Filed Pursuant to Rule 424(b)(5)
Registration File No. 333-265492
The information in this preliminary
prospectus supplement is not complete and may be changed. This preliminary prospectus supplement and the accompanying prospectus are
part of an effective registration statement filed with the Securities and Exchange Commission. This preliminary prospectus supplement
and the accompanying prospectus are not an offer to sell these securities, and we are not soliciting an offer to buy these securities,
in any jurisdiction where the offer or sale is not permitted.
Subject to completion, dated February 26,
2024
Preliminary
Prospectus Supplement
(To Prospectus dated June 21, 2022)

Shares of Common Stock
Pre-Funded Warrants to Purchase Shares of
Common Stock
We are offering shares
of our common stock, par value, $0.001 per share, and pre-funded warrants to purchase (the “Pre-Funded Warrants”) to purchase
up to shares of common stock (and the shares of common stock issuable from time to time upon exercise of the Pre-Funded Warrants)
in this offering. The public offering price of each share of common stock is $ and
the public offering price of each Pre-Funded Warrant to purchase one share of common stock is $ .
We are offering the Pre-Funded Warrants to purchase
shares of common stock (and the shares of common stock issuable from time to time upon exercise of the Pre-Funded Warrants), in lieu of
shares of common stock, to investors whose purchase of shares of common stock in this offering would otherwise result in any such investor,
together with its affiliates, beneficially owning more than 4.99% (or, at the election of such investor, 9.99%) of our outstanding shares
of common stock immediately following the consummation of this offering, in lieu of shares of common stock that would otherwise result
in such investor’s beneficial ownership exceeding 4.99% (or, at the election of such investor, 9.99%) of our outstanding shares
of common stock. Each Pre-Funded Warrant will be exercisable for one share of common stock at an exercise price of $0.001 per share of
common stock, will be exercisable immediately and will expire when exercised in full. The offering price per Pre-Funded Warrant is equal
to the offering price per share of common stock, less $0.001. We are also offering the shares of common stock that are issuable from time
to time upon exercise of the Pre-Funded Warrants.
There is no established public trading market
for the Pre-Funded Warrants, and we do not expect a market to develop. In addition, we do not intend to list the Pre-Funded Warrants,
nor do we expect the Pre-Funded Warrants to be quoted, on the Nasdaq Capital Market (“Nasdaq”) or any other national securities
exchange or any other nationally recognized trading system. Without an active trading market, the liquidity of the Pre-Funded Warrants
will be limited.
Our common stock and our public warrants (the
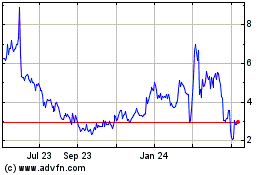
“Public Warrants”) are listed on Nasdaq under the symbols “NRXP” and “NRXPW”, respectively. On February 23,
2024, the closing price of our common stock was $0.44 and the closing price of our Public Warrants was $0.1101.
As of the date of this prospectus supplement, the aggregate market value
of our common stock held by our non-affiliates, as calculated pursuant to the rules of the Securities and Exchange Commission (the
“SEC”), was $34,842,686, based upon 68,052,122 shares of our outstanding common stock held by non-affiliates at the per share
price of $0.512, the closing sale price of our common stock on Nasdaq on February 9, 2024. Pursuant to General Instruction I.B.6 of Form S-3,
in no event will we sell securities in a public offering with a value exceeding more than one-third of our “public float”
(i.e., the market value of our common stock held by our non-affiliates) in any 12-month period so long as our public float remains below
$75.0 million. We have offered $8,285,501 of securities pursuant to General Instruction I.B.6 of Form S-3 during the 12 calendar
months prior to and including the date of this prospectus supplement.
Investing in our securities involves a high
degree of risk. You should read this prospectus supplement and the accompanying prospectus carefully before you make your investment decision.
See “Risk Factors” beginning on page S-5 of this prospectus supplement, page 10 of the accompanying prospectus,
and the other documents we file or have filed with the SEC that are incorporated by reference in this prospectus supplement and in the
accompanying prospectus, for a discussion of the factors you should consider before investing in our securities.
Neither the SEC nor any state securities commission
has approved or disapproved of these securities or determined if this prospectus is truthful or complete. Any representation to the contrary
is a criminal offense.
| |
|
Per Share |
|
Per Pre-
Funded
Warrant |
|
Total |
|
| Public offering price |
|
$ |
|
|
$ |
|
$ |
|
|
| Underwriting discounts and commissions(1) |
|
$ |
|
|
$ |
|
$ |
|
|
| Proceeds, before expenses, to us |
|
$ |
|
|
$ |
|
$ |
|
|
(1) Does
not include a non-accountable expense allowance of 1.0% of the gross proceeds of this offering or other expenses payable by the Company
to the underwriter in connection with this offering. In addition, we have agreed to issue the representative of the underwriters, or its
designees, warrants to purchase a number of shares of common stock equal to 5.0% of the aggregate number of shares of common stock sold
in this offering with an assumed exercise price of $ per share, or 110% of the public offering price per share (the “underwriter's
warrants”). See “Underwriting” on page S-10 for additional disclosure regarding underwriting dicsounts, commissions
and expenses.
We have granted the representative of the underwriters a 45-day option
to purchase up to additional shares of common stock and/or Pre-Funded Warrants from us at the same terms and conditions set forth
above.
We are a “smaller reporting company”
under the federal securities laws and, as such, we have elected to comply with certain reduced public company reporting requirements and
scaled disclosures for this prospectus and future filings. See “Prospectus Supplement Summary—Implication of Being a Smaller
Reporting Company.”
Delivery of the shares of common stock and the
Pre-Funded Warrants offered pursuant to this prospectus supplement and accompanying prospectus is expected to be made on or about ,
2024, subject to satisfaction of customary closing conditions.
Sole Book-Running Manager

The date of this prospectus supplement is February ,
2024
TABLE OF CONTENTS
Prospectus
Supplement
PROSPECTUS
About
This Prospectus Supplement
This prospectus supplement relates to the offering
of our securities. Before buying the securities offered hereby, we urge you to read carefully this prospectus supplement, the accompanying
prospectus, and any free writing prospectus that we have authorized for use in connection with this offering, together with the information
incorporated by reference herein, as described under the heading “Incorporation of Certain Documents by Reference.” These
documents contain important information that you should consider when making your investment decision. This prospectus supplement contains
information about the securities offered hereby.
This document is in two parts. The first part
is this prospectus supplement, which describes the specific terms of the securities we are offering. The second part is the accompanying
prospectus, including the information incorporated by reference therein, which provides more general information, some of which may not
apply to this offering. This prospectus supplement and the information incorporated by reference in this prospectus supplement also may
add to, update and change information contained in, or incorporated by reference into, the accompanying prospectus. Generally, when we
refer to this prospectus, we are referring to both parts of this document combined. To the extent there is a conflict between (i) the
information contained in this prospectus supplement and (ii) the information contained in the accompanying prospectus or in any information
incorporated by reference that was filed with the SEC before the date of this prospectus supplement, you should rely on the information
in this prospectus supplement. If any statement in one of these documents is inconsistent with a statement in another document having
a later date, for example, information incorporated by reference in this prospectus supplement or the accompanying prospectus, the statement
in the document having the later date modifies or supersedes the earlier statement.
We further note that the representations, warranties
and covenants made by us in any agreement that is filed as an exhibit to the registration statement to which the accompanying prospectus
forms a part or to any document that is incorporated by reference in this prospectus supplement or the accompanying prospectus were made
solely for the benefit of the parties to such agreement, including, in some cases, for the purpose of allocating risk among the parties
to such agreements, and should not be deemed to be a representation, warranty or covenant to you. Moreover, such representations, warranties
or covenants were accurate only as of the date when made. Accordingly, such representations, warranties and covenants should not be relied
on as accurately representing the current state of our affairs.
The accompanying prospectus is part of a registration
statement that we filed with the SEC using a shelf registration process on Form S-3 (File No. 333-265492), which such registration
statement was originally filed on June 9, 2022, and declared effective by the SEC on June 21, 2022. Under the shelf registration
process, from time to time, we may offer and sell any of the securities described in the accompanying prospectus separately or together
with other securities described therein.
You should rely only on the information contained
in, or incorporated by reference into, this prospectus supplement, the accompanying prospectus, the information incorporated by reference
herein, and any related free writing prospectus that we authorized to be distributed to you. Neither we nor the underwriters have authorized
anyone to provide you with different or additional information. If anyone provides you with different or additional information, you should
not rely on it. Neither we nor the underwriters is making an offer to sell the securities in any jurisdiction where the offer or sale is
not permitted, and you should not consider this prospectus supplement or the accompanying prospectus to be an offer or solicitation relating
to the securities in any jurisdiction in which such an offer or solicitation relating to the securities is not authorized. You should
assume that the information contained in this prospectus supplement, the accompanying prospectus, any related free writing prospectus
that we have authorized to be delivered to you and the information incorporated by reference herein and therein is accurate only as of
their respective dates, regardless of the time of delivery of this prospectus supplement, the accompanying prospectus, and any related
free writing prospectus or of any sale of securities. Our business, financial condition, results of operations and prospects may have
changed since those dates. Furthermore, you should not consider this prospectus supplement or the accompanying prospectus to be an offer
or solicitation relating to the securities if the person making the offer or solicitation is not qualified to do so, or if it is unlawful
for you to receive such an offer or solicitation.
Unless otherwise indicated, information contained
in this prospectus supplement and the accompanying prospectus or the information incorporated by reference herein or therein concerning
our industry and the markets in which we operate, including our general expectations and market position, market opportunity and market
share, is based on information from our own management estimates and research, as well as from industry and general publications and
research, surveys and studies conducted by third parties. Management estimates are derived from publicly available information, our knowledge
of our industry and assumptions based on such information and knowledge, which we believe to be reasonable. In addition, assumptions
and estimates of our and our industry's future performance are necessarily subject to a high degree of uncertainty and risk due to a
variety of factors, including those described in the section entitled “Risk Factors” in this prospectus supplement and the
accompanying prospectus, and in our Annual
Report on Form 10-K for the fiscal year ended December 31, 2022, as filed with the SEC on May 1, 2023, and in our
Quarterly Reports on Form 10-Q for the quarters ended March 31, 2023, June 30, 2023, and September 30, 2023, filed
with the SEC on May 15,
2023, August 14,
2023, and November 14,
2023, respectively, which are incorporated by reference into this prospectus supplement. These and other important factors could
cause our future performance to differ materially from our assumptions and estimates. See “Cautionary Note Regarding Forward-Looking
Statements.”
This prospectus supplement, the accompanying
prospectus, and the information incorporated herein and therein by reference includes trademarks, service marks and trade names owned
by us or other companies. All trademarks, service marks and trade names included or incorporated by reference into this prospectus supplement
or the accompanying prospectus are the property of their respective owners.
In this prospectus, the terms “NRx,”
“the Company,” “we,” “us” and “our” refer to NRx Pharmaceuticals, Inc.
We are offering to sell, and are seeking offers
to buy, the securities only in jurisdictions where such offers and sales are permitted. The distribution of this prospectus supplement
and the accompanying prospectus and the offering of the securities in certain jurisdictions may be restricted by law. Persons outside
the United States who come into possession of this prospectus supplement and the accompanying prospectus must inform themselves about
and observe any restrictions relating to the offering of the securities and the distribution of this prospectus supplement and the accompanying
prospectus outside the United States. This prospectus supplement and the accompanying prospectus do not constitute, and may not be used
in connection with, an offer to sell, or a solicitation of an offer to buy, any securities offered by this prospectus supplement and
the accompanying prospectus to or by any person in any jurisdiction in which it is unlawful for such person to make such an offer or
solicitation.
CAUTIONARY
NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus supplement and the information
incorporated by reference herein include “forward-looking statements” within the meaning of the “safe harbor”
provisions of the U.S. Private Securities Litigation Reform Act of 1995, which may include, but are not limited to, statements regarding
our financial outlook, product development, business prospects, and market and industry trends and conditions, as well as our strategies,
plans, objectives, and goals. These forward-looking statements are based on current beliefs, expectations, estimates, forecasts, and
projections of, as well as assumptions made by, and information currently available to, our management. Words such as “expect,”
“anticipate,” “should,” “believe,” “hope,” “target,” “project,”
“goals,” “estimate,” “potential,” “predict,” “may,” “will,” “might,”
“could,” “would,” “seek,” “plan,” “intend,” “shall,” and variations
of these terms or the negative of these terms and similar expressions are intended to identify these forward-looking statements.
These forward- looking statements are, by their
nature, subject to significant risks and uncertainties, many of which involve factors or circumstances that are beyond our control. These
risks and uncertainties include, but are not limited to, our relatively limited operating history; our ability to expand, retain and
motivate our employees and manage our growth; risks associated with general industry conditions and competition; general economic factors,
including interest rate and currency exchange rate fluctuations; the ongoing effects of the COVID-19 endemic; the impact of pharmaceutical
industry regulation and health care legislation in the United States and internationally; global trends toward health care cost containment;
technological advances, new products and patents attained by competitors; challenges inherent in new product development, including obtaining
regulatory approval; our ability to accurately predict future market conditions; our ability to regain and maintain compliance with Nasdaq’s
listing standards; maintain compliance with the terms of our indebtedness; manufacturing difficulties or delays; changes in laws, rules or
regulations relating to any aspect of our business operations, or general economic, market and business conditions; financial instability
of international economies and sovereign risk; dependence on the effectiveness of our patents and other protections for innovative products;
and the exposure to litigation, including patent litigation, and/or regulatory actions. Furthermore, there can be no guarantees with
respect to pipeline products that the products will receive the necessary regulatory approvals or that they will prove to be commercially
successful. If underlying assumptions prove inaccurate or risks or uncertainties materialize, actual results may differ materially from
those set forth in the forward-looking statements. We assume no obligation and does not intend to update or otherwise revise any forward-looking
statement, whether as a result of new information, future events or otherwise, except as required by applicable law. As a result of these
and other risks, uncertainties and assumptions, forward-looking events and circumstances discussed herein might not occur in the way
that tour management expects, if at all. Accordingly, you should not place reliance on any forward-looking statement, and all forward-looking
statements are herein qualified by reference to the cautionary statements set forth above.
You also should carefully review the risk factors
and cautionary statements described in the other documents we file or furnish from time to time with the SEC, including our Annual Reports
on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K. The
forward-looking statements included in this prospectus supplement, the accompanying prospectus and any other offering material, or in
the documents incorporated by reference into this prospectus supplement, the accompanying prospectus and any other offering material,
are made only as of the date of the prospectus supplement, the accompanying prospectus, any other offering material or the incorporated
document.
We do not assume any obligation to update any
forward-looking statements. We disclaim any intention or obligation to update or revise any forward-looking statement, whether as a result
of new information, future events or otherwise.
Prospectus
Supplement Summary
This summary does not contain all of the information
that you should consider before investing in our securities offered pursuant to this prospectus supplement and the accompanying prospectus.
Before making an investment decision, you should carefully read the entire prospectus supplement and the accompanying prospectus, including
the “Risk Factors” sections in this prospectus supplement and the accompanying prospectus and in the information incorporated
herein and therein by reference, as well as our financial statements, including the accompanying notes thereto, and the other information
incorporated by reference to this prospectus supplement and the accompanying prospectus, and the information in any related free writing
prospectus that we may authorize for use in connection with this offering of our securities.
Overview
NRx Pharmaceuticals
is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous
system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. The Company is developing NRX-101, an FDA-designated
investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. NRx has partnered with Alvogen
and Lotus around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has
potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
Our patent
estate contains broad disclosure of the synergistic combination of NMDA and 5-HT2A antagonist drugs in the treatment of mental health
disorders and chronic pain. Our foundation product, NRX-101 (D-cycloserine/lurasidone), is being studied initially for the treatment
of bipolar depression in patients with suicidality, has been awarded Fast Track designation, Breakthrough Therapy designation, a Special
Protocol Agreement (SPA), and a Biomarker Letter of Support by the U.S. Food and Drug Administration. To our knowledge, NRX-101 is the
only oral antidepressant demonstrated to reduce suicidal ideation in a phase 2 trial. NRX-101 is covered by four families of U.S. and
foreign patents, including a composition of matter patent (U.S. Patent No. 10,583,138 and foreign counterparts).
Nasdaq
Compliance
Minimum
Bid Price Requirement
On April 18,
2023, we received a written notification from the Listing Qualifications Staff (the “Staff”) of the Nasdaq Stock Market indicating
that we were not in compliance with Nasdaq Listing Rule 5450(a)(1), as closing bid price for our common stock was below $1.00 per
share for the last 30 consecutive business days. We were provided with an initial compliance period of 180 calendar days to regain compliance
with the Nasdaq Listing Rule 5450(a)(1), which expired on October 16, 2023. On October 17, 2023, we received a notice
from the Staff indicating that based upon our non-compliance with Nasdaq Listing Rule 5450(a)(1), our securities were subject to
delisting unless we timely requested a hearing before the Nasdaq Hearings Panel (the “Panel”). We timely requested a hearing
before the Panel, which was held on January 4, 2024.
On January 16,
2024, the Panel granted our request for an exception to the Nasdaq Listing Rules until April 16, 2024, to demonstrate compliance
with the Minimum Bid Price Requirement. Such exception is subject to the following conditions: (1) our filing of all necessary documentation
required to transfer our listing from the Nasdaq Global Market to the Nasdaq on or before January 19, 2024; and (2) our demonstrating
compliance with Nasdaq Listing Rule 5550(a)(2) (the “Minimum Bid Price Requirement”) on or before April 16,
2024. On February 1, 2024, the Nasdaq Stock Market informed us that it had approved our application to transfer our listing. Our
securities were transferred to Nasdaq at the opening of business on January 19, 2024.
Market
Value of Listed Securities Requirement
On July 20,
2023, we received a written notification from the Staff of the Nasdaq Stock Market indicating that we were not in compliance with Nasdaq
Listing Rule 5450(b)(2)(A), as we had not maintained a minimum market value of listed securities (“MVLS”) of $50,000,000
for the last 33 consecutive business days.
We were
provided with an initial compliance period of 180 calendar days, or until January 22, 2024, to regain compliance with the MVLS requirement.
To regain compliance, our MVLS was required to meet or exceed $50,000,000 for a minimum period of ten consecutive business days prior
to January 22, 2024. As explained above, on February 1, 2024, the Nasdaq Stock Market informed us that it had approved our
application to transfer our listing. Our securities were transferred to Nasdaq at the opening of business on January 19, 2024.
Recent Developments
Alvogen Partnership
On February 7, 2024, the Company entered into
the First Amendment (the “Amendment”) to the Exclusive, Global - Development, Supply, Marketing & License Agreement (as
amended, the “License Agreement”), effective as of the same date. Pursant to the term of the Amendment, the Company will receive
$5 million of the first milestone, which NRx will use to fund development of NRX-101 through the phase 2 meeting with Food and Drug Administration
(“FDA”). As compensation for advancing the milestone, Alvogen, Inc. (“Alvogen”) and Lotus Pharmaceutical Co. Ltd.
will receive warrants to purchase up to an aggregate of 4,195,978 shares of the Company's common stock, at a strike price of $0.40 with
a three year term. The second portion of the milestone will be $4 million and, as before, be triggered by a positive response to the Company's
planned end of phase 2 meeting with FDA.
Pursuant to the License Agreement, we remain
eligible to receive up to $320,000,000 in future development and sales milestones, as well as royalty payment escalating to mid-teen
percentages on Net Sales (as defined in the License Agreement), subject to the achievement of certain sales volumes. Additionally, Alvogen
will be responsible for future development and commercialization costs for NRX-101 in Suicidal Bipolar Depression.
Amendment to Convertible Promissory Note
On February 9, 2024, the Company entered into
Amendment #3 to Convertible Promissory Note (the “Third Amendment”), with Streeterville Capital, LLC (“Streeterville”
or the “Lender”). Pursuant to the Third Amendment, the Company and Streeterville agreed to further amend the terms of that
certain Convertible Promissory Note dated November 4, 2022, in the original principal amount of $11,020,000, as amended by the amendments
to the Convertible Promissory Note dated March 30, 2023 and July 7, 2023 (as amended, the “Note”). In accordance with the
Third Amendment, the Company and Streeterville agreed to amend the redemption provisions of the Note. In particular, the Company agreed
to pay Streeterville an amount in cash equal to $1,100,000 on February 12, 2024. In addition, beginning on or before February 29, 2024,
on or before the last day of each month until July 31, 2024, the Company shall pay Streeterville an amount equal to $400,000 in cash,
less any amount satisfied by the delivery of Redemption Conversion Shares (as defined in the Note).
Launch of HOPE Therapeutics, Inc.
On February 26, 2024, the Company launched HOPE
Therapeutics, Inc. (“HOPE Therapeutics”), a subsidiary of the Company, at the BIO CEO & Investor Conference 2024. The
Company’s management is proposing to award 50% of founding shares in HOPE Therapeutics to current shareholders together with a royalty
coupon with an expected ex-dividend date in the near future, all subject to board approval. The dividend is expected to be available to
all shareholders who sign a covenant not to engage in short sales of the Company’s stock, subject to board approval. HOPE Therapeutics
anticipates having manufactured ketamine supplies for shipment under 503a pharmacy regulations by July 1, 2024.
