
Immune Therapeutics, Inc. Files New
Drug Application for Lodonol in
Kenya to Target HIV Epidemic
Kenya's Pharmacy and Poison Board
Accepts Application for Full Review Toward Sales in Q3 and
Q4
Orlando, FL -- April
25,
2018 -- InvestorsHub NewsWire -- Immune Therapeutics,
Inc. (OTCQB:IMUN)
("Immune" "IMUN" or the "Company"), a clinical late stage
biopharmaceutical company focused on the development of therapies
for the treatment of autoimmune diseases, inflammatory diseases,
cancer and HIV/AIDS, is pleased to announce that
the Kenya
Pharmacy and Poison Board filed IMUN's New Drug Application (NDA)
for Lodonal, its lead drug candidate,
thereby accepting the NDA for a full review.
Lodonal is intended to be
used as
an adjunct to the treatment of HIV and adjunct to chemotherapy and
immune dysfunction.
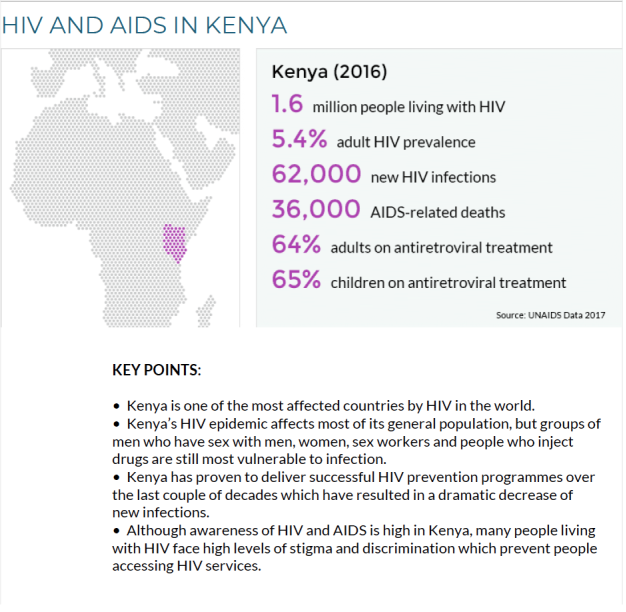
Kenya is
experiencing an HIV epidemic and currently has a reported 1.6
million people affected by HIV, many of which face high levels of
discrimination
"This is a
very serious issue in Kenya and other parts of East Africa," states
CEO Noreen Griffin. "Everyone deserves access to compassionate and
effective care and Immune Therapeutics is confident that our safe
and non-addictive applications of Lodonal will have a significant
impact on the people and communities affected by the HIV
epidemic.
Lodonal is a revolutionary
patented
formula
and
works by
boosting levels of endorphins (peptides produced in the brain and
adrenal glands) that are best known for relieving pain and
enhancing your sense of well-being. Endorphins are natural peptides
that have
proven to be powerful modulators of the
immune system. Low-dose naltrexone
(Lodonal or LDN) is a safe,
inexpensive, yet underused drug that is extremely beneficial for
people with conditions marked by immune system dysfunction such as
HIV/AIDS, among other
applications.
Immune
Therapeutics filed the application using the International Counsel
for Harmonization Guidelines and Application using the white paper
that was completed earlier this year
"I am pleased to
note
that the
acceptance of
filing of our NDA for Lodonal represents a key
achievement toward commercialization and impactful
distribution of our lead drug candidate.
We intend to work closely with Pharmacy and Poison
Board as
it reviews the NDA," states Griffin.
"We look
forward to Pharmacy and poison Board rendering a decision
on approval for marketing of Lodonal during
Q3 and Q4
2018."
ABOUT IMMUNE THERAPEUTICS, INC.
Immune Therapeutics
Inc. a
specialty pharmaceutical company, committed to the
commercialization of our patented therapies focused on the
activation and re-balancing of the body's immune system.
Stimulating
the body's immune system remains one of the most promising
approaches in the treatment of Cancers, HIV, Autoimmune Diseases,
inflammatory conditions and other chronic infectious
diseases.
Forward-Looking
Statements
This release
may contain forward-looking statements. Actual results may differ
from those projected due to a number of risks and uncertainties,
including, but not limited to, the possibility that some or all the
matters and transactions considered by the Company may not proceed
as contemplated, and by all other matters specified in the
Company's filings with the Securities and Exchange Commission.
These statements are made based upon current expectations that are
subject to risk and uncertainty. The Company does not undertake to
update forward-looking statements in this news release to reflect
actual results, changes in assumptions or changes in other factors
affecting such forward-looking information. Assumptions and other
information that could cause results to differ from those set forth
in the forward-looking information can be found in the Company's
filings with the Securities and Exchange Commission (www.sec.gov),
including its recent periodic reports.
$IMUN
CONTACT:
888-613-8802
http://www.immunetherapeutics.com/