- New data demonstrate duration of efficacy up
to 26-weeks and favorable safety profile with up to 5 injection
cycles for Cervical Dystonia -
Revance Therapeutics, Inc. (RVNC) today announced that new data
on DAXXIFY® will be presented at the annual meeting of Association
of Academic Physiatrists (AAP), taking place in Anaheim,
California, February 21-24, 2023. The supplemental biologics
license agreement for DAXXIFY® for the treatment of cervical
dystonia is currently under regulatory review with the Food and
Drug Administration.
A poster by Dr. Cynthia Comella and colleagues reports on 382
patients receiving 1,240 DAXXIFY® treatments across up to five
injection cycles over an 88-week time span, based on the masked,
randomized ASPEN-1 pivotal trial and subsequent ASPEN Open Label
Study (OLS). The findings reinforce the extended duration of
efficacy and favorable safety profile of DAXXIFY®. Notably the
duration of effect, as measured by an >80% loss of peak Toronto Western Spasmodic
Torticollis Rating Scale (TWSTRS) benefit, was maintained for
approximately 20 to 26 weeks across the evaluable injection cycles.
Dysphagia rates (difficulty swallowing) were low (2.7% for ASPEN-1
and 4.2% for ASPEN-OLS), further supporting DAXXIFY’s safety
profile.
A second poster, by Dr. Peter McAllister and colleagues, focuses
on the 357 patients in the ASPEN OLS study in which physicians
began dosing at either 125 or 250 U and were then permitted to
refine dosing based on the clinical response. Symptoms continued to
improve with successive DAXXIFY® treatments. The average dose at
the last visit was 243 U, with 37% of patients receiving up to 300
U during the study. The rate of adverse events remained low with
repeated dosing and at increasing dosing levels.
“These analyses demonstrate that DAXXIFY’s extended duration of
effect is sustained across multiple injection cycles.” said David
A. Hollander, MD, MBA, Chief Medical Officer. “The low rate of
adverse events with repeated treatments and as doses were increased
on an individual patient basis lends even further support that
DAXXIFY® can offer a new and differentiated treatment option for
patients with cervical dystonia.”
Poster Details:
Title: Efficacy of DaxibotulinumtoxinA for Injection Over
Successive Treatments in Adults with Isolated Cervical Dystonia in
the Phase 3 ASPEN-1 and ASPEN-OLS Trials
Authors and Affiliation: Cynthia Comella, Richard
Barbano, Alberto Vasquez, Todd Gross, Roman Rubio, Kristie Kooken,
Domenico Vitarella; Rush University Medical Center, New
Philadelphia, Ohio, USA; University of Rochester, Rochester, New
York, USA; Suncoast Neuroscience Associates, St. Petersburg,
Florida, USA; Revance Therapeutics, Inc., Nashville, Tennessee,
USA; Blue Obsidian Consulting, LLC, Redwood City, California, USA;
Revance Therapeutics, Inc, Nashville, Tennessee, USA
The above poster is available on the conference website. Please
visit www.physiatry.org for the latest information.
Title: A Phase 3, Open-Label, Multi-Center Trial to
Evaluate the Long-Term Safety and Efficacy of Repeat Treatments of
DaxibotulinumtoxinA for Injection in Adults with Isolated Cervical
Dystonia
Authors and Affiliation: Peter McAllister, Jaroslaw
Slawek, Sebastian Paus, Daniel Truong, Todd M. Gross, Roman G.
Rubio, Pat Kesslak, Domenico Vitarella; New England Institute for
Neurology and Headache, Stamford, Connecticut, USA; Medical
University of Gdańsk and St. Adalbert Hospital, Gdansk, Pomorskie,
Poland; University of Bonn and GFO Kliniken Troisdorf, Bonn,
Rheinland-Pfalz, Germany; University of California and the
Parkinson’s & Movement Disorder Institute, Riverside,
California, USA; Blue Obsidian Consulting, LLC, Redwood City,
California, USA; Revance Therapeutics, Inc., Nashville, Tennessee,
USA
The above poster is available on the conference website. Please
visit www.physiatry.org for the latest information.
INDICATION
DAXXIFY® (daxibotulinumtoxinA-lanm) for injection is an
acetylcholine release inhibitor and neuromuscular blocking agent
indicated for the temporary improvement in the appearance of
moderate to severe glabellar lines associated with corrugator
and/or procerus muscle activity in adult patients.
WARNING: DISTANT SPREAD OF TOXIN EFFECT
The effects of DAXXIFY® and all botulinum toxin products may
spread from the area of injection to produce symptoms consistent
with botulinum toxin effects. These symptoms have been reported
hours to weeks after injection. Swallowing and breathing
difficulties can be life threatening and there have been reports of
death. DAXXIFY® is not approved for the treatment of spasticity or
any conditions other than glabellar lines.
IMPORTANT SAFETY INFORMATION
Contraindications
DAXXIFY® contraindications include hypersensitivity to any
botulinum toxin preparation or any of the components in the
formulation and infection at the injection site(s).
Warnings and Precautions
Please refer to Boxed Warning for Distant Spread of Toxin
Effect.