Implications of Being a Smaller Reporting Company
We are a “smaller reporting company”
as defined in Item 10(f)(1) of Regulation S-K. Smaller reporting companies may take advantage of certain reduced disclosure obligations,
including, among other things, providing only two years of audited financial statements. We will remain a smaller reporting company
until the last day of the fiscal year in which (i) the market value of our common stock held by non-affiliates exceeds $250 million
as of the prior June 30, or (ii) our annual revenue exceeded $100 million during such completed fiscal year and the market
value of our common stock held by non-affiliates exceeds $700 million as of the prior June 30.
Corporate Information
We were incorporated under the laws of the State
of Delaware on September 18, 2017 under the name Big Rock Partners Acquisition Corp. Upon the closing of the business combination
with NeuroRx, Inc. on May 24, 2021, we changed our name to NRx Pharmaceuticals, Inc. Our principal executive offices are
located at 1201 Orange Street, Suite 600, Wilmington, Delaware 19801, our mailing address is 1201 North Market Street, Suite 111,
Wilmington, DE 19801 and our telephone number is (484) 254-6134. Our website address is www.nrxpharma.com. The information contained
in, or accessible through, our website does not constitute a part of this prospectus supplement or the accompanying prospectus. We have
included our website address in this prospectus supplement solely as an inactive textual reference.
The
Offering
| Issuer |
NRx Pharmaceuticals, Inc. |
| |
|
| Common stock offered by us |
shares of our common stock. |
| |
|
|
Pre-Funded Warrants offered by us
|
Pre-Funded Warrants to
purchase up
to shares
of common stock to certain investors whose purchase of shares of common stock in this offering would otherwise result in any such
investor, together with such investor’s affiliates and certain related parties, beneficially owning more than 4.99% (or, at
the election of an investor, 9.99%) of our outstanding shares of common stock immediately following the consummation of this
offering. The purchase price of each Pre-Funded Warrant is equal to the price per share at which the shares of common stock are
being sold in this offering, minus $0.001. The exercise price of each Pre-Funded Warrant will equal $0.001 per share. Each
Pre-Funded Warrant will be exercisable upon issuance and will expire when exercised in full. This prospectus supplement and the
accompanying prospectus also relate to the offering of the shares of common stock issuable upon exercise of the Pre-Funded Warrants.
See “Description of Securities Offered” on page S-14 of this prospectus supplement. |
| |
|
| Public offering price |
$ per share of common stock and $ per Pre-Funded Warrant to purchase one share of common stock. |
| |
|
| Shares of common stock to be outstanding after this offering(1) |
shares ( shares if the representative of the underwriters exercises its option in full to purchase up to additional shares of common stock and/or Pre-Funded Warrants in full and assuming full exercise of any Pre-Funded Warrants issued in this offering). |
| |
|
| Underwriters’ option to purchase additional shares of common stock and/or Pre-Funded Warrants |
The representative of the underwriters has an
option, exercisable for 45 days after the date of this prospectus supplement, to purchase up to an additional shares of common stock and/or
Pre-Funded Warrants from us at the public offering prices, less underwriting discounts and commissions.
|
| |
|
| Lock-up agreements |
We and our executive officers and directors have agreed, that subject to certain exceptions, we and our directors and officers will not, until 90 days after the closing of this offering, offer, pledge, sell, contract to sell, grant, lend, or otherwise transfer or dispose of, directly or indirectly of any of our shares of common stock. |
| |
|
| Use of proceeds |
We intend to use the net proceeds from this offering for working capital and general corporate purposes. We may also use the proceeds from this offering to repay the Note issued to Streeterville. The Note bears interest at a rate of 9% per annum and matures in August 2024. See “Use of Proceeds” on page S-8. |
| |
|
| Risk factors |
An investment in our securities involves a high degree of risk. See “Risk Factors” beginning on page S-5 of this prospectus supplement and other information included in this prospectus supplement, the accompanying prospectus and the documents incorporated by reference in this prospectus supplement and the accompanying prospectus for a discussion of factors you should carefully consider before deciding to invest in our securities. |
| |
|
| Nasdaq Capital Market symbol |
Our common stock is traded on Nasdaq under the symbol “NRXP.” There is no established public trading market for the Pre-Funded Warrants and we do not expect a market to develop. We do not intend to apply to list the Pre-Funded Warrants on any national securities exchange or other nationally recognized trading system. Without an active market, the liquidity of the Pre-Funded Warrants will be limited. |
| |
|
(1) The number of shares of our
common stock outstanding after this offering is based on 86,675,580 shares of common stock outstanding as of February 26, 2024 and
excludes:
| |
· |
3,450,000 shares of common
stock issuable upon the exercise of the Public Warrants, at an exercise price of $11.50 per share; |
| |
· |
2,746,961 shares of common stock reserved for future issuance pursuant to outstanding option awards,
with a weighted average exercise price of $2.48 per share; |
| |
· |
2,312,462 shares of common stock available for grants under the NRx 2021 Omnibus Incentive
Plan (the “2021 Plan”); |
| |
· |
up to 45,000,000 shares
of common stock issuable upon conversion of the Note; |
| |
· |
3,000,000 shares of common stock issuable upon the conversion of 3,000,000 shares of Series A Convertible Preferred Stock
(the “Series A Preferred Stock”) at a conversion price of $0.40 per share; |
| |
|
|
| |
· |
3,000,000 shares of common stock issuable upon the exercise of warrants issued to certain investors pursuant to a securities
purchase agreement dated as of August 28, 2023 (the “August 2023 Purchase Agreement”), at an exercise price
of $0.40 per share; and |
| |
· |
4,195,978 shares of common stock issuable upon the exercise of the
Alvogen Warrants issued to Alvogen, at an exercise price of $0.40 per share. |
Except as otherwise indicated, all information
in this prospectus supplement assumes no exercise of the underwriters’ option to purchase up to
additional shares of common
stock and/or Pre-Funded Warrants, no exercise of the underwriter’s warrants, no sale of any Pre-Funded Warrants in this offering,
and no exercise or settlement of outstanding options, warrants or conversion of securities convertible into shares of common stock.
RISK FACTORS
An investment in our securities involves a high degree of risk.
Prior to making a decision about investing in our securities, you should consider carefully the specific risk factors discussed in the
sections entitled “Risk Factors” contained in our most recent Annual Report on Form 10-K for
the year ended December 31, 2022, as filed with the SEC on March 31,
2023, as amended on May 1,
2023, and May 26,
2023, which are incorporated into this prospectus supplement and the accompanying prospectus by reference in their entirety, as updated
or superseded by the risks and uncertainties described under similar headings in our Quarterly Reports on Form 10-Q, our Current
Reports on Form 8-K, and the other documents that are filed after the date hereof and incorporated by reference into this prospectus
supplement and the accompanying prospectus, together with other information in this prospectus supplement and the accompanying prospectus
and any free writing prospectus that we may authorize for use in connection with this offering. These risks and uncertainties are not
the only risks and uncertainties we face. Additional risks and uncertainties not presently known to us, or that we currently view as
immaterial, may also impair our business. Past financial performance may not be a reliable indicator of future performance, and historical
trends should not be unduly relied upon to anticipate results or trends in future periods. If any of the risks or uncertainties described
in our SEC filings or any additional risks and uncertainties actually occur, our business, financial condition, results of operations
and cash flow could be materially and adversely affected. In that case, the trading price of our common stock could decline and you might
lose all or part of your investment. Please also read carefully the section above titled “Cautionary Note Regarding Forward-Looking
Statements.”
Risks Related to our Common Stock and this Offering
If we fail to meet the applicable continued
listing requirements of Nasdaq, our common stock may be delisted, in which case the liquidity and market price of our common stock could
decline.
Our common stock is currently listed on Nasdaq.
In order to maintain that listing, we must satisfy certain continued listing requirements. In the past, we have received deficiency letters
from Nasdaq for failing to maintain compliance with such listing requirements. For example, on July 20, 2023, we received a written
notification from the Staff indicating that we were not in compliance with Nasdaq Listing Rule 5450(b)(2)(A) because we had
not maintained a minimum MVLS of $50,000,000 for the previous 33 consecutive business days. We were provided an initial compliance period
of 180 calendar days, or until January 22, 2024, to regain compliance with the minimum MVLS requirement. Additionally, on April 18,
2023, we received a written notification from the Staff indicating we were not in compliance with
Nasdaq Listing Rule 5450(a)(1), and were provided an initial compliance period of 180 calendar days, or until October 16, 2023,
to regain compliance. On October 17, 2023, we received a written notification from the Staff indicating that based upon our non-compliance
with Nasdaq Listing Rule 5450(a)(1), our securities were subject to delisting unless we timely requested a hearing before
the Panel, which such hearing was timely requested and subsequently held on January 4, 2024. On January 16, 2024, the Panel
granted our request for an exception to the Nasdaq Listing Rules until April 16, 2024, to demonstrate compliance with the Minimum
Bid Price Requirement, subject to our filing all necessary documentation required to transfer our listing from the Nasdaq Global Market
to Nasdaq on or before January 19, 2024, and our demonstrating compliance with the Minimum Bid Price Requirement on or before April 16,
2024. On February 1, 2024, the Nasdaq Stock Market informed us that it had approved our application to transfer our listing to Nasdaq.
Our securities were transferred from the Nasdaq Global Market to Nasdaq at the opening of business on January 19, 2024.
If our common stock is delisted, an active trading
market for our common stock may not be sustained and the market price of our common stock could decline. Delisting of our common stock
could adversely affect our ability to raise additional capital through the public or private sale of equity securities, would significantly
affect the ability of investors to trade our securities and would negatively affect the value and liquidity of our common stock. Delisting
could also have other negative results, including the potential loss of confidence by employees, the loss of institutional investor interest
and fewer business development opportunities.
Fluctuations in the price of our common
stock, including as a result of actual or anticipated sales of shares by us and/or our directors, officers or stockholders, may make
our common stock more difficult to resell.
The market price and trading volume of our common
stock have been, and may continue to be, subject to significant fluctuations due not only to general stock market conditions, but also
to changes in sentiment in the market regarding the industry in which we operate, our operations, business prospects or liquidity, or
this offering. In addition to the risk factors discussed in our periodic reports and in this prospectus supplement, the price and volume
volatility of our common stock may be affected by actual or anticipated sales of common stock by us and/or our directors, officers or
stockholders, whether in the market, in connection with business acquisitions, in this offering or in subsequent public offerings. Stock
markets in general have at times experienced extreme volatility unrelated to the operating performance of particular companies. These
broad market fluctuations may adversely affect the trading price of our common stock, regardless of our operating results.
As a result, these fluctuations in the market
price and trading volume of our common stock may make it difficult to predict the market price of our common stock in the future, cause
the value of your investment to decline and make it more difficult to resell our common stock.
Management will have broad discretion as
to the use of the net proceeds of this offering, and we may use the net proceeds in ways in which you and other stockholders may disagree.
We currently intend to use the net proceeds of
this offering as described in the section entitled “Use of Proceeds” on page S-8. However, our management will have
broad discretion over the use and investment of the net proceeds from this offering, and, accordingly, investors in this offering will
need to rely upon the judgment of our management with respect to the use of proceeds, with only limited information concerning our specific
intentions Our stockholders may not agree with the manner in which our management chooses to allocate and spend the net proceeds.
Investors in this offering will experience
immediate and substantial dilution in the net tangible book value per share of our common stock.
The public offering price of our common
stock will be substantially higher than the net tangible book value per share. Therefore, if you purchase shares of common stock in
this offering, you will pay a price per share that substantially exceeds our net tangible book value per share after this offering.
If you purchase shares of common stock in this offering, you will suffer immediate and substantial dilution of
$ per share, representing the difference between the public
offering price of $ per share in this offering and our as
adjusted net tangible book value per share as of September 30, 2023, after giving effect to this offering, and after
underwriting discounts and commissions and estimated offering expenses payable by us. Please refer to the section below entitled
“Dilution” for more information.
You may experience future dilution as a result of future equity
offerings or acquisitions.
In order to raise additional capital, we may
in the future offer additional shares of our common stock or other securities convertible into or exchangeable for our common stock at
prices that may not be the same as the price per share in this offering. We may sell shares or other securities in any future offering
at a price per share that is less than the price per share paid by investors in this offering, and investors purchasing shares or other
securities in the future could have rights superior to existing stockholders. The price per share at which we sell additional shares
of our common stock, or securities convertible or exchangeable into our common stock, in future transactions or acquisitions may be higher
or lower than the price per share paid by investors in this offering.
In addition, we may engage in one or more potential
acquisitions in the future, which could involve issuing our common stock as some or all of the consideration payable by us to complete
such acquisitions. If we issue common stock or securities linked to our common stock, the newly issued securities may have a dilutive
effect on the interests of the holders of our common stock. Additionally, future sales of newly issued shares used to effect an acquisition
could depress the market price of our common stock.
The Pre-Funded Warrants are speculative
in nature and may never have any value.
Commencing on the date of issuance, holders of
Pre-Funded Warrants may exercise their rights to acquire our common stock and pay an exercise price per share equal to $0.001 per share,
subject to certain adjustments, without expiration. Following this offering, the market value of the Pre-Funded Warrants, if any, is uncertain
and there can be no assurance that the market price of our common stock will ever equal or exceed their imputed offering price.
There is no public market for the Pre-Funded
Warrants being offered in this offering.
There is no established public trading market
for the Pre-Funded Warrants to purchase shares of our common stock being offered in this offering, and we do not expect a market to develop.
In addition, we do not intend to apply to list the Warrants on any national securities exchange or other nationally recognized trading
system. Without an active market, the liquidity of the Pre-Funded Warrants will be limited.
Holders of Pre-Funded Warrants will have
no rights as a common stockholder until such holders exercise their Pre-Funded Warrants and acquire our common stock.
Until holders of Pre-Funded Warrants acquire shares
of our common stock upon exercise of such warrants, holders of the Warrants will have no rights with respect to the shares of our common
stock underlying such Pre-Funded Warrants. Upon exercise of the Pre-Funded Warrants, the holders will be entitled to exercise the rights
of a common stockholder only as to matters for which the record date occurs after the exercise date.
We do not anticipate paying cash dividends
and, accordingly, stockholders must rely on share appreciation for any return on their investment.
We currently intend to retain our future earnings,
if any, to fund the development and growth of our businesses and do not anticipate that we will declare or pay any cash dividends on
our capital stock in the foreseeable future. See the section titled “Dividend Policy” in this prospectus supplement. In addition,
our ability to pay dividends is limited by covenants of our existing and outstanding indebtedness and may be limited by covenants of
any future indebtedness we incur. As a result, capital appreciation, if any, of our common stock will be your sole source of gain on
your investment for the foreseeable future. Investors seeking cash dividends should not invest in our common stock.
Our
common stock may become the target of a “short squeeze”.
In recent years, the securities of several companies
have increasingly experienced significant and extreme volatility in stock price due to short sellers of common stock and buy-and-hold
decisions of longer investors, resulting in what is sometimes described as a “short squeeze.” Short squeezes have caused
extreme volatility in those companies and in the market and have led to the price per share of those companies to trade at a significantly
inflated rate that is disconnected from the underlying value of the company. Sharp rises in a company’s stock price may force traders
in a short position to buy the shares to avoid even greater losses. Many investors who have purchased shares in those companies at an
inflated rate face the risk of losing a significant portion of their original investment as the price per share has declined steadily
as interest in those shares have abated. We may be a target of a short squeeze, and investors may lose a significant portion or all of
their investment if they purchase our shares at a rate that is significantly disconnected from our underlying value.
We are subject to certain contractual obligations
and limitations on our ability to consummate future financings under the Promissory Note issued by us to Streeterville on November 4,
2022, as amended in March 2023, July 2023, and February 2024.
Pursuant to the securities purchase agreement
we entered into in connection with the issuance of the Note to Streeterville, we are subject to certain restrictions on our ability to
issue securities during the term of the Note. Specifically, we agreed to obtain the Lender’s consent prior to issuing any debt
securities or certain equity securities where the pricing of such equity securities is tied to the public trading price of our common
stock. Furthermore, we also must offer the Lender the right to purchase up to 10% of future equity and debt securities offerings, subject
to certain exceptions and limitations, during the term of the Note.
Further, we have agreed to make certain monthly
redemption payments at the request of the Lender. Our failure to pay such redemptions, when due, may result in defaults under our agreements
with the Lender. If we are in default with respect to our obligations under the Note, the Lender may consider the Note immediately due
and payable and may elect to substantially increase the interest rate of the Note. We may not have the required funds to pay the required
note redemptions and such redemptions, or penalties in connection therewith, may have an adverse effect on our cash flows, results of
operations, and ability to pay our other debts as they come due.
Use
of Proceeds
We estimate that the net proceeds from this offering
will be approximately $ (or approximately $ million if the
underwriters exercise in full their option to purchase additional shares of common stock and/or Pre-Funded Warrants), based on a public
offering price of $ per share or $ per Pre-Funded Warrant, and after deducting
underwriting discounts and commissions and estimated offering expenses payable by us.
We expect to use any proceeds that we receive
from this offering for working capital and general corporate purposes. We may also use the proceeds from this offering to repay the Note
issued to Streeterville. The Note bears interest at a rate of 9% per annum and matures in August 2024. Accordingly, we retain broad
discretion over the use of the net proceeds from this offering. The precise amount and timing of the application of such proceeds will
depend upon our liquidity needs and the availability and cost of other capital over which we have little or no control. As of the date
of this prospectus supplement, we cannot specify with certainty all of the particular uses, and the respective amounts we may allocate
to those uses, for the net proceeds we receive.
DIVIDEND POLICY
We have never declared or paid any cash dividends
on our capital stock. We currently intend to retain all available funds and future earnings, if any, for the operation and expansion
of our business and do not anticipate declaring or paying any dividends in the foreseeable future. Any future determination related to
our dividend policy will be made at the discretion of our board of directors (the “Board”) after considering our financial
condition, results of operations, capital requirements, business prospects and other factors the Board deems relevant, and subject to
the restrictions contained in any financing instruments. Our ability to declare dividends may also be limited by restrictive covenants
pursuant to any other future debt financing agreements.
Dilution
If you invest in our securities, you will experience
immediate dilution to the extent of the difference between the public offering price per share of common stock that you pay in this offering
and the as adjusted net tangible book value per share after this offering (excluding the shares of common stock issuable upon exercise
of the Pre-Funded Warrants). The net tangible book value of our common stock as of September 30, 2023, was approximately $(6,217,449),
or $(0.09) per share. Net tangible book value per share of our common stock is equal to total assets minus intangible assets, goodwill,
and equity investments, less total liabilities, dividend by the total number of shares of common stock issued and outstanding as of September 30,
2023.
After giving effect to the issuance and sale of shares
of common stock offered hereby at a public offering price of $ per share and Pre-Funded Warrants to purchase
up to shares of common stock at a public offering price of $ per Pre-Funded Warrant, and after deducting underwriting discounts
and commissions and estimated offering expenses payable by us, our as adjusted net tangible book value as of September 30, 2023,
would have been approximately $ million, or $ per
share. The change represents an immediate increase in the net tangible book value per share of our common stock of $ per
share to existing stockholders and an immediate dilution of $ per share to new investors purchasing common
stock in this offering.
If holders of Pre-Funded Warrants exercise the
Pre-Funded Warrants in full, the as adjusted net tangible book value per share of common stock after giving effect to this offering (but
not the exercise of the underwriters’ option to purchase additional shares and/or Pre-Funded Warrants) would be $ per share, and
the dilution in net tangible book value per share to investors purchasing common stock in this offering would be $ per share.
The following table illustrates this per share
dilution, assuming the the underwriters do not exercise their option to purchase additional shares of common stock and the holders of
the Pre-Funded Warrants do not exercise any of the Pre-Funded Warrants:
| Public offering price per share |
|
|
|
|
|
$ |
|
|
| Net tangible book value per share as of September 30, 2023 |
|
$ |
(0.09 |
) |
|
|
|
|
| Increase in as adjusted net tangible book value per share attributable
to this offering |
|
$ |
|
|
|
|
|
|
| As adjusted net tangible book value per share as of September 30, 2023,
after giving effect to this offering |
|
|
|
|
|
$ |
|
|
| Dilution per share to new investors participating in the offering |
|
|
|
|
|
$ |
|
|
The information above assumes that the
underwriters do not exercise their option to purchase additional shares of common stock and/or Pre-Funded Warrants. If the
underwriters exercise in full their option to purchase additional
shares
of common stock and/or Pre-Funded Warrants, our adjusted net tangible book value after this offering would be approximately $
,
or
$
per share, representing an increase in net tangible book value of approximately $
per
share to existing stockholders and immediate dilution in net tangible book value of approximately $
per
share to investors participating in this offering.
The table above is based on 83,919,554 shares
of common stock outstanding as of September 30, 2023, and excludes:
| |
· |
3,450,000 shares of common
stock issuable upon the exercise of the Public Warrants, at an exercise price of $11.50 per share; |
| |
· |
2,548,849 shares of common
stock reserved for future issuance pursuant to outstanding option awards, with a weighted average exercise price of $3.32 per share; |
| |
· |
964,214 shares of common
stock available for future grants under the 2021 Plan; |
| |
· |
up to 45,000,000 shares
of common stock issuable upon conversion of the Note; |
| |
|
|
| |
· |
3,000,000 shares of
common stock issuable upon the conversion of 3,000,000 shares of Series A Preferred Stock at a conversion price of $0.40 per
share; |
| |
|
|
| |
· |
3,000,000 shares
of common stock issuable upon the exercise of warrants issued to certain investors pursuant to the August 2023 Purchase Agreement,
at an exercise price of $0.40 per share; and |
| |
|
|
| |
· |
the shares of common stock
issuable upon exercise of the underwriters warrants issued in this offering. |
To the extent outstanding options or
warrants are exercised, including any of the Pre-Funded Warrants, or outstanding convertible securities are converted into shares of
common stock, there will be further dilution to investors. In addition, to the extent that we issue additional equity securities in
connection with future capital raising activities, our then-existing stockholders may experience dilution.
