |
us-gaap_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_SubsequentEventTypeAxis=us-gaap_SubsequentEventMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
Petlife Pharmaceuticals (CE) (USOTC:PTLF)
Historical Stock Chart
From Sep 2024 to Oct 2024
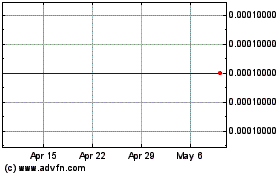
Petlife Pharmaceuticals (CE) (USOTC:PTLF)
Historical Stock Chart
From Oct 2023 to Oct 2024