The potency units of DAXXIFY® are not interchangeable with other
preparations of other botulinum toxin products. Recommended dose
and frequency of administration should not be exceeded. Patients
should seek immediate medical attention if respiratory, speech or
swallowing difficulties occur. Use caution when administering to
patients with pre-existing cardiovascular disease. Concomitant
neuromuscular disorders may exacerbate clinical effects of
treatment.
Adverse Reactions
The most commonly observed adverse reactions (≥1%) were headache
(6%), eyelid ptosis (2%) and facial paresis (1%).
Drug Interactions
Co-administration of DAXXIFY® and aminoglycoside antibiotics,
anticholinergic agents or any other agents interfering with
neuromuscular transmission or muscle relaxants should only be
performed with caution as the effect of DAXXIFY® may be
potentiated. The effect of administering different botulinum
neurotoxins during course of treatment with DAXXIFY® is
unknown.
Use in Specific Populations
DAXXIFY® is not recommended for use in children or pregnant
women.
Please see DAXXIFY® full Prescribing Information, including
Boxed Warning and Medication Guide.
About DAXXIFY®
DAXXIFY® (DaxibotulinumtoxinA-lanm) for injection is the first
and only FDA approved long-lasting peptide-formulated
neuromodulator product for use in adults for the temporary
improvement of moderate to severe frown lines (glabellar lines).
1-7 DAXXIFY® has the ability to deliver year-long results for
patients with potentially only two treatments per year and has been
proven to be effective, and generally safe and well
tolerated.2,8-10* DAXXIFY® is powered by a cell-penetrating peptide
technology (Peptide Exchange Technology™), Revance's proprietary,
synthetic, 35-amino-acid stabilizing excipient with a highly
positive charge, and is free of human serum albumin or animal-based
components. 1,2,7 Manufactured exclusively in the U.S., DAXXIFY® is
the first true innovation in neuromodulator product formulation in
over 30 years. Learn more at DAXXIFY.com.
Revance has evaluated this neuromodulator formulation in other
Phase 2 clinical studies in aesthetics, including the full upper
face, forehead lines and crow’s feet as well as in therapeutic
indications, including cervical dystonia and upper limb spasticity.
Learn more at RevanceAesthetics.com.
* At least 50% of patients treated with DAXXIFY® in SAKURA 1 and
2 had none or mild frown lines per investigator or patient
assessments for 24 weeks and 23.9 weeks (6 months) or longer
(respectively) after treatment.
Please see DAXXIFY® important safety information below and full
Prescribing Information, including Boxed Warning and Medication
Guide.
About Cervical Dystonia
According to the Dystonia Medical Research Foundation, cervical
dystonia is a painful condition in which the neck muscles contract
involuntarily, causing abnormal movements and awkward posture of
the head and neck. The movements may be sustained (tonic), jerky
(clonic), or a combination. Cervical dystonia (also referred to as
spasmodic torticollis) may be primary (meaning that it is the only
apparent neurological disorder, with or without a family history)
or may be the result of secondary causes (such as physical
trauma).
First-line treatment for cervical dystonia is usually
neuromodulator (botulinum toxin) injections, but additional
treatments can include oral medications, surgery, and complementary
therapies. Neuromodulators block the communication between the
nerve and the muscle, relaxing the muscle, which alleviates
abnormal involuntary movements and postures. Current neuromodulator
treatments for cervical dystonia have a duration of effect of
approximately three months. Cervical dystonia can occur at any age,
although most individuals first experience symptoms in middle age.
The condition affects a few hundred thousand adults and children in
the United States alone. The U.S. and global market opportunities
for cervical dystonia are approximately $325 million and $438
million, respectively as of 2021.2 The U.S. and global market
opportunities for treating muscle movement disorders with botulinum
toxins, which include cervical dystonia and spasticity (upper and
lower limb), are approximately $929 million and $1.2 billion,
respectively as of 2022.2
About Revance
Revance is a biotechnology company setting the new standard in
healthcare with innovative aesthetic and therapeutic offerings that
elevate patient and physician experiences. Revance’s aesthetics
portfolio of expertly created products and services, including
DAXXIFY® (DaxibotulinumtoxinA-lanm) for injection, the RHA®
Collection of dermal fillers, and OPUL®, the first-of-its-kind
Relational Commerce platform for aesthetic practices, deliver a
differentiated and exclusive offering for the company’s elite
practice partners and their consumers. Revance has also partnered
with Viatris Inc. to develop a biosimilar to onabotulinumtoxinA for
injection, which will compete in the existing short-acting
neuromodulator marketplace. Revance’s therapeutics pipeline is
currently focused on muscle movement disorders including evaluating
DAXXIFY® in two debilitating conditions, cervical dystonia and
upper limb spasticity.
Revance is headquartered in Nashville, Tennessee, with
additional office locations in Newark, Pleasanton and Irvine,
California. Learn more at www.Revance.com,
www.RevanceAesthetics.com, www.DAXXIFY.com or connect with us on
LinkedIn.