UNDERWRITING
EF Hutton LLC is acting as representative of each
of the underwriters named below (the “Representative”). Subject to the terms and conditions set forth in an underwriting agreement
between us and the Representative, we have agreed to sell to each underwriter named below such securities set forth opposite its name
in the below table at the public offering price, less the underwriting discounts and commissions set forth on the cover page of this prospectus.
|
Underwriter |
|
Number of
Shares of
Common
Stock |
|
|
Number of
Pre-Funded
Warrants |
|
| EF Hutton LLC |
|
|
|
|
|
|
|
|
| Total |
|
|
|
|
|
|
|
|
The underwriting agreement provides that, subject
to the terms and conditions contained therein, the underwriters are obligated to take and pay for all of the shares of common stock in
the offering if any of the shares of common stock are purchased, other than the shares of common stock covered by the over-allotment option
described below. If an underwriter defaults, the underwriting agreement provides that the purchase commitments of the non-defaulting underwriters
may be increased or the underwriting agreement may be terminated.
Over-Allotment Option
We have granted to the Representative an option,
exercisable no later than 45 calendar days after the date of the underwriting agreement, to purchase up to an additional shares
of common stock and/or Pre-Funded Warrants (15% of shares and shares subject to Pre-Funded Warrants sold in this offering) at the public
offering prices, less the underwriting discounts. If the Representative exercises this option, it will be obligated, subject to conditions
contained in the underwriting agreement, to purchase a number of additional shares and/or Pre-Funded Warrants on the same terms as those
on which the shares and Pre-Funded Warrants are being offered.
Discounts and Commissions
The underwriters propose to offer the shares of
common stock and Pre-Funded Warrants initially at the public offering prices on the cover page of this prospectus. After the initial offering,
the public offering prices, concession or any other term of the offering may be changed.
The following table summarizes the underwriting
discounts and commissions and proceeds, before expenses, to us, assuming both no exercise and full exercise of the over-allotment option:
| |
|
Per
Share |
|
|
Per
Pre-Funded
Warrant |
|
|
Total Without
Over-Allotment
Option |
|
|
Total With
Over-Allotment
Option |
|
| Public offering price |
|
$ |
|
|
|
$ |
|
|
|
$ |
|
|
|
$ |
|
|
| Underwriting discounts and commissions (8.0%)(1) |
|
$ |
|
|
|
$ |
|
|
|
$ |
|
|
|
$ |
|
|
| Proceeds to us, before fees and expenses |
|
$ |
|
|
|
$ |
|
|
|
$ |
|
|
|
$ |
|
|
| (1) |
We have agreed to pay a non-accountable expense allowance to the Representative equal to 1% of the gross proceeds received in this offering which is not included in the underwriting discounts and commission. |
We estimate expenses payable by us in connection
with this offering, other than the underwriting discounts and commissions and non-accountable expense allowance referred to above, will
be approximately $ ($ if the over-allotment option is exercised in full), which includes certain expenses incurred by the underwriters
in connection with this offering that will be reimbursed by us. We have agreed to reimburse the Representative for all reasonable out-of-pocket
costs and expenses incident to the performance of its obligations under the underwriting agreement (including, without limitation, the
fees and expenses of its outside attorneys), provided that, excluding certain expenses related to indemnification and Blue-Sky and FINRA
filings, if any, such costs and expenses shall not exceed $100,000.
We have also agreed to indemnify the underwriters
against certain liabilities, including civil liabilities under the Securities Act or to contribute to payments that the underwriters may
be required to make in respect of those liabilities.
Underwriters’ Warrants
Upon
closing of this offering, we have agreed to issue to the Representative warrants, or the underwriter warrants, to purchase shares
of our common stock (5% of the aggregate number of shares of common stock and Pre-Funded Warrants sold in this offering) as a portion
of the underwriting compensation payable to the underwriters in connection with this offering. The underwriter warrants will be exercisable
at a per share exercise price equal to 110% of the public offering price per share in this offering. The underwriter warrants are exercisable
at any time and from time to time, in whole or in part, during the four and one half year period commencing 180 days from the commencement
of sales in this offering.
In addition, the warrants provide for registration
rights upon request, in certain cases. The sole demand registration right provided will not be greater than four and one-half years beginning
on the Initial Exercise Date in compliance with FINRA Rule 5110(g)(8)(C). The piggyback registration rights provided will not be greater
than five years from the initial exercise date of the underwriter warrants in compliance with FINRA Rule 5110(g)(8)(D). We will bear all
fees and expenses attendant to registering the securities issuable on exercise of the warrants other than underwriting commissions incurred
and payable by the holders. The exercise price and number of shares issuable upon exercise of the warrants may be adjusted in certain
circumstances including in the event of a stock dividend or our recapitalization, reorganization, merger or consolidation. However, the
warrant exercise price or underlying shares will not be adjusted for issuances of shares of common stock at a price below the warrant
exercise price.
Tail Financing
We agreed to pay the Representative a cash fee
equal to eight percent (8%) of the gross proceeds received by us from the sale of any equity, debt and/or equity derivative instruments
to any investor actually introduced by the Representative to us during the term of engagement with it, in connection with any public or
private financing or capital raise (each a “Tail Financing”), and such Tail Financing is consummated at any time during the
term of our engagement with them or within the six month period following the expiration or termination of our engagement with them (the
“Tail Period”), provided that such Tail Financing is by a party actually introduced to the Company in an offering in which
we had direct knowledge of such party’s participation
Lock-Up Agreements
In connection with this offering, our executive
officers and directors have agreed with the Representative that, subject to certain customary exceptions, without the prior written consent
of the Representative, for a period commencing on the date of the lock-up agreement and ending on the date that is the 90 days after the
closing of the offering, that they shall not offer, pledge, sell, contract to sell, sell any option or contract to purchase, purchase
any option or contract to sell, grant any option, right or warrant to purchase, lend, or otherwise transfer or dispose of, directly or
indirectly, any shares of capital stock of the Company or any securities convertible into or exercisable or exchangeable for shares of
capital stock of the Company.
Company Standstill
In connection with this offering, we have agreed
that, without the prior written consent of the Representative, for a period commencing on the date of the underwriting agreement and ending
on the date that is 90 days after the closing of the offering, we will not (i) offer, pledge, sell, contract to sell, sell any option
or contract to purchase, purchase any option or contract to sell, grant any option, right or warrant to purchase, lend, or otherwise transfer
or dispose of, directly or indirectly, any shares of capital stock of the Company or any securities convertible into or exercisable or
exchangeable for shares of capital stock of the Company, (ii) file or caused to be filed any registration statement with the Commission
relating to the offering of any shares of capital stock of the Company or any securities convertible into or exercisable or exchangeable
for shares of capital stock of the Company, (iii) complete any offering of debt securities of the Company, other than entering into a
line of credit with a traditional bank, or (iv) enter into any swap or other arrangement that transfers to another, in whole or in part,
any of the economic consequences of ownership of capital stock of the Company, whether any such transaction described in clause (i), (ii),
(iii) or (iv) above is to be settled by delivery of shares of capital stock of the Company or such other securities, in cash or otherwise.
These restrictions above do not apply in certain
situations, including, among others:
| |
● |
the issuance and sale of shares of common stock and/or Pre-Funded Warrants (including any shares of common stock issuable upon exercise of the Pre-Funded Warrants) to be sold on connection with the offering; |
| |
● |
the issuance by the Company of shares of common stock upon the exercise of a stock option or warrant or the conversion of a security outstanding on the date hereof; and |
| |
● |
the issuance by the Company of stock options, shares of capital stock of the Company or other awards under any equity compensation plan of the Company. |
Nasdaq Listing
Our
common stock and the Public Warrants are listed on Nasdaq under the symbols “NRXP” and “NRXPW”, respectively.
Price Stabilization, Short Positions and Penalty
Bids
In connection with this offering, the underwriters
may engage in transactions that stabilize, maintain or otherwise affect the price of our common stock. Specifically, the underwriters
may over-allot in connection with this offering by selling more shares of common stock than are set forth on the cover page of this prospectus.
This creates a short position in our common stock for its own account. The short position may be either a covered short position or a
naked short position. In a covered short position, the number of shares of common stock over-allotted by the underwriters is not greater
than the number of shares of our common stock that they may purchase in the over-allotment option. In a naked short position, the number
of shares of our common stock involved is greater than the number of shares of common stock in the over-allotment option. To close out
a short position, the underwriters may elect to exercise all or part of the over-allotment option. The underwriters may also elect to
stabilize the price of our common stock or reduce any short position by bidding for, and purchasing, our common stock in the open market.
The underwriters may also impose a penalty bid.
This occurs when a particular underwriter or dealer repays selling concessions allowed to it for distributing a security in this offering
because the underwriter repurchases that security in stabilizing or short covering transactions.
Finally, the underwriters may bid for, and purchase,
shares of our common stock in market making transactions, including “passive” market making transactions as described below.
These activities may stabilize or maintain the
market price of our common stock at a price that is higher than the price that might otherwise exist in the absence of these activities.
The underwriters are not required to engage in these activities and may discontinue any of these activities at any time without notice.
In connection with this offering, the underwriters
and selling group members, if any, or their affiliates may engage in passive market making transactions in our common stock immediately
prior to the commencement of sales in this offering, in accordance with Rule 103 of Regulation M under the Exchange Act. Rule 103
generally provides that:
| |
● |
a passive market maker may not effect transactions or display bids for our common stock in excess of the highest independent bid price by persons who are not passive market makers; |
| |
● |
net purchases by a passive market maker on each day are generally limited to 30% of the passive market maker’s average daily trading volume in our common stock during a specified two-month prior period or 200 shares, whichever is greater, and must be discontinued when that limit is reached; and |
| |
● |
passive market making bids must be identified as such. |
Electronic Distribution
A prospectus in electronic format may be made
available on the websites maintained by the underwriters. The Representative may agree to allocate a number of shares to underwriters
for sale to their online brokerage account holders. Internet distributions will be allocated by the Representative to underwriters that
may make internet distributions on the same basis as other allocations. In connection with the offering, the underwriters or syndicate
members may distribute prospectuses electronically. No forms of electronic prospectus other than prospectuses that are printable as Adobe®
PDF will be used in connection with this offering.
Other than the prospectus in electronic format,
the information on any underwriter’s website and any information contained in any other website maintained by an underwriter is
not part of the prospectus or the registration statement of which this prospectus forms a part, has not been approved and/or endorsed
by us or any underwriter in its capacity as underwriter and should not be relied upon by investors.
Affiliations
Each underwriter and its affiliates may provide,
from time to time, investment banking and financial advisory services to us in the ordinary course of business, for which they may receive
customary fees and commissions.
Foreign Regulatory Restrictions on Purchase
of our Shares
We have not taken any action to permit a public
offering of our shares outside the United States or to permit the possession or distribution of this prospectus outside the United States.
People outside the United States who come into possession of this prospectus must inform themselves about and observe any restrictions
relating to this offering of our shares and the distribution of this prospectus outside the United States.
DESCRIPTION
OF SECURITIES BEING OFFERED
The following is a description of our capital
stock and certain provisions of our Second Amended and Restated Certificate of Incorporation, Amended and Restated By-Laws, and certain
provisions of applicable law. The following is only a summary and is qualified by applicable law and by the provisions of our Second Amended
and Restated Certificate of Incorporation and Amended and Restated By-Laws, copies of which are included as exhibits to the registration
statement of which this prospectus forms a part. We are incorporated in the State of Delaware. The rights of our stockholders are generally
covered by Delaware law and our Second Amended and Restated Certificate of Incorporation and Amended and Restated By-Laws. The terms of
our capital stock are therefore subject to Delaware law.
We are offering shares of our common stock
and Pre-Funded Warrants to purchase shares of our common stock. We are also registering the shares of common stock issuable from time
to time upon exercise of the Pre-Funded Warrants offered hereby.
General
Our authorized capital stock consists of 500,000,000
shares of common stock, $0.001 par value per share, and 50,000,000 shares of preferred stock, $0.001 par value per share. As of the date
of this prospectus supplement (i) 86,675,580 shares of common stock were issued and outstanding; and (ii) 3,000,000 shares of
Series A Preferred Stock were issued and outstanding.
Common Stock
The material terms and provisions of our common
stock are described under the caption “Description of Capital Stock” in the accompanying prospectus and are incorporated herein
by reference.
Pre-Funded Warrants
The following summary of certain terms and provisions
of the Pre-Funded Warrants that are being offered hereby is not complete and is subject to, and qualified in its entirety by, the provisions
of the Pre-Funded Warrant, the form of which will be filed as an exhibit to a Current Report on Form 8-K in connection with this offering
and incorporated by reference into the registration statement of which this prospectus supplement forms a part. Prospective investors
should carefully review the terms and provisions of the form of Pre-Funded Warrant for a complete description of the terms and conditions
of the Pre-Funded Warrants.
The Pre-Funded Warrants will be issued in certificated
form only.
Duration and exercise price
Each Pre-Funded Warrant offered hereby has an
initial exercise price per share equal to $0.001. The Pre-Funded Warrants are immediately exercisable and have an indefinite term. The
exercise price and number of shares of common stock issuable upon exercise is subject to appropriate adjustment in the event of stock
dividends, stock splits, reorganizations or similar events affecting our common stock and the exercise price.
Exercisability
The Pre-Funded Warrants will be exercisable, at
the option of each holder, in whole or in part, by delivering to us a duly executed exercise notice accompanied by payment in full for
the number of shares of our common stock purchased upon such exercise (except in the case of a cashless exercise as discussed below).
A holder (together with its affiliates) may not exercise any portion of such holder’s Pre-Funded Warrant to the extent that the
holder would own more than 4.99% (or, at the election of the purchaser, 9.99%) of the outstanding shares of common stock immediately after
exercise, except that upon prior notice from the holder to us, the holder may increase the amount of ownership of outstanding shares of
common stock after exercising the holder’s Pre-Funded Warrants up to 9.99% of the number of shares of common stock outstanding immediately
after giving effect to the exercise, as such percentage ownership is determined in accordance with the terms of the Pre-Funded Warrants.
No fractional shares of common stock will be issued in connection with the exercise of a Pre-Funded Warrant. In lieu of fractional shares,
we will either pay the holder an amount in cash equal to the fractional amount multiplied by the exercise price or round up to the next
whole share.
Cashless exercise
The Pre-Funded Warrants may be exercised, in whole
or in part, by means of cashless exercise. In lieu of making the cash payment otherwise contemplated to be made to us upon such exercise
in payment of the aggregate exercise price, the holder may elect instead to receive upon such exercise (either in whole or in part) the
net number of shares of common stock determined according to a formula set forth in the Pre-Funded Warrants.
Fundamental transactions
In the event of any fundamental transaction, as
described in the Pre-Funded Warrants and generally including any merger with or into another entity, sale of all or substantially all
of our assets, tender offer or exchange offer, or reclassification of our shares of common stock, upon any subsequent exercise of a Pre-Funded
Warrant, the holder will have the right to receive as alternative consideration, for each share of common stock that would have been issuable
upon such exercise immediately prior to the occurrence of such fundamental transaction, the number of shares of common stock of the successor
or acquiring corporation of our company, if it is the surviving corporation, and any additional consideration receivable upon or as a
result of such transaction by a holder of the number of shares of common stock for which the Pre-Funded Warrant is exercisable immediately
prior to such event.
Transferability
Subject to applicable laws, a Pre-Funded Warrant
or the rights thereunder may be transferred or assigned, in whole or in part. The ownership of the Pre-Funded Warrants and any transfers
of the Pre-Funded Warrants will be registered in a warrant register maintained by the warrant agent. We will initially act as warrant
agent.
Exchange listing
There is no trading market available for the Pre-Funded
Warrants on any securities exchange or nationally recognized trading system. We do not intend to list the Pre-Funded Warrants on any securities
exchange or nationally recognized trading system.
Right as a stockholder
Except as otherwise provided in the Pre-Funded
Warrants or by virtue of such holder’s ownership of shares of our common stock, the holders of the Pre-Funded Warrants do not have
the rights or privileges of holders of our common stock, including any voting rights, until they exercise their Pre-Funded Warrants.
MATERIAL
U.S. Federal Income TAX CONSIDERATIONS
The following is a summary of the material U.S.
federal income tax considerations relating to the purchase, ownership and disposition of our common stock purchased in this offering
but is for general information purposes only and does not purport to be a complete analysis of all the potential tax considerations relating
thereto. This summary is based upon the provisions of the Internal Revenue Code of 1986, as amended (the “Code”), existing
and proposed Treasury regulations promulgated thereunder, administrative rulings, and judicial decisions, all as of the date hereof.
These authorities may be changed, possibly retroactively, so as to result in U.S. federal income tax consequences different from those
set forth below. There can be no assurance that the Internal Revenue Service (the “IRS”) will not challenge one or more of
the tax consequences described herein, and we have not obtained, and do not intend to obtain, an opinion of counsel or ruling from the
IRS with respect to the U.S. federal income tax considerations relating to the purchase, ownership or disposition of our common stock.
This summary does not address any alternative
minimum tax considerations, any considerations regarding the tax on net investment income, or the tax considerations arising under the
laws of any state, local or non-U.S. jurisdiction, or under any non-income tax laws, including U.S. federal gift and estate tax
laws. In addition, this summary does not address tax considerations applicable to an investor’s particular circumstances or to
investors that may be subject to special tax rules, including, without limitation:
| · | banks,
insurance companies, or other financial institutions; |
| · | tax-exempt
organizations or governmental organizations; |
| · | regulated
investment companies and real estate investment trusts; |
| · | controlled
foreign corporations, passive foreign investment companies and corporations that accumulate
earnings to avoid U.S. federal income tax; |
| · | brokers
or dealers in securities or currencies; |
| · | traders
in securities that elect to use a mark-to-market method of accounting for their securities
holdings; |
| · | persons
that own, or are deemed to own, more than five percent of our common stock (except to the
extent specifically set forth below); |
| · | tax-qualified
retirement plans; |
| · | certain
former citizens or long-term residents of the United States; |
| · | partnerships
or entities or arrangements classified as partnerships for U.S. federal income tax purposes
and other pass-through entities (and investors therein); |
| · | persons
who hold our common stock as a position in a hedging transaction, “straddle,”
“conversion transaction,” or other risk reduction transaction or integrated investment; |
| · | persons
who hold or receive our common stock pursuant to the exercise of any employee stock option
or otherwise as compensation; |
| · | persons
who do not hold our common stock as a capital asset within the meaning of Section 1221 of
the Code; or |
| · | persons
deemed to sell our common stock under the constructive sale provisions of the Code. |
In addition, if a partnership (or entity or arrangement
classified as a partnership for U.S. federal income tax purposes) holds our common stock, the tax treatment of a partner in such partnership
generally will depend on the status of the partner and upon the activities of the partnership. Accordingly, partnerships that hold our
common stock, and partners in such partnerships, should consult their tax advisors.
You
are urged to consult your own tax advisors with respect to the application of the U.S. federal income tax laws to your particular situation,
as well as any tax consequences of the purchase, ownership, and disposition of our common stock arising under the U.S.
federal estate or gift tax laws or under the laws of any state, local, non-U.S., or other taxing jurisdiction, or under any applicable
tax treaty.
Consequences to U.S. Holders
The following is a summary of the U.S. federal
income tax consequences that will apply to a U.S. holder of our common stock. For purposes of this discussion, you are a “U.S.
holder” if, for U.S. federal income tax purposes, you are a beneficial owner of our common stock, other than a partnership, that
is:
| · | an
individual citizen or resident of the United States; |
| · | a
corporation or other entity taxable as a corporation created or organized in the United States
or under the laws of the United States, any State thereof or the District of Columbia; |
| · | an
estate whose income is subject to U.S. federal income tax regardless of its source; or |
| · | a
trust (x) whose administration is subject to the primary supervision of a U.S. court and
which has one or more “United States persons” (within the meaning of Section
7701(a)(30) of the Code) who have the authority to control all substantial decisions of the
trust or (y) which has made a valid election to be treated as a “United States person.” |
Distributions
As described in the section titled “Dividend
Policy,” we have not declared or paid cash dividends on our common stock and do not anticipate paying any dividends on our common
stock in the foreseeable future. However, if we do make distributions on our common stock, those payments will constitute dividends for
U.S. federal income tax purposes to the extent paid from our current or accumulated earnings and profits, as determined under U.S. federal
income tax principles. To the extent those distributions exceed both our current and our accumulated earnings and profits, the excess
will constitute a return of capital and will first reduce your basis in our common stock, but not below zero, and then will be treated
as gain from the sale of stock as described below under “—Sale, Exchange or Other Taxable Disposition of Our Common Stock.”
Dividend income may be taxed to an individual
U.S. holder at rates applicable to long-term capital gains, provided that a minimum holding period and other limitations and requirements
are satisfied. Any dividends that we pay to a U.S. holder that is a taxable corporation generally will qualify for a dividends-received
deduction allowed to U.S. corporations in respect of dividends received from other U.S. corporations equal to a portion of any dividends
received, subject to generally applicable limitations on that deduction. U.S. holders should consult their own tax advisors regarding
the holding period and other requirements that must be satisfied in order to qualify for the reduced tax rate on dividends or the dividends-received deduction.