“Revance” and the Revance logo, DAXXIFY®, and OPUL® are
registered trademarks of Revance Therapeutics, Inc.
Resilient Hyaluronic Acid® and RHA® are trademarks of TEOXANE
SA.
Forward-Looking Statements
Any statements in this press release that are not statements of
historical fact, including statements related to the approval of
DAXXIFY® for the treatment of cervical dystonia; our market
opportunity; the potential benefits, safety, efficacy and duration
of DAXXIFY®; our opportunity in therapeutics; the potential to set
a new standard of care; development of a biosimilar to BOTOX® with
our partner, Viatris; statements about our business strategy,
timeline and other goals, plans and prospects; and the outcomes for
and experiences of patients; constitute forward-looking statements
within the meaning of the Private Securities Litigation Reform Act
of 1995, Section 27A of the Securities Act of 1933, as amended, and
Section 21E of the Securities Exchange Act of 1934, as amended. You
should not rely upon forward-looking statements as predictions of
future events. Although we believe that the expectations reflected
in the forward-looking statements are reasonable, we cannot
guarantee that the future results, levels of activity, performance,
events, circumstances or achievements reflected in the
forward-looking statements will ever be achieved or occur.
Forward-looking statements are subject to risks and
uncertainties that could cause actual results to differ materially
from our expectations. These risks and uncertainties relate, but
are not limited to: our ability to successfully commercialize
DAXXIFY® and to continue to successfully commercialize the RHA®
Collection of dermal fillers and OPUL®; the results, timing, costs,
and completion of our research and development activities and
regulatory approvals; our ability to obtain funding for our
operations; the timing of capital expenditures; the accuracy of our
estimates regarding future expenses, revenues and capital
requirements, our financial performance and the economics of
DAXXIFY®, the RHA® Collection of dermal fillers and OPUL®; the
impact of the COVID-19 pandemic and other macroeconomic factors on
our manufacturing operations, supply chain, end user demand for our
products and services, the aesthetics market, commercialization
efforts, business operations, regulatory meetings, inspections and
approvals, clinical trials and other aspects of our business and on
the market; our ability and the ability of our partners to
manufacture supplies for DAXXIFY® and our product candidates and to
acquire supplies of the RHA® Collection of dermal fillers; the
uncertain clinical development process; the risk that clinical
trials may not have an effective design or generate positive
results or that positive results would assure regulatory approval
or commercial success; the applicability of clinical study results
to actual outcomes; the rate and degree of economic benefit,
safety, efficacy, commercial acceptance, market, competition and/or
size and growth potential of DAXXIFY®, the RHA® Collection of
dermal fillers, and our drug product candidates, if approved; the
rate and degree of commercial acceptance, market, competition and
growth potential of OPUL® ; reports of adverse events or safety
concerns involving DAXXIFY® or the RHA® Collection of dermal
fillers; the timing and cost of commercialization activities; the
proper training and administration of our products by physicians
and medical staff; our ability to expand sales and marketing
capabilities; the status of commercial collaborations; changes in
and failures to comply with privacy and data protection laws; our
ability to effectively manage our expanded operations in connection
with the acquisition of Hint, Inc; our ability to continue
obtaining and maintaining intellectual property protection for
DAXXIFY® and our drug product candidates; the cost and our ability
to defend ourselves in product liability, intellectual property,
class action or other lawsuits; the volatility of our stock price;
and other risks. Detailed information regarding factors that may
cause actual results to differ materially from the results
expressed or implied by statements in this press release may be
found in our periodic filings with the Securities and Exchange
Commission (SEC), including factors described in the section
entitled "Risks Factors" on our Form 10-K filed with the SEC on
February 28, 2022 and including, without limitation, our Form 10-Q
for the quarter ended September 30, 2022, filed with the SEC on
November 8, 2022. The forward-looking statements in this press
release speak only as of the date hereof. We disclaim any
obligation to update these forward-looking statements
SOURCES
- Data on File. Draft DAXXIFY® Package Insert. Newark, CA:
Revance Therapeutics, Inc, 2022.
- Decision Resources Group Therapeutic Botulinum Toxin Market
Analysis Global 2021.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20230223005449/en/
Investors Revance Therapeutics, Inc.: Jessica Serra,
510-279-6886 jessica.serra@revance.com
or Gilmartin Group, LLC.: Laurence Watts, 619-916-7620
laurence@gilmartinir.com
Media Revance Therapeutics, Inc.: Sara J. Fahy,
949-887-4476 sfahy@revance.com
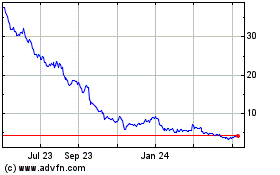
Revance Therapeutics (NASDAQ:RVNC)
Historical Stock Chart
From Jun 2024 to Jul 2024
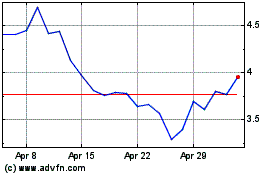
Revance Therapeutics (NASDAQ:RVNC)
Historical Stock Chart
From Jul 2023 to Jul 2024