Sale, Exchange, or Other Taxable Disposition of Our Common Stock
A U.S. holder will generally recognize capital
gain or loss on the sale, exchange, or other taxable disposition of our common stock. The amount of gain or loss will equal the difference
between the amount realized on the sale and such U.S. holder’s tax basis in such common stock. The amount realized will include
the amount of any cash and the fair market value of any other property received in exchange for such common stock. Gain or loss will
be long-term capital gain or loss if the U.S. holder has held the common stock for more than one year. Long-term capital gains
of non-corporate U.S. holders are generally taxed at preferential rates. The deductibility of capital losses is subject to certain
limitations.
Consequences to Non-U.S. Holders
The following is a summary of the U.S. federal
income tax consequences that will apply to a non-U.S. holder of our common stock. A “non-U.S. holder” is a beneficial owner
of our common stock (other than a partnership or an entity or arrangement treated as a partnership for U.S. federal income tax purposes)
that, for U.S. federal income tax purposes, is not a U.S. holder.
Distributions
Distributions will be treated as described above
under “Consequences to U.S. Holders—Distributions.” Subject to the discussion below regarding effectively connected
income, any dividend paid to a non-U.S. holder generally will be subject to U.S. withholding tax either at a rate of 30% of the gross
amount of the dividend or such lower rate as may be specified by an applicable income tax treaty. In order to receive a reduced treaty
rate, a non-U.S. holder must provide us with an IRS Form W-8BEN, IRS Form W-8BEN-E or other applicable IRS Form W-8 properly
certifying qualification for the reduced rate. These forms must be updated periodically. A non-U.S. holder eligible for a reduced rate
of U.S. withholding tax pursuant to an income tax treaty may obtain a refund of any excess amounts withheld by timely filing an appropriate
claim for a refund with the IRS. If a non-U.S. holder holds our common stock through a financial institution or other agent acting on
the non-U.S. holder’s behalf, the non-U.S. holder will be required to provide appropriate documentation to the agent, which then
may be required to provide certification to us or our paying agent, either directly or through other intermediaries.
Dividends received by a non-U.S. holder that
are effectively connected with its conduct of a U.S. trade or business (and, if required by an applicable income tax treaty, attributable
to a permanent establishment or fixed base maintained by the non-U.S. holder in the United States) are generally exempt from such withholding
tax if the non-U.S. holder satisfies certain certification and disclosure requirements. In order to obtain this exemption, the non-U.S.
holder must provide us with an IRS Form W-8ECI or other applicable IRS Form W-8 properly certifying such exemption. Such effectively
connected dividends, although not subject to withholding tax, are taxed at the same graduated U.S. federal income tax rates applicable
to U.S. holders, net of certain deductions and credits. In addition, dividends received by a corporate non-U.S. holder that are effectively
connected with its conduct of a U.S. trade or business may also be subject to a branch profits tax at a rate of 30% or such lower rate
as may be specified by an applicable income tax treaty. Non-U.S. holders should consult their own tax advisors regarding any applicable
tax treaties that may provide for different rules.
Gain on Sale, Exchange, or Other Taxable Disposition of Our
Common Stock
Subject to the discussion below regarding backup
withholding and foreign accounts, a non-U.S. holder generally will not be required to pay U.S. federal income tax on any gain realized
upon the sale, exchange, or other taxable disposition of our common stock unless:
| · | the
gain is effectively connected with the non-U.S. holder’s conduct of a U.S. trade or
business (and, if required by an applicable income tax treaty, the gain is attributable to
a permanent establishment or fixed base maintained by the non-U.S. holder in the United States); |
| · | the
non-U.S. holder is a non-resident alien individual who is present in the United States for
a period or periods aggregating 183 days or more during the calendar year in which the sale
or disposition occurs and certain other conditions are met; or |
| · | shares
of our common stock constitute U.S. real property interests by reason of our status as a
“United States real property holding corporation” (a “USRPHC”) for
U.S. federal income tax purposes at any time within the shorter of the five-year period preceding
the non-U.S. holder’s disposition of, or the non-U.S. holder’s holding period
for, our common stock. |
We believe that we are not currently a USRPHC
for U.S. federal income tax purposes, and the remainder of this discussion so assumes. However, because the determination of whether
we are a USRPHC depends on the fair market value of our U.S. real property relative to the fair market value of our other business assets,
there can be no assurance that we will not become a USRPHC in the future. Even if we become a USRPHC, however, if our common stock becomes
regularly traded on an established securities market (as defined by applicable Treasury regulations), such common stock will be treated
as U.S. real property interests only if the non-U.S. holder actually or constructively held more than five percent of such regularly
traded common stock at any time during the shorter of the five-year period preceding the non-U.S. holder’s disposition of,
or the non-U.S. holder’s holding period for, our common stock.
If the non-U.S. holder is described in the first
bullet above, it will be required to pay tax on the net gain derived from the sale, exchange, or other taxable disposition under regular
graduated U.S. federal income tax rates, and a corporate non-U.S. holder described in the first bullet above also may be subject to the
branch profits tax at a rate of 30%, or such lower rate as may be specified by an applicable income tax treaty. An individual non-U.S.
holder described in the second bullet above will be required to pay a flat 30% tax (or such lower rate specified by an applicable income
tax treaty) on the gain derived from the sale, exchange, or other taxable disposition, which gain may be offset by U.S. source capital
losses for the year (provided the non-U.S. holder has timely filed U.S. federal income tax returns with respect to such losses). Non-U.S.
holders should consult their own tax advisors regarding the withholding rules that may be applicable to the non-U.S. holder.
Backup Withholding and Information Reporting
Generally, we must report annually to the IRS
the amount of dividends paid to you, your name and address, and the amount of tax withheld, if any. A similar report will be sent to
you. Pursuant to applicable income tax treaties or other agreements, the IRS may make these reports available to tax authorities in your
country of residence if you reside outside of the United States.
Payments of dividends on or of proceeds from
the disposition of our common stock made to you may be subject to information reporting and backup withholding. Backup withholding may
apply at a current rate of 24% unless you (i) provide the payor with a correct taxpayer identification number and comply with applicable
certification requirements, or (ii) establish an exemption, for example, by properly certifying your non-U.S. status on an IRS Form W-8BEN or
IRS Form W-8BEN-E or other applicable IRS Form W-8. Notwithstanding the foregoing, backup withholding and information reporting
may apply if either we or our paying agent has actual knowledge, or reason to know, that you are a U.S. person that is not an exempt
recipient.
Backup withholding is not an additional tax;
rather, the U.S. federal income tax liability of persons subject to backup withholding will be reduced by the amount of tax withheld.
If withholding results in an overpayment of taxes, a refund or credit may generally be obtained from the IRS, provided that the required
information is furnished to the IRS in a timely manner.
Foreign Account Tax Compliance Act
The Foreign Account Tax Compliance Act (“FATCA”)
generally imposes withholding tax at a rate of 30% on dividends on our common stock paid to a “foreign financial institution”
(as specially defined under these rules), unless such institution enters into an agreement with the U.S. government to, among other things,
withhold on certain payments and to collect and provide to the U.S. tax authorities substantial information regarding the U.S. account
holders of such institution (which includes certain equity and debt holders of such institution, as well as certain account holders that
are foreign entities with U.S. owners) or otherwise establishes an exemption. FATCA also generally imposes a U.S. federal withholding
tax of 30% on dividends on our common stock paid to a “non-financial foreign entity” (as specially defined for purposes
of these rules) unless such entity provides the withholding agent with a certification identifying certain substantial direct and indirect
U.S. owners of the entity, certifies that there are no U.S. owners, or otherwise establishes an exemption. The withholding provisions
under FATCA generally apply to dividends paid by us. Under certain circumstances, a non-U.S. holder might be eligible for refunds or
credits of such taxes. An intergovernmental agreement between the United States and an applicable foreign country may modify the requirements
described in this paragraph. Non-U.S. holders should consult their own tax advisors regarding the possible implications of this legislation
on their investment in our common stock.
Each prospective investor should consult its
own tax advisor regarding the particular U.S. federal, state and local and non-U.S. consequences of purchasing, owning and disposing
of our common stock, including the consequences of any proposed changes in applicable laws.
Legal
Matters
The validity of the common stock being offered
hereby will be passed upon for us by Haynes and Boone, LLP, New York, New York. Sichenzia Ross Ference Carmel LLP, New York, New York,
is counsel to the underwriters in connection with this offering.
Experts
The consolidated financial statements of NRx
Pharmaceuticals, Inc. as of December 31, 2022 and 2021, and for each of the years then ended have been incorporated by reference
herein in reliance upon the report of KPMG LLP, independent registered public accounting firm, incorporated by reference herein, and
upon the authority of such firm as experts in accounting and auditing.
Where
You Can Find More Information
We file annual, quarterly and current reports,
proxy statements and other information with the SEC. The SEC maintains an Internet website at http://www.sec.gov which contains reports,
proxy and information statements and other information regarding issuers that file electronically with the SEC. Our SEC filings are available
to the public from the SEC’s Internet website.
This prospectus supplement and the accompanying
prospectus are part of a registration statement that we have filed with the SEC relating to the securities to be offered. This prospectus
supplement and the accompanying prospectus omit some of the information we have included in the registration statement and the accompanying
exhibits and schedules in accordance with the rules and regulations of the SEC, and we refer you to the omitted information. The
statements this prospectus supplement makes pertaining to the content of any contract, agreement or other document that is an exhibit
to the registration statement necessarily are summaries of their material provisions and do not describe all provisions, exceptions and
qualifications contained in those contracts, agreements or documents. You should read those contracts, agreements or documents for information
that may be important to you. The registration statement, exhibits and schedules are available at the SEC’s Public Reference Room
or through its Internet website.
Incorporation
of Certain Documents by Reference
The SEC allows us to “incorporate by reference”
much of the information we file with the SEC, which means that we can disclose important information to you by referring you to those
publicly available documents. The information that we incorporate by reference in this prospectus supplement is considered to be part
of this prospectus supplement. Because we are incorporating by reference future filings with the SEC, this prospectus supplement is continually
updated and those future filings may modify or supersede some of the information included or incorporated in this prospectus supplement.
This means that you must look at all of the SEC filings that we incorporate by reference to determine if any of the statements in this
prospectus supplement or in any document previously incorporated by reference have been modified or superseded. This prospectus supplement
incorporates by reference the documents listed below and any future filings we make with the SEC under Sections 13(a), 13(c), 14 or 15(d) of
the Securities Exchange Act of 1934, as amended (in each case, other than information furnished under Item 2.02 or Item 7.01 of Form 8-K),
on or after the date of this prospectus supplement until the earlier of the date on which all of the securities registered hereunder
have been sold or the registration statement of which this prospectus supplement and the accompanying prospectus form a part of have
been withdrawn:
| |
· |
Our Annual Report on Form 10-K
for the fiscal year ended December 31, 2022, filed with the SEC on March 31,
2023, as amended on Form 10-K/A on May 1,
2023, and May 26,
2023, respectively; |
| |
· |
Our Quarterly Reports on Form 10-Q for
the quarters ended March 31, 2023, June 30, 2023, and September 30, 2023, filed with the SEC on May 15,
2023, August 14,
2023, and November 14,
2023, respectively;
|
| |
· |
Our Current Reports on
Form 8-K, filed with the SEC on February 2,
2023, February 13,
2023, February 23,
2023, February 24,
2023, March 3,
2023, March 9,
2023 (as amended on Form 8-K/A on March 14,
2023), April 21,
2023, June 5,
2023, June 6,
2023 (as amended on Form 8-K/A on June 7,
2023), July 28,
2023, August 14,
2023, August 31,
2023, September 1,
2023, September 14,
2023, October 23,
2023, November 21,
2023, November 22,
2023, December 21,
2023, December 29,
2023, January 23,
2024, February 2, 2024, February 14, 2024 and February 14, 2024; and |
We
maintain an Internet website at www.nrxpharma.com. The information contained in, or accessible through, our website does not constitute
a part of this prospectus supplement or the accompanying prospectus. We have included our website address in this prospectus supplement
solely as an inactive textual reference.
You should not assume that the information in
this prospectus supplement and the accompanying prospectus, or any document incorporated by reference herein or therein is accurate as
of any date other than the date of the applicable document. Any statement contained in a document incorporated or deemed to be incorporated
by reference into this prospectus supplement and the accompanying prospectus will be deemed to be modified or superseded for purposes
of this prospectus supplement to the extent that a statement contained in this prospectus supplement or the accompanying prospectus or
any other subsequently filed document that is deemed to be incorporated by reference into this prospectus supplement modifies or supersedes
the statement. Any statement so modified or superseded will not be deemed, except as so modified or superseded, to constitute a part
of this prospectus supplement.
You may request a copy of any or all documents
referred to above that have been or may be incorporated by reference into this prospectus supplement (excluding certain exhibits to the
documents) at no cost, by writing or calling us at the following address or telephone number:
NRx Pharmaceuticals, Inc.
Richard Narido
Interim Chief Financial Officer
1201 North Market Street, Suite 111
Wilmington, Delaware 19801
(484) 254-6134
TABLE
OF CONTENTS
PROSPECTUS
NRX
Pharmaceuticals, Inc.
$150,000,000
Common
Stock
Preferred
Stock
Debt
Securities
Depositary
Shares
Warrants
Rights
Purchase
Contracts
Units
We may offer
and sell from time to time shares of our common stock, par value $0.001 per share (the “Common Stock”), shares of
our preferred stock, debt securities, depositary shares, warrants, rights, purchase contracts or units, or any combination
thereof, in one or more offerings in amounts, at prices and on terms that we determine at the time of the offering, with an aggregate
initial offering price of up to $150,000,000 (or its equivalent in foreign currencies, currency units or composite currencies).
Each time we
offer securities pursuant to this prospectus, we will provide a prospectus supplement containing more information about the particular
offering together with this prospectus. The prospectus supplement also may add, update or change information contained in this
prospectus. This prospectus may not be used to offer and sell securities without a prospectus supplement.
These securities
may be sold on a continuous or delayed basis directly to or through agents, dealers or underwriters as designated from time to
time, or through a combination of these methods.
Our Common
Stock is listed on the Nasdaq Global Market (“Nasdaq”) under the symbol “NRXP” and our warrants are listed
on the Nasdaq under the symbol “NRXPW”. If we decide to list or seek a quotation for any other securities, the prospectus
supplement relating to those securities will disclose the exchange or market on which those securities will be listed or quoted.
Investing
in these securities involves significant risks. We strongly recommend that you read carefully the risks we describe in this prospectus
as well as in any accompanying prospectus supplement and the risk factors that are incorporated by reference into this prospectus
from our filings made with the Securities and Exchange Commission. See “Risk Factors” beginning on page 10
of this prospectus.
Neither
the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or
determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The date
of this prospectus is June 21, 2022.
TABLE
OF CONTENTS
TABLE OF
CONTENTS
| |
|
|
|
|
|
1 |
|
|
| |
|
|
|
|
|
2 |
|
|
| |
|
|
|
|
|
3 |
|
|
| |
|
|
|
|
|
4 |
|
|
| |
|
|
|
|
|
5 |
|
|
| |
|
|
|
|
|
10 |
|
|
| |
|
|
|
|
|
11 |
|
|
| |
|
|
|
|
|
12 |
|
|
| |
|
|
|
|
|
16 |
|
|
| |
|
|
|
|
|
26 |
|
|
| |
|
|
|
|
|
29 |
|
|
| |
|
|
|
|
|
31 |
|
|
| |
|
|
|
|
|
32 |
|
|
| |
|
|
|
|
|
33 |
|
|
| |
|
|
|
|
|
34 |
|
|
| |
|
|
|
|
|
37 |
|
|
| |
|
|
|
|
|
37 |
|
|
TABLE
OF CONTENTS
To understand
the terms of the securities offered by this prospectus, you should carefully read this prospectus and any applicable prospectus
supplement. You should also read the documents referred to under the heading “Where You Can Find More Information”
for information on us and the business conducted by us.
This prospectus
is part of a registration statement that we filed with the Securities and Exchange Commission (the “SEC”), using
a “shelf” registration process. Under this shelf registration process, we may offer and sell from time to time shares
of our Common Stock, shares of our preferred stock, debt securities, depositary shares, warrants, rights, purchase contracts
or units, or any combination thereof, in one or more offerings in amounts, at prices and on terms that we determine at the
time of the offering, with an aggregate initial offering price of up to $150,000,000 (or its equivalent in foreign currencies,
currency units or composite currencies). This prospectus provides you with a general description of the securities. Each
time we offer the securities, we will provide a prospectus supplement that describes the terms of the offering. The prospectus
supplement also may add, update or change information contained in this prospectus. Before making an investment decision, you
should read carefully both this prospectus and any prospectus supplement together with the documents incorporated by reference
into this prospectus as described below under the heading “Incorporation by Reference.”
The registration
statement that contains this prospectus, including the exhibits to the registration statement and the information incorporated
by reference, provides additional information about us and our securities. That registration statement can be found on the SEC’s
website at www.sec.gov.
You should
rely only on the information provided in the registration statement, this prospectus and in any prospectus supplement, including
the information incorporated by reference. We have not authorized anyone to provide you with different information. You should
not assume that the information in this prospectus or any supplement to this prospectus is accurate at any date other than the
date indicated on the cover page of these documents. We are not making an offer to sell the securities in any jurisdiction where
the offer or sale is not permitted.
We may sell
the securities to or through underwriters, dealers or agents or directly to purchasers. The securities may be sold for U.S. dollars,
foreign-denominated currency, currency units or composite currencies. Amounts payable with respect to any securities may
be payable in U.S. dollars or foreign-denominated currency, currency units or composite currencies as specified in the applicable
prospectus supplement. We and our agents reserve the sole right to accept or reject in whole or in part any proposed purchase
of the securities. The prospectus supplement, which we will provide each time we offer the securities, will set forth the names
of any underwriters, dealers or agents involved in the sale of the securities, and any related fee, commission or discount arrangements.
See “Plan of Distribution.”
The prospectus
supplement may also contain information about any material U.S. federal income tax considerations relating to the securities
covered by the prospectus supplement.
In this prospectus,
the terms “NRx,” “we,” “us” and “our” refer to NRX Pharmaceuticals, Inc.
WHERE
YOU CAN FIND MORE INFORMATION
As required
by the Securities Act, we filed a registration statement relating to the securities offered by this prospectus with the SEC.
This prospectus is a part of that registration statement, which includes additional information.
We are subject
to the reporting requirements of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), and are required
to file with the SEC annual, quarterly and current reports, proxy statements and other information. Such reports include our
audited financial statements. Our publicly available filings can be found on the SEC’s website at www.sec.gov. Our filings,
including the audited financial and additional information that we have made public to investors, may also be found on our website
at www.nrxpharma.com. Information on or accessible through our website does not constitute part of this prospectus (except for
SEC reports expressly incorporated by reference herein).
As permitted
by SEC rules, this prospectus does not contain all of the information we have included in the registration statement and the
accompanying exhibits and schedules we file with the SEC. You may refer to the registration statement, exhibits and schedules
for more information about us and the securities. The registration statement, exhibits and schedules are available through the
SEC’s website.
INCORPORATION
BY REFERENCE
The SEC allows
us to “incorporate by reference” the information we file with the SEC, which means that we can disclose important
information to you by referring you to those documents. The information incorporated by reference is considered to be part of
this prospectus. Information that we file later with the SEC will automatically update and supersede information in this prospectus.
In all cases, you should rely on the later information over different information included in this prospectus or the prospectus
supplement. The following documents have been filed by us with the SEC and are incorporated by reference into this prospectus:
•
our Annual Report on Form 10-K and Form 10-K/A for the year ended December 31, 2021
as filed with the SEC on March 31,
2022 and April 29,
2022, respectively;
•
•
our Current Reports on Form 8-K as filed with the SEC on February 3,
2022, February 11,
2022, March 9,
2022, March 18,
2022, May 25,
2022, and May 31,
2022, and June
8, 2022.
All reports
and other documents that we subsequently file with the SEC (other than any portion of such filings that are furnished under applicable
SEC rules rather than filed) pursuant to Section 13(a), 13(c), 14 or 15(d) of the Exchange Act after the date of this prospectus
and before the later of (1) the completion of the offering of the securities described in this prospectus and any prospectus
supplement and (2) the date we stop offering securities pursuant to this prospectus and any prospectus supplement, will
be deemed to be incorporated by reference into this prospectus and to be part of this prospectus from the date of filing of such
reports and documents. The information contained on our website (www.nrxpharma.com) is not incorporated into this prospectus.
You should
not assume that the information in this prospectus, the prospectus supplement, any applicable pricing supplement or any document
incorporated by reference is accurate as of any date other than the date of the applicable document. Any statement contained
in a document incorporated or deemed to be incorporated by reference into this prospectus will be deemed to be modified or superseded
for purposes of this prospectus to the extent that a statement contained in this prospectus or any other subsequently filed document
that is deemed to be incorporated by reference into this prospectus modifies or supersedes the statement. Any statement so modified
or superseded will not be deemed, except as so modified or superseded, to constitute a part of this prospectus.
You may request
a copy of any or all documents referred to above that have been or may be incorporated by reference into this prospectus (excluding
certain exhibits to the documents) at no cost, by writing or calling us at the following address or telephone number:
NRX Pharmaceuticals,
Inc.
Alessandra Daigneault
Chief Corporate Officer, General Counsel & Secretary
1201 North Market Street, Suite 111
Wilmington, Delaware 19801
(484) 254-6134
CAUTIONARY
NOTE REGARDING FORWARD-LOOKING STATEMENTS
This document
and the information incorporated by reference herein include “forward-looking statements” within the meaning of the
“safe harbor” provisions of the U.S. Private Securities Litigation Reform Act of 1995, which may include, but are
not limited to, statements regarding our financial outlook, product development, business prospects, and market and industry
trends and conditions, as well as the Company’s strategies, plans, objectives, and goals. These forward-looking statements
are based on current beliefs, expectations, estimates, forecasts, and projections of, as well as assumptions made by, and information
currently available to, the Company’s management. Words such as “expect,” “anticipate,” “should,”
“believe,” “hope,” “target,” “project,” “goals,” “estimate,”
“potential,” “predict,” “may,” “will,” “might,” “could,”
“would,” “seek,” “plan,” “intend,” “shall,” and variations of these
terms or the negative of these terms and similar expressions are intended to identify these forward-looking statements. These
forward-looking statements are, by their nature, subject to significant risks and uncertainties, many of which involve factors
or circumstances that are beyond the Company’s control. These risks and uncertainties include, but are not limited to,
our relatively limited operating history; our ability to expand, retain and motivate our employees and manage our growth; risks
associated with general industry conditions and competition; general economic factors, including interest rate and currency exchange
rate fluctuations; the impact of the global outbreak of the novel coronavirus disease (“COVID-19”); the impact of
pharmaceutical industry regulation and health care legislation in the United States and internationally; global trends toward
health care cost containment; technological advances, new products and patents attained by competitors; challenges inherent in
new product development, including obtaining regulatory approval; the Company’s ability to accurately predict future market
conditions; manufacturing difficulties or delays; changes in laws, rules or regulations relating to any aspect of the Company’s
business operations, or general economic, market and business conditions; financial instability of international economies and
sovereign risk; dependence on the effectiveness of the Company’s patents and other protections for innovative products;
and the exposure to litigation, including patent litigation, and/or regulatory actions. Furthermore, there can be no guarantees
with respect to pipeline products that the products will receive the necessary regulatory approvals or that they will prove to
be commercially successful. If underlying assumptions prove inaccurate or risks or uncertainties materialize, actual results
may differ materially from those set forth in the forward-looking statements. The Company assumes no obligation and does not
intend to update or otherwise revise any forward-looking statement, whether as a result of new information, future events or
otherwise, except as required by applicable law. As a result of these and other risks, uncertainties and assumptions, forward-looking
events and circumstances discussed herein might not occur in the way that the Company’s management expects, if at all.
Accordingly, you should not place reliance on any forward-looking statement, and all forward-looking statements are herein qualified
by reference to the cautionary statements set forth above.
Overview
NRx is a clinical-stage
pharmaceutical company which develops, through its wholly-owned operating subsidiaries, NeuroRx, Inc., (“NeuroRx”)
a Delaware corporation, and NeuroRx 2015 LTD, an Israeli limited liability company, novel therapeutics for the treatment of central
nervous system disorders and life-threatening pulmonary diseases. Our strategy is to apply innovative science to known molecules
in the pursuit of therapies for high unmet needs, including lethal conditions. Given the recent geopolitical changes and other
developments, our company recently re-prioritized its development activities and geographic focus. We are currently developing
the following pharmaceutical products across our psychiatry and pulmonary areas:
•
NRX-100 and NRX-101. NRX-100 and NRX-101 are NMDA-targeted medicines designed to address
both depression and suicidal ideation. NRX-101 is a patented, fixed dose combination of D-cycloserine and lurasidone. The NMDA
receptor of the brain is increasingly viewed as a key target for treating suicidal depression. However, to the best of our knowledge,
NRX-101 is the first investigational medicine to advance to Phase 3 for severe bipolar depression in patients with Acute
Suicidal Ideation and Behavior (“ASIB”). NRX-101 has not shown neurotoxicity or addiction. NRX-101 has been granted
Fast Track Designation, Breakthrough Therapy Designation, and a Special Protocol Agreement (“SPA”) by the U.S. Food
and Drug Administration (the “FDA”) for the treatment of severe bipolar depression in patients with ASIB after initial
stabilization with ketamine or other effective therapy.
•
ZYESAMI® (aviptadil),
a sterile liquid for intravenous administration, was developed as an FDA Fast Track-designated, investigational drug for COVID-19
related respiratory failure. Aviptadil has previously been used in studies of Acute Respiratory Distress Syndrome (ARDS) and
other respiratory conditions. Although the Company’s Phase IIb/III clinical study in patients with Acute Respiratory Failure
in COVID-19 did not meet its primary endpoint of Alive and Free of Respiratory Failure at Day 60, data indicated a reduction
in mortality compared to placebo. However, the U.S. National Institutes of Health (“NIH”) recently halted recruitment
of a Phase III study of ZYESAMI for intravenous administration in patients with Critical COVID-19 (ACTIV-3b TESICO study)
due to futility. As a result, the Company is shifting focus away from COVID-19 efforts, reallocating resources in favor of NRX-101,
and will begin evaluating the options for ZYESAMI®
in protecting the lungs in other respiratory disorders, as well as its potential in other
therapeutic areas. Although the Company will evaluate the data from the NIH study when it becomes available, the likelihood and
timing of a submission of ZYESAMI for any New Drug Application cannot be ascertained at this time.
We have re-initiated
our psychiatry clinical work and expect to start a new registrational study of NRX-101 for severe bipolar depression in patients
with ASIB after initial stabilization with ketamine (NRX-100), using newly-manufactured commercial level material in the second
half of 2022. In addition, we have initiated a Phase II clinical study for bipolar depression with sub-acute suicidal ideation
and behavior (“SSIB”). Furthermore, we are evaluating the potential of NRX-101 in post-traumatic stress disorder
(“PTSD”), another area of unmet need which is also associated with suicidality. NRX-100 is ketamine, which is a generic
anesthetic, that is being used off-label in psychiatry. NRX-100 is part of a regimen of two sequential studies that we have agreed
to with the FDA as part of our SPA for NRX-101 in the treatment of severe bipolar depression with ASIB. NRX-100 is not required
as part of our outpatient protocol for treatment of SSIB.
Background
We were incorporated
as Big Rock Partners Acquisition Corp. (“BRPA”) on September 18, 2017. On May 24, 2021, BRPA consummated
the business combination (the “Business Combination”) with NeuroRx pursuant to the Agreement and Plan of Merger dated
December 13, 2020, as amended (the “Merger Agreement”), by and among our BRPA, NeuroRx and Big Rock Merger Corp
(“Merger Sub”). As a result of the Business Combination, NeuroRx became a wholly-owned subsidiary of BRPA, and BRPA
changed its name to NRX Pharmaceuticals, Inc. While BRPA was the legal acquirer of NeuroRx in the Business
TABLE
OF CONTENTS
Combination, NeuroRx is deemed to be the accounting
acquirer, and the historical financial statements of NeuroRx became the historical financial statements of BRPA upon the closing
of the Business Combination.
At the effective
time of the Business Combination (the “Effective Time”), each share of preferred stock and common stock of NeuroRx
issued and outstanding immediately prior to the Effective Time was converted into the right to receive 3.16 shares of our Common
Stock (or an aggregate of 44,419,279 shares) plus two earn out rights. The first earn out right was the right to receive 1.58
additional shares of our Common Stock (or an aggregate of 21,804,164 shares) if, prior to December 31, 2022, ZYESAMI receives
Emergency Use Authorization (“EUA”) by the FDA, and NRx submits and the FDA files for review a new drug application
for ZYESAMI (the “Earnout Shares Milestone”). The second earn out right was the right to receive approximately USD$5.00
per share of NeuroRx common stock (or an aggregate of approximately $88,800,000 after adjustments) upon the earlier to occur
of (a) FDA approval of ZYESAMI and the listing of ZYESAMI in the FDA’s “Orange Book”, and (b) FDA’s
approval of NRX-100 and NRX-101 and the listing of NRX- 100 and NRX-101 in the FDA’s “Orange Book”, in each
case prior to December 31, 2022 (the occurrence of either clauses (a) or (b), the “Earnout Cash Milestone”).
If the Earnout Shares Milestone is achieved, the shares of our Common Stock will be issued within five (5) business days
after the occurrence of the Earnout Shares Milestone. If the Earnout Cash Milestone is achieved, the Merger Agreement does not
require the earnout cash to be delivered to the former NeuroRx securityholders within any specified period of time, and the board
of directors of NRx (the “Board”) will use its good faith judgment to determine the date to pay such cash.
On August 23,
2021, we consummated a private placement (the “August Private Placement”) pursuant to the terms and conditions of
the Securities Purchase Agreement, dated as of August 19, 2021. At the closing of the August Private Placement, we issued
2,727,273 shares of Common Stock for a purchase price of $11.00 per share and preferred investment options to purchase up to
an aggregate of 2,727,273 shares of Common Stock for an exercise price of $12.00 per share. On August 23, 2021, we also
issued preferred investment options to purchase 136,364 shares of Common Stock for an exercise price of $13.75 per share to the
placement agents of the August Private Placement in lieu of certain professional fees associated with the August Private Placement
(the “August 2021 Placement Agent Options”).
On February 2,
2022, we consummated a private placement (the “Private Placement”) pursuant to the terms and conditions of the Securities
Purchase Agreement, dated as of January 30, 2022 (the “Purchase Agreement”), with the selling securityholders.
At the closing of the Private Placement, we issued 7,824,727 shares of Common Stock for a purchase price of $3.195 per share
and Investment Options to purchase up to an aggregate of 7,824,727 shares of Common Stock for an exercise price of $3.07 per
share.
On February 2,
2022, we issued placement agent options to purchase 391,236 shares of Common Stock for an exercise price of $3.99375 per share
to the placement agents of the Private Placement in lieu of certain professional fees associated with the Private Placement (the
“Placement Agent Options”). The investment options issued to the selling security holders and Placement Agent Options
issued to the placement agent may be exercised starting six months from the date of issuance.
The rights
of holders of our Common Stock and warrants are governed by our second amended and restated certificate of incorporation (the
“Charter”), our second amended and restated bylaws (the “Bylaws”), and the Delaware General Corporation
Law (the “DGCL”), and, in the case of the warrants, the Warrant Agreement, dated November 20, 2017, as amended,
between us and Continental Stock Transfer and Trust Company (the “Warrant Agreement”). See the “Description
of Capital Stock” section of this prospectus.
Summary of
Risk Factors
Our business
is subject to a number of risks of which you should be aware before making an investment decision. These risks are discussed
more fully in the “Risk Factors” section of our annual report on Form 10-K, filed with the SEC on March 31,
2022. Some of the risks related to our business and industry are summarized below. Such risks include, but are not limited to:
•
We have a limited operating history upon which to base an investment decision. We have not
been profitable historically and may not achieve or maintain profitability in the future.
TABLE
OF CONTENTS
•
We
need to raise additional capital to operate our business. If we fail to obtain the capital necessary to fund our operations,
we will be unable to continue or complete our product development.
•
NRX-100, NRX-101, and ZYESAMI are still in Phase IIb/III of clinical testing, and our
initial application to the FDA for Emergency Use Authorization for ZYESAMI was not granted.
•
We have not yet scaled manufacturing of our drug products to levels that are required for
sustained sales.
•
The Company has been, and may become involved in, disputes, claims, arbitration and litigation,
including our dispute with Relief Therapeutics.
•
If we fail to obtain or maintain FDA and other regulatory clearances for our products, or
if such clearances are delayed, we will be unable to commercially distribute and market our products in the U.S.
•
Our products will face significant competition in the markets for such products and future
products may never achieve market acceptance. We are faced with rapid technological change and developments by competitors may
render our products or technologies obsolete or non-competitive.
•
We do not anticipate obtaining orphan drug protection for the treatment of COVID-19.
•
Our business activities have been disrupted due to the outbreak of the COVID-19 pandemic.
Likewise, we are currently operating in a period of economic uncertainty and capital markets disruption, which has been significantly
impacted by geopolitical instability due to the ongoing military conflict between Russia and Ukraine.
•
Our relationships with customers and payors will be subject to applicable anti-kickback,
fraud and abuse, transparency, and other healthcare laws and regulations, which could expose us to criminal sanctions, civil
penalties, contractual damages, reputational harm, and administrative burdens.
•
Managing our growth as we expand operations may strain our resources.
•
Failure to achieve and maintain effective internal controls over financial reporting in accordance
with Section 404 of the Sarbanes-Oxley Act could impair our ability to produce timely and accurate financial statements
or comply with applicable regulations and have a material adverse effect on our business.
•
Even if a drug product is approved, the regulators may impose limitations on the use or marketing
of such product.
•
If we are unable to design, conduct and complete clinical trials successfully, our drug candidates
will not be able to receive regulatory approval. We cannot predict whether regulatory agencies will determine that the data from
our clinical trials of our product candidates supports marketing approval.
•
There is no guarantee that regulators will grant NDA approval of our current or future product
candidates.
•
If an adverse event occurs during a clinical trial, the regulators or an internal review
board may delay or terminate the trial.
•
Discussions and guidance of clinical trials are not binding obligations on the part of regulatory
authorities. The results of our current or future clinical trials may not support our product candidate claims or may result
in the discovery of unexpected adverse side effects.
•
Delays in the commencement or completion of pharmaceutical development, manufacturing or
clinical efficacy and safety testing could result in increased costs to us and delay our ability to generate revenues.
•
Even if our products are approved by regulatory authorities, if we or our suppliers fail
to comply with ongoing FDA regulation or if we experience unanticipated problems with our products, these products could be subject
to market restrictions or withdrawals.
•
Conducting clinical trials of our drug candidates or commercial sales of a drug candidate
may expose us to expensive product liability claims and we may not be able to maintain product liability insurance on reasonable
terms or at all.
TABLE
OF CONTENTS
•
The
use of a controlled substance in our NRX-100 drug candidate subjects us to DEA scrutiny and compliance, which may result in additional
expense and clinical delays, and may generate controversy. In addition, the use of controlled substances may limit the availability
of the active ingredients needed for NRX-100 and NRX-101.
•
We may need to focus our future efforts in new therapeutic areas where we have little or
no experience.
•
Modifications to our products may require new NDA approvals and some of our other product
candidates will require Risk Evaluation and Mitigation Strategies.
•
Our formulation of ZYESAMI is not covered by an issued patent and may be subject to future
generic competition. Our business relies on certain licensing rights that can be terminated in certain circumstances.
•
Our business depends upon securing and protecting critical intellectual property. Our patent
position is highly uncertain. If we are found to be infringing on patents or trade secrets owned by others, we may be forced
to cease or alter our product development efforts, obtain a license to continue development or sale of our products, and/or pay
damages.
•
Breaches by our employees or other parties may allow our trade secrets to become known to
our competitors.
•
We may not receive royalty or milestone revenue relating to our product candidates under
our collaboration and future license agreements for several years, or at all.
•
We do not have direct control of third parties performing preclinical and clinical trials.
If such third parties do not perform as contractually required or expected, we may not be able to obtain regulatory approval
for or commercialize our products.
•
We have no manufacturing capabilities and depend on other parties for manufacturing operations.
These manufacturers may fail to satisfy our requirements and applicable regulatory requirements.
•
Upon commercialization of our products, we may be dependent on third parties to market, distribute
and sell our products. We may not be successful in contracting with third parties for these services on favorable terms.
•
Our issuance of additional shares of Common Stock or convertible securities could make it
difficult for another company to acquire us, may dilute your ownership of us and could adversely affect our stock price. Future
sales, or the perception of sales, of our Common Stock by us or our existing stockholders could cause the market price for our
Common Stock to decline.
•
We qualify as an “emerging growth company” as well as a “smaller reporting
company” within the meaning of the Securities Act, which could make our securities less attractive to investors and may
make it more difficult to evaluate our performance.
•
Anti-takeover provisions in our governing documents and under Delaware law could make an
acquisition of us more difficult, limit attempts by our stockholders to replace or remove our current management and limit the
market price of our Common Stock.
•
Certain of our stockholders have effective control of NRx, and their interests may conflict
with NRx’s or yours in the future. We are no longer a “controlled company” under the corporate governance rules
of Nasdaq. However, during the applicable phase-in periods we may continue to rely on exemptions from certain corporate governance
standards.
•
The obligations associated with being a public company will involve significant expenses,
and will require significant resources and management attention, which may divert from our business operations.
•
We do not intend to pay dividends on our Common Stock for the foreseeable future.
Corporate
Information
We were incorporated
under the laws of the State of Delaware on September 18, 2017 under the name Big Rock Partners Acquisition Corp. (Nasdaq:
BRPA). Upon the closing of the Business Combination, we
TABLE
OF CONTENTS
changed our name to NRX Pharmaceuticals, Inc.
The Company does not maintain a principal executive office. Our registered office is located at 1201 Orange Street, Suite 600,
Wilmington, Delaware 19801 and our mailing address is at 1201 North Market Street, Suite 111, Wilmington, Delaware 19801. Our
telephone number is (484) 254-6134. Our website address is www.nrxpharma.com. The information contained in, or accessible
through, our website does not constitute a part of this prospectus. We have included our website address in this prospectus solely
as an inactive textual reference.
Implications
of Being an Emerging Growth Company
We qualify
as an “emerging growth company” as defined in Section 2(a)(19) of the Securities Act of 1933, as amended (the
“Securities Act”), as modified by the JOBS Act. As such, we will be eligible for and intend to take advantage of
certain exemptions from various reporting requirements applicable to other public companies that are not emerging growth companies
for as long as it continues to be an emerging growth company, including (a) the exemption from the auditor attestation requirements
with respect to internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act, (b) the exemptions
from say-on-pay, say-on-frequency and say-on-golden parachute voting requirements and (c) reduced disclosure obligations
regarding executive compensation in its periodic reports and proxy statements.
We will remain
an emerging growth company until December 31, 2022. In addition, Section 107 of the JOBS Act also provides that an
emerging growth company can take advantage of the exemption from complying with new or revised accounting standards provided
in Section 7(a)(2)(B) of the Securities Act as long as we are an emerging growth company. An emerging growth company can
therefore delay the adoption of certain accounting standards until those standards would otherwise apply to private companies.
We have elected not to opt out of such extended transition.
Investing in
our securities involves risk. Before you decide whether to purchase any of our securities, you should carefully consider the
specific risks discussed in, or incorporated by reference into, the applicable prospectus supplement, together with all the other
information contained in the prospectus supplement or incorporated by reference into this prospectus and the applicable prospectus
supplement. You should also consider the risks, uncertainties and assumptions discussed under the caption “Risk Factors”
included in our annual
report on Form 10-K, filed with the SEC on March 31, 2022, as supplemented and updated by subsequent quarterly reports
on Form 10-Q and current reports on Form 8-K that we have filed or will file with the SEC, which are incorporated by reference
into this prospectus. These risk factors may be amended, supplemented or superseded from time to time by other reports we file
with the SEC in the future. For more information, please see “Where You Can Find More Information” and “Incorporation
by Reference.” These risks could materially and adversely affect our business, results of operations and financial condition
and could result in a partial or complete loss of your investment.
Unless
we specify another use in the applicable prospectus supplement, we will use the net proceeds from the sale of the securities
offered by us for general corporate purposes, which may include, among other things, general corporate expenses, funding of clinical
trial programs and working capital purposes. We may also use such proceeds to fund acquisitions of businesses or technologies
that complement our current business. We may set forth additional information on the use of net proceeds from the sale of the
securities we offer under this prospectus in a prospectus supplement related to a specific offering.
DESCRIPTION
OF CAPITAL STOCK
General
The following
description summarizes some of the terms of our Charter and Bylaws and of the DGCL. This description is summarized from, and
qualified in its entirety by reference to, our Charter and Bylaws, each of which has been publicly filed with the SEC, as well
as the relevant provisions of the DGCL.
Capital Stock
Our authorized
capital stock consists of 500,000,000 shares of Common Stock, par value $0.001 per share, and 50,000,000 shares of preferred
stock, par value $0.001 per share. As of May 11, 2022, there were 66,641,314 shares of Common Stock outstanding. No shares
of preferred stock have been issued or are outstanding. Unless our Board determines otherwise, we will issue all shares of our
capital stock in uncertificated form.
Common Stock
Holders of
shares of Common Stock are entitled to one vote for each share held of record on all matters submitted to a vote of stockholders.
The holders of Common Stock do not have cumulative voting rights in the election of directors.
In the event
of our liquidation, dissolution or winding up and after payment in full of all amounts required to be paid to creditors and to
any future holders of preferred stock having liquidation preferences, if any, the holders of Common Stock will be entitled to
receive pro rata our remaining assets available for distribution. Holders of Common Stock do not have preemptive, subscription,
redemption or conversion rights. There are no redemption or sinking fund provisions applicable to the Common Stock. The rights,
powers, preferences and privileges of holders of the Common Stock are subject to those of the holders of any shares of preferred
stock that the Board may authorize and issue in the future.
Preferred
Stock
Under the
terms of the Charter, our Board is authorized to direct us to issue shares of preferred stock in one or more series without stockholder
approval. Our Board has the discretion to determine the rights, powers, preferences, privileges and restrictions, including voting
rights, dividend rights, conversion rights, redemption privileges and liquidation preferences, of each series of preferred stock.
The purpose
of authorizing our Board to issue preferred stock and determine its rights and preferences is to eliminate delays associated
with a stockholder vote on specific issuances. The issuance of preferred stock, while providing flexibility in connection with
possible acquisitions, future financings and other corporate purposes, could have the effect of making it more difficult for
a third party to acquire, or could discourage a third party from seeking to acquire, a majority of the outstanding voting stock.
Additionally, the issuance of preferred stock may adversely affect the holders of Common Stock by restricting dividends on the
Common Stock, diluting the voting power of the Common Stock or subordinating the liquidation rights of the Common Stock. As a
result of these or other factors, the issuance of preferred stock could have an adverse impact on the market price of the Common
Stock. No shares of preferred stock have been issued.
Dividends
Declaration
and payment of any dividend is subject to the discretion of our Board. The time and amount of dividends is dependent upon, among
other things, our business prospects, results of operations, financial condition, cash requirements and availability, debt repayment
obligations, capital expenditure needs, contractual restrictions, covenants in the agreements governing current and future indebtedness,
industry trends, the provisions of Delaware law affecting the payment of dividends and distributions to stockholders and any
other factors or considerations our Board may regard as relevant.
We currently
intend to retain all available funds and any future earnings to fund the development and growth of our business, and therefore
do not anticipate declaring or paying any cash dividends on Common Stock in the foreseeable future.
TABLE
OF CONTENTS
Anti-Takeover Provisions
The Charter
and Bylaws contain provisions that may delay, defer or discourage another party from acquiring control of us. We expect that
these provisions, which are summarized below, will discourage coercive takeover practices or inadequate takeover bids.
These provisions
are also designed to encourage persons seeking to acquire control of us to first negotiate with our Board, which may result in
an improvement of the terms of any such acquisition in favor of our stockholders. However, they also give our Board the power
to discourage acquisitions that some stockholders may favor.
Authorized
but Unissued Shares
The authorized
but unissued shares of Common Stock and preferred stock are available for future issuance without stockholder approval, subject
to any limitations imposed by the listing standards of Nasdaq. These additional shares may be used for a variety of corporate
finance transactions, acquisitions and employee benefit plans. The existence of authorized but unissued and unreserved Common
Stock and preferred stock could make more difficult or discourage an attempt to obtain control of us by means of a proxy contest,
tender offer, merger or otherwise.
Classified
Board of Directors
Our Charter
provides that our Board is divided into three classes of directors, with the classes to be as nearly equal in number as possible,
and with each director serving a three-year term. As a result, approximately one-third of our Board will be elected each year.
The classification of directors has the effect of making it more difficult for stockholders to change the composition of our
Board.
Stockholder
Action; Special Meetings of Stockholders
Our Charter
provides that, unless Jonathan Javitt and Daniel Javitt own at least a majority of the shares of the Common Stock, stockholders
may not take action by written consent, but may only take action at annual or special meetings of stockholders. As a result,
a holder controlling a majority of capital stock would not be able to amend the Bylaws or remove directors without holding a
meeting of stockholders called in accordance with the Bylaws. Further, our Charter provides that only the chairperson of our
Board, a majority of our Board, our Chief Executive Officer or our President may call special meetings of stockholders, thus
prohibiting a stockholder from calling a special meeting. These provisions might delay the ability of stockholders to force consideration
of a proposal or for stockholders controlling a majority of capital stock to take any action, including the removal of directors.
Advance Notice
Requirements for Stockholder Proposals and Director Nominations
In addition,
our Bylaws establish an advance notice procedure for stockholder proposals to be brought before an annual meeting or special
meeting of stockholders. Generally, in order for any matter to be “properly brought” before a meeting, the matter
must be (a) specified in a notice of meeting given by or at the direction of our Board, (b) if not specified in a notice
of meeting, otherwise brought before the meeting by our Board or the chairperson of the meeting, or (c) otherwise properly
brought before the meeting by a stockholder present in person who (i) was a stockholder both at the time of giving the notice
and at the time of the meeting, (ii) is entitled to vote at the meeting, and (iii) has complied with the advance notice
procedures specified in our Bylaws or properly made such proposal in accordance with Rule 14a-8 under the Exchange Act and
the rules and regulations thereunder, which proposal has been included in the proxy statement for the annual meeting. Further,
for business to be properly brought before an annual meeting by a stockholder, the stockholder must (a) provide Timely Notice
in writing and in proper form to the secretary and (b) provide any updates or supplements to such notice at the times and
in the forms required by our Bylaws. To be timely, a stockholder’s notice must be delivered to, or mailed and received
at, our principal executive offices not less than 90 days nor more than 120 days prior to the one-year anniversary
of the preceding year’s annual meeting; provided, however, that if the date of the annual meeting is more than 30 days
before or more than 60 days after such anniversary date, notice by the stockholder to be timely must be so delivered, or
mailed and received, not later than the 90th day prior to such annual meeting or, if later, the
TABLE
OF CONTENTS
10th day following the day on which public
disclosure of the date of such annual meeting was first made (such notice within such time periods, “Timely Notice”).
Stockholders
at an annual meeting or special meeting may only consider proposals or nominations specified in the notice of meeting or brought
before the meeting by or at the direction of our Board or by a qualified stockholder of record on the record date for the meeting,
who is entitled to vote at the meeting and who has delivered timely written notice in proper form to our secretary of the stockholder’s
intention to bring such business before the meeting. These provisions could have the effect of delaying stockholder actions that
are favored by the holders of a majority of the outstanding voting securities until the next stockholder meeting.
Amendment
of Charter or Bylaws
Our Bylaws
may be amended or repealed by a majority vote of our Board or by the holders of at least sixty-six and two-thirds percent
(662∕3%)
of the voting power of all of the then-outstanding shares entitled to vote generally in the election of directors, voting together
as a single class. The affirmative vote of a majority of our Board and at least sixty-six and two-thirds percent (662∕3%)
in voting power of the outstanding shares entitled to vote would be required to amend certain provisions of our Charter.
Limitations
on Liability and Indemnification of Officers and Directors
Our Charter
and Bylaws provide indemnification and advancement of expenses for our directors and officers to the fullest extent permitted
by the DGCL, subject to certain limited exceptions. We have entered into indemnification agreements with each of our directors
and officers. In some cases, the provisions of those indemnification agreements may be broader than the specific indemnification
provisions contained under Delaware law. In addition, as permitted by Delaware law, our Charter and Bylaws include provisions
that eliminate the personal liability of directors for monetary damages resulting from breaches of certain fiduciary duties as
a director. The effect of this provision is to restrict our rights and the rights of our stockholders in derivative suits to
recover monetary damages against a director for breach of fiduciary duties as a director. These provisions may be held not to
be enforceable for violations of the federal securities laws of the United States.
Dissenters’
Rights of Appraisal and Payment
Under the
DGCL, with certain exceptions, our stockholders have appraisal rights in connection with a merger or consolidation of our company.
Pursuant to Section 262 of the DGCL, stockholders who properly demand and perfect appraisal rights in connection with such
merger or consolidation have the right to receive payment of the fair value of their shares as determined by the Delaware Court
of Chancery.
Stockholders’
Derivative Actions
Under the
DGCL, any of our stockholders may bring an action in our name to procure a judgment in its favor, also known as a derivative
action, provided that the stockholder bringing the action is a holder of the our shares at the time of the transaction to which
the action relates.
Forum Selection
Our Charter
and Bylaws provide that unless we consent in writing to the selection of an alternative forum, the (a) Chancery Court of
the State of Delaware (or, in the event that the Chancery Court does not have jurisdiction, the federal district court for the
District of Delaware or other state courts of the State of Delaware) shall, to the fullest extent permitted by law, be the sole
and exclusive forum for: (i) any derivative action brought by a stockholder on our behalf, (ii) any action, suit or
proceeding asserting a claim of breach of fiduciary duty owed by any of our directors, officers, stockholders to us or to our
stockholders, (iii) any action, suit or proceeding asserting a claim arising pursuant to the DGCL, the Charter or the Bylaws,
or (iv) any action, suit or proceeding asserting a claim governed by the internal affairs doctrine; and (b) subject
to the foregoing, the federal district courts of the United States of America shall be the exclusive forum for the resolution
of any complaint asserting a cause of action arising under the Securities Act. Notwithstanding the foregoing, such forum selection
provisions shall not apply to suits brought to enforce any liability or duty
TABLE
OF CONTENTS
created by the Exchange Act or any other claim
for which the federal courts of the United States have exclusive jurisdiction. The choice of forum provision may limit a stockholder’s
ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors, officers, or other
employees, which may discourage such lawsuits against us and our directors, officers, and other employees. Alternatively, if
a court were to find the choice of forum provision contained in the Charter to be inapplicable or unenforceable in an action,
we may incur additional costs associated with resolving such action in other jurisdictions, which could harm our business, results
of operations, and financial condition.
Additionally,
Section 22 of the Securities Act creates concurrent jurisdiction for federal and state courts over all suits brought to
enforce any duty or liability created by the Securities Act or the rules and regulations thereunder. As noted above, the Charter
and the Bylaws will provide that the federal district courts of the United States of America shall have jurisdiction over any
action arising under the Securities Act. Accordingly, there is uncertainty as to whether a court would enforce such provision.
Our stockholders will not be deemed to have waived our compliance with the federal securities laws and the rules and regulations
thereunder.
Transfer Agent
and Registrar
The transfer
agent and registrar for the Common Stock is Continental Stock Transfer & Trust Company, One State Street Plaza, New York,
New York 10004.
Trading Symbol
and Market
Our Common
Stock is listed on Nasdaq under the symbol “NRXP” and certain of our warrants are listed on Nasdaq under the symbol
“NRXPW”.
DESCRIPTION
OF THE DEBT SECURITIES
The following
description of the terms of the debt securities sets forth certain general terms and provisions of the debt securities to which
any prospectus supplement may relate. The particular terms of the debt securities offered by any prospectus supplement and the
extent, if any, to which these general provisions may apply to those debt securities will be described in the prospectus supplement
relating to those debt securities. Accordingly, for a description of the terms of a particular issue of debt securities, reference
must be made to both the prospectus supplement relating thereto and to the following description.
We may issue
debt securities from time to time in one or more series. The debt securities will be general obligations of NRx Pharmaceuticals,
Inc. The debt securities may be fully and unconditionally guaranteed on a secured or unsecured senior or subordinated basis,
jointly and severally, by guarantors, if any. In the event that any series of debt securities will be subordinated to other indebtedness
that we have outstanding or may incur, the terms of the subordination will be set forth in the prospectus supplement relating
to the subordinated debt securities. Debt securities will be issued under one or more indentures between us and a trustee. The
following discussion of certain provisions of the indenture is a summary only and should not be considered a complete description
of the terms and provisions of the indenture. Accordingly, the following discussion is qualified in its entirety by reference
to the provisions of the indenture, including the definition of certain terms used below. You should refer to the indenture for
the complete terms of the debt securities.
General
The debt securities
will represent direct, general obligations of NRx Pharmaceuticals, Inc. and:
•
may rank equally with other unsubordinated debt or may be subordinated to other debt we have
or may incur;
•
may be issued in one or more series with the same or various maturities;
•
may be issued at a price of 100% of their principal amount or at a premium or discount;
•
may be issued in registered or bearer form and certificated or uncertificated form; and
•
may be represented by one or more global debt securities registered in the name of a designated
depositary’s nominee, and if so, beneficial interests in the global note will be shown on and transfers will be made only
through records maintained by the designated depositary and its participants.
The aggregate
principal amount of debt securities that we may authenticate and deliver is unlimited. Subject to limitations contained in the
indenture, we may from time to time, without notice to or the consent of the holders of a series of debt securities, issue additional
debt securities of any such series on the same terms and conditions as the debt securities of such series, except for any differences
in the issue price and, if applicable, the initial interest accrual date and interest payment date; provided that if the
additional debt securities are not fungible with the debt securities of such series for U.S. federal income tax purposes, such
additional debt securities will have one or more separate CUSIP numbers. You should refer to the applicable prospectus supplement
for the following terms of the debt securities of the series with respect to which that prospectus supplement is being delivered:
•
the title of the debt securities of the series (which will distinguish the debt securities
of that particular series from the debt securities of any other series) and ranking (including the terms of any subordination
provisions);
•
the price or prices of the debt securities of the series at which such debt securities will
be issued;
•
whether the debt securities are entitled to the benefit of any guarantee by any guarantor;
•
any limit on the aggregate principal amount of the debt securities of the series that may
be authenticated and delivered under the indenture (except for debt securities authenticated and delivered upon registration
or transfer of, or in exchange for, or in lieu of, other debt securities of the series);
•
the date or dates on which the principal and premium with respect to the debt securities
of the series are payable;
TABLE
OF CONTENTS
•
the
person to whom any interest on a security of the series shall be payable if other than the person in whose name that security
is registered at the close of business on the record date;
•
the rate or rates (which may be fixed or variable) at which the debt securities of the series
will bear interest (if any) or the method of determining such rate or rates (including, but not limited to, any commodity, commodity
index, stock exchange index or financial index), the date or dates from which such interest, if any, will accrue, the interest
payment dates on which such interest, if any, will be payable or the method by which such dates will be determined, the record
dates for the determination of holders thereof to whom such interest is payable (in the case of securities in registered form),
and the basis upon which interest will be calculated if other than that of a 360-day year of twelve 30-day months;
•
the currency or currencies in which debt securities of the series will be denominated and/or
in which payment of the principal, premium, if any, and interest of any of the securities shall be payable, if other than U.S.
dollars, the place or places, if any, in addition to or instead of the corporate trust office of the trustee (in the case of
securities in registered form) where the principal, premium and interest, if any, with respect to debt securities of the series
will be payable, where notices and demands to or upon us in respect of the debt securities and the indenture may be delivered,
and the method of such payment, if by wire transfer, mail or other means;
•
the price or prices at which, the period or periods within which, and the terms and conditions
upon which debt securities of the series may be redeemed, in whole or in part, at our option or otherwise;
•
whether debt securities of the series are to be issued as securities in registered form or
securities in bearer form or both and, if securities in bearer form are to be issued, whether coupons will be attached to them,
whether securities in bearer form of the series may be exchanged for securities in registered form of the series, and the circumstances
under which and the places at which any such exchanges, if permitted, may be made;
•
if any debt securities of the series are to be issued as securities in bearer form or as
one or more global securities representing individual securities in bearer form of the series, whether certain provisions for
the payment of additional interest or tax redemptions will apply; whether interest with respect to any portion of a temporary
bearer security of the series payable with respect to any interest payment date prior to the exchange of such temporary bearer
security for definitive securities in bearer form of the series will be paid to any clearing organization with respect to the
portion of such temporary bearer security held for its account and, in such event, the terms and conditions (including any certification
requirements) upon which any such interest payment received by a clearing organization will be credited to the persons entitled
to interest payable on such interest payment date; and the terms upon which a temporary bearer security may be exchanged for
one or more definitive securities in bearer form of the series;
•
the obligation or right, if any, to redeem, purchase or repay debt securities of the series
pursuant to any sinking fund or analogous provisions or at the option of a holder of such debt securities and the price or prices
at which, the period or periods within which, and the terms and conditions upon which, debt securities of the series will be
redeemed, purchased or repaid, in whole or in part, pursuant to such obligations;
•
the terms, if any, upon which the debt securities of the series may be convertible into or
exchanged for any issuer’s common stock, preferred stock, depositary shares, other debt securities or warrants for common
stock, preferred stock, depositary shares, indebtedness or other securities of any kind and the terms and conditions upon which
such conversion or exchange will be effected, including the initial conversion or exchange price or rate, the conversion or exchange
period and any other additional provisions;
•
if other than minimum denominations of $2,000 or any integral multiple of $1,000 in excess
thereof, the denominations in which debt securities of the series will be issuable;
•
if the amount of principal, premium or interest with respect to the debt securities of the
series may be determined with reference to an index or pursuant to a formula, the manner in which such amounts will be determined;
TABLE
OF CONTENTS
•
if
the principal amount payable at the stated maturity of debt securities of the series will not be determinable as of any one or
more dates prior to such stated maturity, the amount that will be deemed to be such principal amount as of any such date for
any purpose, including the principal amount thereof which will be due and payable upon any maturity other than the stated maturity
or which will be deemed to be outstanding as of any such date (or, in any such case, the manner in which such deemed principal
amount is to be determined), and if necessary, the manner of determining the equivalent thereof in U.S. dollars;
•
any changes or additions to the provisions of the indenture dealing with defeasance;
•
if other than the principal amount thereof, the portion of the principal amount of debt securities
of the series that will be payable upon declaration of acceleration of the maturity thereof or provable in bankruptcy;
•
the terms, if any, of the transfer, mortgage, pledge or assignment as security for the debt
securities of the series of any properties, assets, moneys, proceeds, securities or other collateral and any corresponding changes
to provisions of the indenture as then in effect;
•
any addition to or change in the events of default with respect to the debt securities of
the series and any change in the right of the trustee or the holders to declare the principal, premium and interest, if any,
with respect to such debt securities due and payable;
•
if the debt securities of the series will be issued in whole or in part in the form of a
global security, the terms and conditions, if any, upon which such global security may be exchanged in whole or in part for other
individual debt securities in definitive registered form, the depositary (as defined in the applicable prospectus supplement)
for such global security and the form of any legend or legends to be borne by any such global security in addition to or in lieu
of the legend referred to in the indenture;
•
any trustee, authenticating or paying agent, transfer agent or registrar or any other agent
with respect to the debt securities;
•
the applicability of, and any addition to, deletion of or change in, the covenants and definitions
then set forth in the indenture or in the terms then set forth in the indenture relating to permitted consolidations, mergers
or sales of assets;
•
the terms, if any, of any guarantee of the payment of principal, premium and interest with
respect to debt securities of the series and any corresponding changes to the provisions of the indenture as then in effect;
•
the subordination, if any, of the debt securities of the series pursuant to the indenture
and any changes or additions to the provisions of the indenture relating to subordination;
•
with regard to debt securities of the series that do not bear interest, the dates for certain
required reports to the trustee;
•
any provisions granting special rights to holders when a specified event occurs;
•
any co-issuer;
•
the place or places where the principal of and interest, if any, on the debt securities will
be payable, where the debt securities may be surrendered for registration of transfer or exchange and where notices and demands
to or upon us in respect of the debt securities and the indenture may be served, and the method of such payment, if by wire transfer,
mail or other means; and
•
any other terms of the debt securities of the series (which terms will not be prohibited
by the provisions of the indenture).
TABLE
OF CONTENTS
The prospectus supplement
will also describe any material U.S. federal income tax consequences or other special considerations applicable to the series
of debt securities to which such prospectus supplement relates, including those applicable to:
•
securities in bearer form;
•
debt securities with respect to which payments of principal, premium or interest are determined
with reference to an index or formula (including changes in prices of particular securities, currencies or commodities);
•
debt securities with respect to which principal or interest is payable in a foreign or composite
currency;
•
debt securities that are issued at a discount below their stated principal amount, bearing
no interest or interest at a rate that at the time of issuance is below market rates or original issue discount debt securities;
and
•
variable rate debt securities that are exchangeable for fixed rate debt securities.
Unless otherwise
provided in the applicable prospectus supplement, securities in registered form may be transferred or exchanged at the office
of the trustee at which its corporate trust business is principally administered in the United States, subject to the limitations
provided in the indenture, without the payment of any service charge, other than any tax or governmental charge payable in connection
therewith. Securities in bearer form will be transferable only by delivery. Provisions with respect to the exchange of securities
in bearer form will be described in the prospectus supplement relating to those securities in bearer form.
All funds
that we pay to a paying agent for the payment of principal, premium or interest with respect to any debt securities that remain
unclaimed at the end of two years after that principal, premium or interest will have become due and payable will be repaid
to us, and the holders of those debt securities or any related coupons will thereafter look only to us for payment thereof.
Global Securities
The debt
securities of a series may be issued in whole or in part in the form of one or more global securities. A global security is a
debt security that represents, and is denominated in an amount equal to the aggregate principal amount of, all outstanding debt
securities of a series, or any portion thereof, in either case having the same terms, including the same original issue date,
date or dates on which principal and interest are due, and interest rate or method of determining interest. A global security
will be deposited with, or on behalf of, a depositary, which will be identified in the prospectus supplement relating to such
debt securities. Global securities may be issued in either registered or bearer form and in either temporary or definitive form.
Unless and until it is exchanged in whole or in part for the individual debt securities represented thereby, a global security
may not be transferred except as a whole by the depositary to a nominee of the depositary, by a nominee of the depositary to
the depositary or another nominee of the depositary, or by the depositary or any nominee of the depositary to a successor depositary
or any nominee of such successor.
The terms
of the depositary arrangement with respect to a series of debt securities will be described in the prospectus supplement relating
to such debt securities. We anticipate that the following provisions will generally apply to depositary arrangements, in all
cases subject to any restrictions or limitations described in the prospectus supplement relating to such debt securities.
Upon the
issuance of a global security, the depositary for such global security will credit, on its book entry registration and transfer
system, the respective principal amounts of the individual debt securities represented by such global security to the accounts
of persons that have accounts with the depositary. Such accounts will be designated by the dealers or underwriters with respect
to such debt securities or, if such debt securities are offered and sold directly by us or through one or more agents, by us
or such agents. Ownership of beneficial interests in a global security will be limited to participants or persons that hold beneficial
interests through participants. Ownership of beneficial interests in such global security will be shown on, and the transfer
of that ownership will be effected only through, records maintained by the depositary (with respect to interests of participants)
or records maintained by participants (with respect to interests of persons other than participants). The laws of some states
require that certain purchasers of
TABLE
OF CONTENTS
securities take physical delivery of such securities
in definitive form. Such limitations and laws may impair the ability to transfer beneficial interests in a global security.
So long as
the depositary for a global security, or its nominee, is the registered owner or holder of such global security, such depositary
or nominee, as the case may be, will be considered the sole owner or holder of the individual debt securities represented by
such global security for all purposes under the indenture. Except as provided below, owners of beneficial interests in a global
security will not be entitled to have any of the individual debt securities represented by such global security registered in
their names, will not receive or be entitled to receive physical delivery of any of such debt securities in definitive form and
will not be considered the owners or holders thereof under the indenture.
Payments of
principal, premium and interest with respect to individual debt securities represented by a global security will be made to the
depositary or its nominee, as the case may be, as the registered owner or holder of such global security. Neither we, the trustee,
any paying agent or registrar for such debt securities nor any agent of ours or the trustee will have any responsibility or liability
for:
•
any aspect of the records relating to or payments made by the depositary, its nominee or
any participants on account of beneficial interests in the global security or for maintaining, supervising or reviewing any records
relating to such beneficial interests;
•
the payment to the owners of beneficial interests in the global security of amounts paid
to the depositary or its nominee; or
•
any other matter relating to the actions and practices of the depositary, its nominee or
its participants.
Neither we,
the trustee, any paying agent or registrar for such debt securities nor any agent of ours or the trustee will be liable for any
delay by the depositary, its nominee or any of its participants in identifying the owners of beneficial interests in the global
security, and we and the trustee may conclusively rely on, and will be protected in relying on, instructions from the depositary
or its nominee for all purposes.
We expect that
the depositary for a series of debt securities or its nominee, upon receipt of any payment of principal, premium or interest
with respect to a definitive global security representing any of such debt securities, will immediately credit participants’
accounts with payments in amounts proportionate to their respective beneficial interests in the principal amount of such global
security, as shown on the records of the depositary or its nominee. We also expect that payments by participants to owners of
beneficial interests in such global security held through such participants will be governed by standing instructions and customary
practices, as is now the case with securities held for the accounts of customers and registered in “street name.”
Such payments will be the responsibility of such participants. See “— Limitations on Issuance of Securities in Bearer
Form” below.
If the depositary
for a series of debt securities is at any time unwilling, unable or ineligible to continue as depositary, we will appoint a successor
depositary. If a successor depositary is not appointed by us within 90 days, we will issue individual debt securities of
such series in exchange for the global security representing such series of debt securities. In addition, we may at any time
and in our sole discretion determine to no longer have debt securities of a series represented by a global security and, in such
event, will issue individual debt securities of such series in exchange for the global security representing such series of debt
securities. Furthermore, if we so specify with respect to the debt securities of a series, an owner of a beneficial interest
in a global security representing debt securities of such series may, on terms acceptable to us, the trustee and the depositary
for such global security, receive individual debt securities of such series in exchange for such beneficial interests. In any
such instance, an owner of a beneficial interest in a global security will be entitled to physical delivery of individual debt
securities of the series represented by such global security equal in principal amount to such beneficial interest and to have
such debt securities registered in its name (if the debt securities are issuable as securities in registered form). Individual
debt securities of such series so issued generally will be issued:
•
as securities in registered form in minimum denominations, unless otherwise specified by
us, of $2,000 and any integral multiples of $1,000 in excess thereof if the debt securities are issuable as securities in registered
form;
TABLE
OF CONTENTS
•
as
securities in bearer form in the denomination or denominations specified by us if the debt securities are issuable as securities
in bearer form; or
•
as either securities in registered form or securities in bearer form as described above if
the debt securities are issuable in either form.
Limitations
on Issuance of Securities in Bearer Form
The debt
securities of a series may be issued as securities in registered form (which will be registered as to principal and interest
in the register maintained by the registrar for such debt securities) or securities in bearer form (which will be transferable
only by delivery). If such debt securities are issuable as securities in bearer form, the applicable prospectus supplement will
describe certain special limitations and considerations that will apply to such debt securities.
Certain Covenants
If debt securities
are issued, the indenture, as supplemented for a particular series of debt securities, will contain certain covenants for the
benefit of the holders of such series of debt securities, which will be applicable (unless waived or amended) so long as any
of the debt securities of such series are outstanding, unless stated otherwise in the prospectus supplement. The specific terms
of the covenants, and summaries thereof, will be set forth in the prospectus supplement relating to such series of debt securities.
Subordination
Debt securities
of a series and any guarantees, may be subordinated, which we refer to as subordinated debt securities, to senior indebtedness
(as defined in the applicable prospectus supplement) to the extent set forth in the prospectus supplement relating thereto. To
the extent we conduct operations through subsidiaries, the holders of debt securities (whether or not subordinated debt securities)
will be structurally subordinated to the creditors of our subsidiaries, except to the extent such subsidiary is a guarantor of
such series of debt securities.
Events of
Default
Each of the
following will constitute an event of default under the form of indenture with respect to any series of debt securities:
•
default in payment of the principal amount of the debt securities of that series, when such
amount becomes due and payable at maturity, upon acceleration, required redemption or otherwise;
•
failure to pay interest on the debt securities of that series within 30 days of the
due date;
•
failure to comply with the obligations described under “— Mergers and Sales of
Assets” below;
•
failure to comply for 90 days after notice with any of our other agreements in the debt
securities of that series or the indenture or supplemental indenture related to that series of debt securities; or
•
certain events of bankruptcy, insolvency or reorganization affecting us.
A prospectus
supplement may omit, modify or add to the foregoing events of default.
An event of
default under one series of debt securities does not necessarily constitute an event of default under any other series of debt
securities. A default under the fourth bullet above will not constitute an event of default until the trustee notifies us or
the holders of 30% in principal amount of the outstanding debt securities of such series notify us and the trustee of the default
and we do not cure such default within the time specified after receipt of such notice.
If any event
of default (other than an event of default relating to certain events of bankruptcy, insolvency or reorganization) occurs and
is continuing with respect to a particular series of debt securities, either the trustee or the holders of not less than 30%
in aggregate principal amount of the debt securities of that series then outstanding by written notice to us (and to the trustee
if such notice is given by the holders), may declare the principal amount of (or in the case of original issue discount debt
securities, the portion
TABLE
OF CONTENTS
thereby specified in the terms thereof), and
accrued interest on the debt securities of that series to be immediately due and payable. In the case of certain events of bankruptcy,
insolvency or reorganization, the principal amount of, and accrued interest on the debt securities of that series will automatically
become and be immediately due and payable without any declaration or other act on the part of the trustee or any holders. Upon
a declaration by the trustee or the holders, we will be obligated to pay the principal amount plus accrued and unpaid interest
of each affected series of debt securities so declared due and payable.
The holders
of a majority in aggregate principal amount of the debt securities of any series then outstanding by notice to the trustee under
the indenture may on behalf of the holders of all of such series of debt securities waive any existing default or event of default
and its consequences under the applicable indenture except a continuing default or event of default in the payment of interest
on, or the principal of, the debt securities of such series.
Subject to
the provisions of the indenture relating to the duties of the trustee in case an event of default will occur and be continuing,
the trustee will be under no obligation to exercise any of its rights or powers under the indenture or debt securities at the
request or direction of any of the holders of any series of debt securities, unless such holders have offered to the trustee
indemnity or security satisfactory to the trustee against any cost, loss, liability or expense. Subject to such provisions for
the indemnification of the trustee, the holders of at least a majority in aggregate principal amount of the outstanding debt
securities of a series have the right to direct the time, method and place of conducting any proceeding for any remedy available
to the trustee or exercising any trust or power conferred on the trustee with respect to such series of debt securities. The
trustee, however, may refuse to follow any direction that conflicts with law or the indenture or that the trustee determines
is unduly prejudicial to the rights of any other holder of such series of debt securities (it being understood that the trustee
does not have an affirmative duty to ascertain whether or not any such direction unduly prejudices the rights of such holders)
or that would involve the trustee in personal liability. Prior to taking any action under the indenture, the trustee will be
entitled to indemnification satisfactory to it in its sole discretion against all costs, losses, liabilities and expenses caused
by taking or not taking such action.
Except to
enforce the right to receive payment of principal, premium, if any, or interest when due, no holder of debt securities of a series
has any right to institute any proceeding with respect to the indenture or debt securities, or for the appointment of a receiver
or a trustee, or for any other remedy thereunder, unless:
•
such holder has previously given to the trustee written notice of a continuing event of default
with respect to such series of debt securities;
•
the holder or holders of at least 30% in aggregate principal amount of the outstanding debt
securities of that series have made written request, and such holder or holders have offered security or indemnity satisfactory
to the trustee against any loss, liability or expense, to the trustee to institute such proceeding as trustee; and
•
the trustee has failed to institute such proceeding, and has not received from the holders
of a majority in aggregate principal amount of the outstanding debt securities of that series a direction inconsistent with such
request, within 60 days after such notice, request and offer.
However,
such limitations do not apply to a suit instituted by a holder of a debt security of such series for the enforcement of payment
of the principal, premium, if any, or interest on such debt security on or after the applicable due date specified in such debt
security.
The indenture
will provide that if a default with respect to a series of debt securities occurs and is continuing and is actually known to
a trust officer of the trustee, the trustee must send to each holder of such debt securities notice of the default within 90 days
after it is actually known to a trust officer of the trustee. Except in the case of a default in the payment of the principal
or premium, if any, upon acceleration, redemption or otherwise with respect to any debt security of a series when such amount
becomes due and payable, the trustee may withhold notice if and so long as a committee of its trust officers in good faith determines
that withholding notice is not opposed to the interests of the holders.
The indenture
will require us to furnish to the trustee, within 120 days after the end of each fiscal year, a statement by certain of
our officers as to whether or not we, to their knowledge, are in default in the
TABLE
OF CONTENTS
performance or observance of any of the terms,
provisions and conditions of the indenture and, if so, specifying all such known defaults. We will also be required to deliver
to the trustee, within 30 days after the occurrence thereof, written notice of any event which would constitute a default;
provided, however, that failure to provide such written notice will not in and of itself result in a default under
the indenture.
Street name
and other indirect holders should consult their banks and brokers for information on their requirements for giving notice or
taking other actions upon a default.
Modification
and Waiver
Subject to
certain exceptions, modifications and amendments of the indenture, any supplemental indenture and any series of debt securities
may be made by us and the trustee with the consent of the holders of at least a majority in aggregate principal amount of the
outstanding debt securities of any series affected by such modification or amendment.
No such modification
or amendment may, without the consent of each holder affected thereby:
•
reduce the percentage of principal amount of the outstanding debt securities, the consent
of whose holders is required for any amendment;
•
reduce the principal amount of, or interest on, or extend the Stated Maturity or interest
payment periods of, any debt securities;
•
change the provisions applicable to the redemption of any debt securities;
•
make any debt securities payable in money or securities other than those stated in the debt
securities;
•
impair the contractual right of any holder of the debt securities to receive payment of principal
of and interest on such holder’s debt securities on or after the due dates therefor or to institute suit for the enforcement
of any payment on or with respect to such holder’s debt securities;
•
except as otherwise provided as described under “— Satisfaction and Discharge”
and “— Defeasance” herein, release any security or guarantee that may have been granted with respect to any
debt securities;
•
in the case of any subordinated securities, or coupons appertaining thereto, make any change
in the provisions of the indenture relating to subordination that adversely affects the rights of any holder under such provisions
(including any contractual subordination of senior unsubordinated debt securities); or
•
make any change in the amendment provisions which require each holder’s consent or
in the waiver provisions
Without the
consent of any holder, we and the trustee may amend the indenture for one or more of the following purposes:
•
to cure any ambiguity, omission, defect or inconsistency;
•
to surrender any right or power conferred upon the Company by the indenture, to add to the
covenants of the Company such further covenants, restrictions, conditions or provisions for the protection of the holders of
all or any series of debt securities as the board of directors of the Company will consider to be for the protection of the holders
of such debt securities, and to make the occurrence, or the occurrence and continuance, of a default in respect of any such additional
covenants, restrictions, conditions or provisions a default or an event of default under the indenture; provided, however,
that with respect to any such additional covenant, restriction, condition or provision, such amendment may provide for a period
of grace after default, which may be shorter or longer than that allowed in the case of other defaults, may provide for an immediate
enforcement upon such default, may limit the remedies available to the trustee upon such default or may limit the right of holders
of a majority in aggregate principal amount of the debt securities of any series to waive such default;
•
to provide for the assumption by a successor company of the obligations of the Company under
the indenture;
TABLE
OF CONTENTS
•
to
add guarantees with respect to the debt securities or to secure the debt securities;
•
to make any change that does not adversely affect in any material respect the rights of any
holder of the debt securities;
•
to add to, change, or eliminate any of the provisions of the indenture with respect to one
or more series of debt securities, so long as any such addition, change or elimination not otherwise permitted under the indenture
will (a) neither apply to any debt securities of any series created prior to the execution of such supplemental indenture
and entitled to the benefit of such provision nor modify the rights of the holders of any such debt securities with respect to
the benefit of such provision or (b) become effective only when there is no such debt securities outstanding;
•
to evidence and provide for the acceptance of appointment by a successor or separate trustee
with respect to the debt securities of one or more series and to add to or change any of the provisions of the indenture as shall
be necessary to provide for or facilitate the administration of the indenture by more than one trustee;
•
to add or to change any of the provisions of the indenture to provide that debt securities
in bearer form may be registrable as to principal, to change or eliminate any restrictions on the payment of principal or premium
with respect to debt securities in registered form or of principal, premium or interest with respect to debt securities in bearer
form, or to permit debt securities in registered form to be exchanged for debt securities in bearer form, so as to not adversely
affect the interests of the holders of debt securities or any coupons of any series in any material respect or permit or facilitate
the issuance of debt securities of any series in uncertificated form;
•
in the case of subordinated debt securities, to make any change in the provisions of the
indenture or any supplemental indenture relating to subordination that would limit or terminate the benefits available to any
holder of senior Indebtedness under such provisions (but only if each such holder of senior Indebtedness consents to such change);
•
to comply with any requirement of the SEC in connection with the qualification of the indenture
or any supplemental indenture under the Trust Indenture Act;
•
to conform any provision in the indenture or the debt securities to the description of any
debt securities in an offering document;
•
to approve a particular form of any proposed amendment;
•
to provide for the issuance of additional debt securities of any series;
•
to establish the form or terms of debt securities and coupons of any series pursuant to the
indenture;
•
to comply with the rules of any applicable depositary;
•
to make any amendment to the provisions of the indenture relating to the transfer and legending
of debt securities; provided, however, that (a) compliance with the indenture as so amended would not result
in debt securities being transferred in violation of the Securities Act or any other applicable securities law and (b) such
amendment does not materially and adversely affect the rights of holders of debt securities to transfer debt securities; or
•
to convey, transfer, assign, mortgage or pledge any property to or with the trustee, or to
make such other provisions in regard to matters or questions arising under the indenture as shall not adversely affect, in any
material respect, the interests of any holders of debt securities of any series.
Mergers and
Sales of Assets
The indenture
will provide that we will not consolidate with or merge with or into, or convey, transfer or lease in one transaction or a series
of related transactions, directly or indirectly, all or substantially all of our properties and assets to, another person, unless
(i) the resulting, surviving or transferee person, if not NRX Pharmaceuticals, Inc., is a person organized and existing
under the laws of the United States of America, any state thereof or the District of Columbia; (ii) immediately after giving
effect to such transaction, no default or event of default has occurred and is continuing under the indenture; (iii) the
resulting,
TABLE
OF CONTENTS
surviving or transferee person, if not NRX Pharmaceuticals,
Inc., expressly assumes by supplemental indenture in a form satisfactory to the trustee all of our obligations under the debt
securities and the indenture; and (iv) we or the successor person has delivered to the trustee the certificates and opinions
of counsel required under the indenture. Upon any such consolidation, merger or transfer, the resulting, surviving or transferee
person shall succeed to, and may exercise every right and power of, NRX Pharmaceuticals, Inc. under the indenture.
Satisfaction
and Discharge of the Indenture; Defeasance
Unless otherwise
provided for in the prospectus supplement, the indenture will generally cease to be of any further effect with respect to a series
of debt securities if (a) we have delivered to the trustee for cancellation all debt securities of such series (with certain
limited exceptions) or (b) all debt securities and coupons of such series not theretofore delivered to the trustee for cancellation
will have become due and payable, or are by their terms to become due and payable within one year or are to be called for redemption
within one year, and we will have irrevocably deposited with the trustee as trust funds the entire amount sufficient to pay at
maturity or upon redemption all such debt securities and coupons (and if, in either case, we will also pay or cause to be paid
all other sums payable under the indenture by us).
In addition,
we will have a “legal defeasance option” (pursuant to which we may terminate, with respect to the debt securities
of a particular series, all of our obligations under such debt securities and the indenture with respect to such debt securities)
and a “covenant defeasance option” (pursuant to which we may terminate, with respect to the debt securities
of a particular series, our obligations with respect to such debt securities under certain specified covenants contained in the
indenture). If we exercise our legal defeasance option with respect to a series of debt securities, payment of such debt securities
may not be accelerated because of an event of default. If we exercise our covenant defeasance option with respect to a series
of debt securities, payment of such debt securities may not be accelerated because of an event of default related to the specified
covenants.
The applicable
prospectus supplement will describe the procedures we must follow in order to exercise our defeasance options
Regarding
the Trustee
The indenture
will provide that, except during the continuance of an event of default, the trustee will perform only such duties as are specifically
set forth in the indenture. During the existence of an event of default, the trustee may exercise such rights and powers vested
in it under the indenture and use the same degree of care and skill in its exercise as a prudent person would exercise under
the circumstances in the conduct of such person’s own affairs.
The indenture
and provisions of the Trust Indenture Act that are incorporated by reference therein will contain limitations on the rights of
the trustee, should it become one of our creditors, to obtain payment of claims in certain cases or to realize on certain property
received by it in respect of any such claim as security or otherwise. The trustee will be permitted to engage in other transactions
with us or any of our affiliates; provided, however, that if it acquires any conflicting interest (as defined in
the indenture or in the Trust Indenture Act), it must eliminate such conflict, apply to the SEC for permission to continue, or
resign.
Governing
Law
The indenture
and the debt securities will be governed by, and construed in accordance with, the laws of the State of New York.
DESCRIPTION
OF DEPOSITARY SHARES
General
We may, at
our option, elect to offer fractional shares rather than full shares of the preferred stock of a series. In the event that we
determine to do so, we will issue receipts for depositary shares, each of which will represent a fraction (to be set forth in
the prospectus supplement relating to a particular series of preferred stock) of a share of a particular series of preferred
stock as more fully described below.
The shares
of any series of preferred stock represented by depositary shares will be deposited under one or more deposit agreements among
us, a depositary to be named in the applicable prospectus supplement, and the holders from time to time of depositary receipts
issued thereunder. Subject to the terms of the applicable deposit agreement, each holder of a depositary share will be entitled,
in proportion to the applicable fraction of a share of preferred stock represented by the depositary share, to all the rights
and preferences of the preferred stock represented thereby (including, as applicable, dividend, voting, redemption, subscription
and liquidation rights).
The depositary
shares will be evidenced by depositary receipts issued pursuant to the deposit agreement. Depositary receipts will be distributed
to those persons purchasing the fractional shares of the related series of preferred stock.
The following
description sets forth certain general terms and provisions of the depositary shares to which any prospectus supplement may relate.
The particular terms of the depositary shares to which any prospectus supplement may relate and the extent, if any, to which
such general provisions may apply to the depositary shares so offered will be described in the applicable prospectus supplement.
To the extent that any particular terms of the depositary shares or the deposit agreement described in a prospectus supplement
differ from any of the terms described below, then the terms described below will be deemed to have been superseded by that prospectus
supplement relating to such deposited shares. The forms of deposit agreement and depositary receipt will be filed as exhibits
to the documents incorporated or deemed to be incorporated by reference into this prospectus.
The following
summary of certain provisions of the depositary shares and deposit agreement does not purport to be complete and is subject to,
and is qualified in its entirety by express reference to, all the provisions of the deposit agreement and the applicable prospectus
supplement, including the definitions.
Immediately
following our issuance of shares of a series of preferred stock that will be offered as fractional shares, we will deposit the
shares with the depositary, which will then issue and deliver the depositary receipts to the purchasers thereof. Depositary receipts
will only be issued evidencing whole depositary shares. A depositary receipt may evidence any number of whole depositary shares.
Pending the
preparation of definitive depositary receipts, the depositary may, upon our written order, issue temporary depositary receipts
substantially identical to (and entitling the holders thereof to all the rights pertaining to) the definitive depositary receipts
but not in definitive form. Definitive depositary receipts will be prepared thereafter without unreasonable delay, and such temporary
depositary receipts will be exchangeable for definitive depositary receipts at our expense.
Dividends
and Other Distributions
The depositary
will distribute all cash dividends or other cash distributions received in respect of the related series of preferred stock to
the record holders of depositary shares relating to the series of preferred stock in proportion to the number of the depositary
shares owned by the holders.
In the event
of a distribution other than in cash, the depositary will distribute property received by it to the record holders of depositary
shares entitled thereto in proportion to the number of depositary shares owned by the holders, unless the depositary determines
that the distribution cannot be made proportionately among the holders or that it is not feasible to make the distributions,
in which case the depositary may, with our approval, adopt any method as it deems equitable and practicable for the purpose of
effecting the distribution, including the sale (at public or private sale) of the securities or property thus received, or any
part thereof, at the place or places and upon those terms as it may deem proper.
TABLE
OF CONTENTS
The amount distributed in
any of the foregoing cases will be reduced by any amounts required to be withheld by us or the depositary on account of taxes
or other governmental charges.
Redemption
of Depositary Shares
If any series
of the preferred stock underlying the depositary shares is subject to redemption, the depositary shares will be redeemed from
the proceeds received by the depositary resulting from any redemption, in whole or in part, of the series of the preferred stock
held by the depositary. The redemption price per depositary share will be equal to the applicable fraction of the redemption
price per share payable with respect to the series of the preferred stock. If we redeem shares of a series of preferred stock
held by the depositary, the depositary will redeem as of the same redemption date the number of depositary shares representing
the shares of preferred stock so redeemed. If less than all the depositary shares are to be redeemed, the depositary shares to
be redeemed will be selected by lot or substantially equivalent method determined by the depositary.
After the
date fixed for redemption, the depositary shares so called for redemption will no longer be deemed to be outstanding and all
rights of the holders of the depositary shares will cease, except the right to receive the monies payable upon redemption and
any money or other property to which the holders of the depositary shares were entitled upon such redemption, upon surrender
to the depositary of the depositary receipts evidencing the depositary shares. Any funds deposited by us with the depositary
for any depositary shares that the holders thereof fail to redeem will be returned to us after a period of two years from
the date the funds are so deposited.
Voting the
Underlying Preferred Stock
Upon receipt
of notice of any meeting at which the holders of any series of the preferred stock are entitled to vote, the depositary will
mail the information contained in the notice of meeting to the record holders of the depositary shares relating to the series
of preferred stock. Each record holder of the depositary shares on the record date (which will be the same date as the record
date for the related series of preferred stock) will be entitled to instruct the depositary as to the exercise of the voting
rights pertaining to the number of shares of the series of preferred stock represented by that holder’s depositary shares.
The depositary will endeavor, insofar as practicable, to vote or cause to be voted the number of shares of preferred stock represented
by the depositary shares in accordance with the instructions, provided the depositary receives the instructions sufficiently
in advance of the meeting to enable it to so vote or cause to be voted the shares of preferred stock, and we will agree to take
all reasonable action that may be deemed necessary by the depositary in order to enable the depositary to do so. The depositary
will abstain from voting shares of the preferred stock to the extent it does not receive specific instructions from the holders
of depositary shares representing the preferred stock.
Withdrawal
of Stock
Upon surrender
of the depositary receipts at the corporate trust office of the depositary and upon payment of the taxes, charges and fees provided
for in the deposit agreement and subject to the terms thereof, the holder of the depositary shares evidenced thereby will be
entitled to delivery at such office, to or upon his or her order, of the number of whole shares of the related series of preferred
stock and any money or other property, if any, represented by the depositary shares. Holders of depositary shares will be entitled
to receive whole shares of the related series of preferred stock, but holders of the whole shares of preferred stock will not
thereafter be entitled to deposit the shares of preferred stock with the depositary or to receive depositary shares therefor.
If the depositary receipts delivered by the holder evidence a number of depositary shares in excess of the number of depositary
shares representing the number of whole shares of the related series of preferred stock to be withdrawn, the depositary will
deliver to the holder or upon his or her order at the same time a new depositary receipt evidencing the excess number of depositary
shares.
Amendment
and Termination of a Deposit Agreement
The form
of depositary receipt evidencing the depositary shares of any series and any provision of the applicable deposit agreement may
at any time and from time to time be amended by agreement between us
TABLE
OF CONTENTS
and the depositary. However, any amendment that
materially adversely alters the rights of the holders of depositary shares of any series will not be effective unless the amendment
has been approved by the holders of at least a majority of the depositary shares of the series then outstanding. Every holder
of a depositary receipt at the time the amendment becomes effective will be deemed, by continuing to hold the depositary receipt,
to be bound by the deposit agreement as so amended. Notwithstanding the foregoing, in no event may any amendment impair the right
of any holder of any depositary shares, upon surrender of the depositary receipts evidencing the depositary shares and subject
to any conditions specified in the deposit agreement, to receive shares of the related series of preferred stock and any money
or other property represented thereby, except in order to comply with mandatory provisions of applicable law. The deposit agreement
may be terminated by us at any time upon not less than 60 days prior written notice to the depositary, in which case, on
a date that is not later than 30 days after the date of the notice, the depositary shall deliver or make available for delivery
to holders of depositary shares, upon surrender of the depositary receipts evidencing the depositary shares, the number of whole
or fractional shares of the related series of preferred stock as are represented by the depositary shares. The deposit agreement
shall automatically terminate after all outstanding depositary shares have been redeemed or there has been a final distribution
in respect of the related series of preferred stock in connection with any liquidation, dissolution or winding up of us and the
distribution has been distributed to the holders of depositary shares.
Charges of
Depositary
We will pay
all transfer and other taxes and the governmental charges arising solely from the existence of the depositary arrangements. We
will pay the charges of the depositary, including charges in connection with the initial deposit of the related series of preferred
stock and the initial issuance of the depositary shares and all withdrawals of shares of the related series of preferred stock,
except that holders of depositary shares will pay transfer and other taxes and governmental charges and any other charges as
are expressly provided in the deposit agreement to be for their accounts.
Resignation
and Removal of Depositary
The depositary
may resign at any time by delivering to us written notice of its election to do so, and we may at any time remove the depositary.
Any resignation or removal will take effect upon the appointment of a successor depositary, which successor depositary must be
appointed within 60 days after delivery of the notice of resignation or removal and must be a bank or trust company having
its principal office in the United States and having a combined capital and surplus of at least $50,000,000.
Miscellaneous
The depositary
will forward to the holders of depositary shares all reports and communications from us that are delivered to the depositary
and which we are required to furnish to the holders of the related preferred stock.
The depositary’s
corporate trust office will be identified in the applicable prospectus supplement. Unless otherwise set forth in the applicable
prospectus supplement, the depositary will act as transfer agent and registrar for depositary receipts and if shares of a series
of preferred stock are redeemable, the depositary will also act as redemption agent for the corresponding depositary receipts.
DESCRIPTION
OF THE WARRANTS
The following
description of the terms of the warrants sets forth certain general terms and provisions of the warrants to which any prospectus
supplement may relate. We may issue warrants for the purchase of common stock, preferred stock, debt securities or depositary
shares. Warrants may be issued independently or together with common stock, preferred stock, debt securities or depositary shares
offered by any prospectus supplement and may be attached to or separate from any such offered securities. Each series of warrants
will be issued under a separate warrant agreement to be entered into between us and a bank or trust company, as warrant agent.
The warrant agent will act solely as our agent in connection with the warrants and will not assume any obligation or relationship
of agency or trust for or with any holders or beneficial owners of warrants. The following summary of certain provisions of the
warrants does not purport to be complete and is subject to, and qualified in its entirety by reference to, the provisions of
the warrant agreement that will be filed with the SEC in connection with the offering of such warrants.
Debt Warrants
The prospectus
supplement relating to a particular issue of debt warrants will describe the terms of such debt warrants, including the following:
•
the title of such debt warrants;
•
the offering price for such debt warrants, if any;
•
the aggregate number of such debt warrants;
•
the designation and terms of the debt securities purchasable upon exercise of such debt warrants;
•
if applicable, the designation and terms of the debt securities with which such debt warrants
are issued and the number of such debt warrants issued with each such debt security;
•
if applicable, the date from and after which such debt warrants and any debt securities issued
therewith will be separately transferable;
•
the principal amount of debt securities purchasable upon exercise of a debt warrant and the
price at which such principal amount of debt securities may be purchased upon exercise (which price may be payable in cash, securities
or other property);
•
the date on which the right to exercise such debt warrants shall commence and the date on
which such right shall expire;
•
if applicable, the minimum or maximum amount of such debt warrants that may be exercised
at any one time;
•
whether the debt warrants represented by the debt warrant certificates or debt securities
that may be issued upon exercise of the debt warrants will be issued in registered or bearer form;
•
information with respect to book-entry procedures, if any;
•
the currency or currency units in which the offering price, if any, and the exercise
price are payable;
•
if applicable, a discussion of material United States federal income tax considerations;
•
the antidilution or adjustment provisions of such debt warrants, if any;
•
the redemption or call provisions, if any, applicable to such debt warrants; and
•
any additional terms of such debt warrants, including terms, procedures, and limitations
relating to the exchange and exercise of such debt warrants.
Stock Warrants
The prospectus
supplement relating to any particular issue of common stock warrants, preferred stock warrants or depositary share warrants will
describe the terms of such warrants, including the following:
•
the title of such warrants;
TABLE
OF CONTENTS
•
the
offering price for such warrants, if any;
•
the aggregate number of such warrants;
•
the designation and terms of the offered securities purchasable upon exercise of such warrants;
•
if applicable, the designation and terms of the offered securities with which such warrants
are issued and the number of such warrants issued with each such offered security;
•
if applicable, the date from and after which such warrants and any offered securities issued
therewith will be separately transferable;
•
the number of shares of common stock, preferred stock or depositary shares purchasable upon
exercise of a warrant and the price at which such shares may be purchased upon exercise;
•
the date on which the right to exercise such warrants shall commence and the date on which
such right shall expire;
•
if applicable, the minimum or maximum amount of such warrants that may be exercised at any
one time;
•
the currency or currency units in which the offering price, if any, and the exercise
price are payable;
•
if applicable, a discussion of material United States federal income tax considerations;
•
the antidilution provisions of such warrants, if any;
•
the redemption or call provisions, if any, applicable to such warrants; and
•
any additional terms of such warrants, including terms, procedures and limitations relating
to the exchange and exercise of such warrants.
DESCRIPTION
OF THE RIGHTS
We may issue
rights to purchase our common stock. The rights may or may not be transferable by the persons purchasing or receiving the rights.
In connection with any rights offering, we may enter into a standby underwriting or other arrangement with one or more underwriters
or other persons pursuant to which such underwriters or other persons would purchase any offered securities remaining unsubscribed
for after such rights offering. Each series of rights will be issued under a separate rights agent agreement to be entered into
between us and one or more banks, trust companies or other financial institutions, as rights agent, that we will name in the
applicable prospectus supplement. The rights agent will act solely as our agent in connection with the rights and will not assume
any obligation or relationship of agency or trust for or with any holders of rights certificates or beneficial owners of rights.
The prospectus
supplement relating to any rights that we offer will include specific terms relating to the offering, including, among other
matters:
•
the date of determining the security holders entitled to the rights distribution;
•
the aggregate number of rights issued and the aggregate number of shares of common stock
purchasable upon exercise of the rights;
•
the exercise price;
•
the conditions to completion of the rights offering;
•
the date on which the right to exercise the rights will commence and the date on which the
rights will expire; and
•
any applicable federal income tax considerations.
Each right
would entitle the holder of the rights to purchase for cash the principal amount of shares of common stock at the exercise price
set forth in the applicable prospectus supplement. Rights may be exercised at any time up to the close of business on the expiration
date for the rights provided in the applicable prospectus supplement. After the close of business on the expiration date, all
unexercised rights will become void.
If less than
all of the rights issued in any rights offering are exercised, we may offer any unsubscribed securities directly to persons other
than our security holders, to or through agents, underwriters or dealers or through a combination of such methods, including
pursuant to standby arrangements, as described in the applicable prospectus supplement.
DESCRIPTION
OF THE PURCHASE CONTRACTS
We may issue,
from time to time, purchase contracts, including contracts obligating holders to purchase from us and us to sell to the holders,
a specified principal amount of debt securities, shares of common stock or preferred stock, depositary shares, government securities,
or other securities that we may sell under this prospectus at a future date or dates. The consideration payable upon settlement
of the purchase contracts may be fixed at the time the purchase contracts are issued or may be determined by a specific reference
to a formula set forth in the purchase contracts. The purchase contracts may be issued separately or as part of units consisting
of a purchase contract and other securities or obligations issued by us or third parties, including United States treasury securities,
securing the holders’ obligations to purchase the relevant securities under the purchase contracts. The purchase contracts
may require us to make periodic payments to the holders of the purchase contracts or units or vice versa, and the payments
may be unsecured or prefunded on some basis. The purchase contracts may require holders to secure their obligations under the
purchase contracts.
The prospectus
supplement related to any particular purchase contracts will describe, among other things, the material terms of the purchase
contracts and of the securities being sold pursuant to such purchase contracts, a discussion, if appropriate, of any special
United States federal income tax considerations applicable to the purchase contracts and any material provisions governing the
purchase contracts that differ from those described above. The description in the prospectus supplement will not necessarily
be complete and will be qualified in its entirety by reference to the purchase contracts, and, if applicable, collateral arrangements
and depositary arrangements, relating to the purchase contracts.
We may, from
time to time, issue units comprised of one or more of certain other securities that may be offered under this prospectus,
in any combination. Each unit may also include debt obligations of third parties, such as U.S. Treasury securities. Each unit
will be issued so that the holder of the unit is also the holder of each security included in the unit. Thus, the holder of a
unit will have the rights and obligations of a holder of each included security. The unit agreement under which a unit is issued
may provide that the securities included in the unit may not be held or transferred separately at any time, or at any time before
a specified date.
Any prospectus
supplement related to any particular units will describe, among other things:
•
the material terms of the units and of the securities comprising the units, including
whether and under what circumstances those securities may be held or transferred separately;
•
any material provisions relating to the issuance, payment, settlement, transfer or exchange
of the units or of the securities comprising the units;
•
if appropriate, any special United States federal income tax considerations applicable to
the units; and
•
any material provisions of the governing unit agreement that differ from those described
above.
We may offer
and sell the securities in any one or more of the following ways:
•
to or through underwriters, brokers or dealers;
•
directly to one or more other purchasers;
•
through a block trade in which the broker or dealer engaged to handle the block trade will
attempt to sell the securities as agent, but may position and resell a portion of the block as principal to facilitate the transaction;
•
through agents on a best-efforts basis; or
•
otherwise through a combination of any of the above methods of sale.
In addition,
we may enter into option, share lending or other types of transactions that require us to deliver shares of common stock to an
underwriter, broker or dealer, who will then resell or transfer the shares of common stock under this prospectus. We may also
enter into hedging transactions with respect to our securities. For example, we may:
•
enter into transactions involving short sales of the shares of common stock by underwriters,
brokers or dealers;
•
sell shares of Common Stock short and deliver the shares to close out short positions;
•
enter into option or other types of transactions that require us to deliver shares of Common
Stock to an underwriter, broker or dealer, who will then resell or transfer the shares of Common Stock under this prospectus;
or
•
loan or pledge the shares of Common Stock to an underwriter, broker or dealer, who may sell
the loaned shares or, in the event of default, sell the pledged shares.
We may enter
into derivative transactions with third parties, or sell securities not covered by this prospectus to third parties in privately
negotiated transactions. If the applicable prospectus supplement indicates, in connection with those derivatives, the third parties
may sell securities covered by this prospectus and the applicable prospectus supplement, including in short sale transactions.
If so, the third party may use securities pledged by us or borrowed from us or others to settle those sales or to close out any
related open borrowings of stock, and may use securities received from us in settlement of those derivatives to close out any
related open borrowings of stock. The third party in such sale transactions will be an underwriter and, if not identified in
this prospectus, will be identified in the applicable prospectus supplement (or a post-effective amendment). In addition, we
may otherwise loan or pledge securities to a financial institution or other third party that in turn may sell the securities
short using this prospectus. Such financial institution or other third party may transfer its economic short position to investors
in our securities or in connection with a concurrent offering of other securities.
Each time we
sell securities, we will provide a prospectus supplement that will name any underwriter, dealer or agent involved in the offer
and sale of the securities. The prospectus supplement will also set forth the terms of the offering, including:
•
the purchase price of the securities and the proceeds we will receive from the sale of the
securities;
•
any underwriting discounts and other items constituting underwriters’ compensation;
•
any public offering or purchase price and any discounts or commissions allowed or re-allowed
or paid to dealers;
•
any commissions allowed or paid to agents;
•
any other offering expenses;
•
any securities exchanges on which the securities may be listed;
•
the method of distribution of the securities;
TABLE
OF CONTENTS
•
the
terms of any agreement, arrangement or understanding entered into with the underwriters, brokers or dealers; and
•
any other information we think is important.
If underwriters
or dealers are used in the sale, the securities will be acquired by the underwriters or dealers for their own account. The securities
may be sold from time to time by us in one or more transactions:
•
at a fixed price or prices that may be changed;
•
at market prices prevailing at the time of sale;
•
at prices related to such prevailing market prices;
•
at varying prices determined at the time of sale; or
•
at negotiated prices.
Such sales
may be effected:
•
in transactions on any national securities exchange or quotation service on which the securities
may be listed or quoted at the time of sale;
•
in transactions in the over-the-counter market;
•
in block transactions in which the broker or dealer so engaged will attempt to sell the securities
as agent but may position and resell a portion of the block as principal to facilitate the transaction, or in crosses, in which
the same broker acts as an agent on both sides of the trade;
•
through the writing of options; or
•
through other types of transactions.
The securities
may be offered to the public either through underwriting syndicates represented by one or more managing underwriters or directly
by one or more of such firms. Unless otherwise set forth in the prospectus supplement, the obligations of underwriters or dealers
to purchase the securities offered will be subject to certain conditions precedent and the underwriters or dealers will be obligated
to purchase all the offered securities if any are purchased. Any public offering price and any discount or concession allowed
or reallowed or paid by underwriters or dealers to other dealers may be changed from time to time.
Any shares
of Common Stock covered by this prospectus that qualify for sale pursuant to Rule 144 under the Securities Act may be sold
under Rule 144 rather than pursuant to this prospectus. Any shares of Common Stock offered under this prospectus will be
listed on The Nasdaq Global Market (or other such exchange or automated quotation system on which the Common Stock is listed),
subject to official notice of issuance.
The securities
may be sold directly by us or through agents designated by us from time to time. Any agent involved in the offer or sale of the
securities in respect of which this prospectus is delivered will be named, and any commissions payable by us to such agent will
be set forth in, the prospectus supplement. Unless otherwise indicated in the prospectus supplement, any such agent will be acting
on a best efforts basis for the period of its appointment.
Offers to
purchase the securities offered by this prospectus may be solicited, and sales of the securities may be made by us directly to
institutional investors or others, who may be deemed to be underwriters within the meaning of the Securities Act with respect
to any resale of the securities. The terms of any offer made in this manner will be included in the prospectus supplement relating
to the offer.
If indicated
in the applicable prospectus supplement, underwriters, dealers or agents will be authorized to solicit offers by certain institutional
investors to purchase securities from us pursuant to contracts providing for payment and delivery at a future date. Institutional
investors with which these contracts may be made include, among others:
•
commercial and savings banks;
•
insurance companies;
TABLE
OF CONTENTS
•
pension
funds;
•
investment companies; and
•
educational and charitable institutions.
In all cases,
these purchasers must be approved by us. Unless otherwise set forth in the applicable prospectus supplement, the obligations
of any purchaser under any of these contracts will not be subject to any conditions except that (a) the purchase of the
securities must not at the time of delivery be prohibited under the laws of any jurisdiction to which that purchaser is subject,
and (b) if the securities are also being sold to underwriters, we must have sold to these underwriters the securities not
subject to delayed delivery. Underwriters and other agents will not have any responsibility in respect of the validity or performance
of these contracts.
Some of the
underwriters, dealers or agents used by us in any offering of securities under this prospectus may be customers of, engage in
transactions with, and perform services for us or affiliates of ours in the ordinary course of business. Underwriters, dealers,
agents and other persons may be entitled under agreements which may be entered into with us to indemnification against and contribution
toward certain civil liabilities, including liabilities under the Securities Act, and to be reimbursed by us for certain expenses.
Subject to
any restrictions relating to debt securities in bearer form, any securities initially sold outside the United States may be resold
in the United States through underwriters, dealers or otherwise.
Any underwriters
to which offered securities are sold by us for public offering and sale may make a market in such securities, but those underwriters
will not be obligated to do so and may discontinue any market making at any time.
The anticipated
date of delivery of the securities offered by this prospectus will be described in the applicable prospectus supplement relating
to the offering.
To comply with
the securities laws of some states, if applicable, the securities may be sold in these jurisdictions only through registered
or licensed brokers or dealers. In addition, in some states the securities may not be sold unless they have been registered or
qualified for sale or an exemption from registration or qualification requirements is available and is complied with.
Unless otherwise
indicated in the applicable prospectus supplement, certain legal matters will be passed upon for us by Paul, Weiss, Rifkind,
Wharton & Garrison LLP, New York, New York. If legal matters in connection with offerings made pursuant to this prospectus
are passed upon by counsel for underwriters, dealers or agents, if any, such counsel will be named in the prospectus supplement
relating to such offering.
The consolidated
financial statements of NRX Pharmaceuticals, Inc. as of December 31, 2021 and 2020, and for each of the years then
ended have been incorporated by reference herein in reliance upon the report of KPMG LLP, independent registered public accounting
firm, incorporated by reference herein, and upon the authority of such firm as experts in accounting and auditing.

Shares of Common Stock
Pre-Funded Warrants to
Purchase Shares of Common Stock
PRELIMINARY PROSPECTUS SUPPLEMENT

February ,
2024
NRX Pharmaceuticals (NASDAQ:NRXP)
Historical Stock Chart
From Jun 2024 to Jul 2024
NRX Pharmaceuticals (NASDAQ:NRXP)
Historical Stock Chart
From Jul 2023 to Jul 2024