UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16 UNDER
THE SECURITIES EXCHANGE ACT OF 1934
For the Month of December 2023
Commission File Number: 001-38104
IMMURON LIMITED
(Name of Registrant)
Level 3, 62 Lygon Street, Carlton South,
Victoria, 3053, Australia
(Address of principal executive office)
Indicate
by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F.
Form
20-F ☒ Form 40-F ☐
Indicate
by check mark whether by furnishing the information contained in this Form, the registrant is also thereby furnishing the information
to the Commission pursuant to Rule 12g3-2(b) under the Securities Exchange Act of 1934.
Yes
☐ No ☒
If “Yes”
is marked, indicate below the file number assigned to the registrant in connection with Rule 12g3-2(b): 82-
IMMURON LIMITED
EXPLANATORY NOTE
Immuron Limited (the “Company”) published
one announcement (the “Public Notices”) to the Australian Securities Exchange on December 4, 2023 titled:
| |
- |
“First Patients Enrolled in Campylobacter Clinical Study” |
A copy of the Public Notice is attached as an exhibit to this report
on Form 6-K.
This report on Form 6-K (including the exhibit
hereto) shall not be deemed to be “filed” for purposes of the Securities Exchange Act of 1934, as amended (the “Exchange
Act”) and shall not be incorporated by reference into any filing under the Securities Act of 1933, as amended, except as shall be
expressly set forth by specific reference in such filing.
EXHIBITS
SIGNATURE
Pursuant to the requirements
of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto
duly authorized.
| |
IMMURON LIMITED |
| |
|
|
| |
 |
| |
|
|
| Date: December 4, 2023 |
By: |
/s/ Phillip Hains |
| |
|
Phillip Hains |
| |
|
Company Secretary |
3
Exhibit 99.1

Immuron Announces First Patients Enrolled in
Campylobacter Challenge Clinical Study
Highlights:
| ● | First participants enrolled in the US Naval
Medical Research Command Clinical Trial |
| ● | Clinical Study initiated to evaluate the efficacy
of a new Immuron clinical product to protecting volunteers against moderate to severe campylobacteriosis |
| ● | New CampETEC product is a prophylactic therapeutic
designed to protect against Campylobacter and Enterotoxigenic Escherichia coli (ETEC) infections, two of the major causes of Travelers’
diarrhea |
| ● | The in-patient stage of the study is anticipated
to be completed by the end of December 2023 |
Melbourne, Australia, December 04, 2023: Immuron
Limited (ASX: IMC; NASDAQ: IMRN), an Australian based and globally integrated biopharmaceutical company is pleased to announce that the
US Naval Medical Research Command (NMRC) has initiated the clinical evaluation of a new oral therapeutic targeting Campylobacter and Enterotoxigenic
Escherichia coli (ETEC) developed in collaboration with Immuron. The NMRC has prioritized
the clinical development of the study to evaluate the efficacy of the new therapeutic product to prevent infectious diarrhea caused
by Campylobacter.
The clinical study is being led by Principal Investigator
Dr Kawsar Talaat, MD at the Johns Hopkins University (JHU) Center for Immunization Research (CIR) Inpatient Unit, located at the JHU Bayview
Medical Campus, Baltimore, Maryland. U.S. The Phase 2 clinical trial is designed to evaluate the safety and protective efficacy of the
new product compared to a placebo in a controlled human infection model (CHIM). The primary efficacy outcome is prevention and/or reduction
of moderate to severe diarrhea. ClinicalTrials.gov Identifier: NCT06122870.
The dosing, challenge and the in-patient stage
of the study is anticipated to be completed by the third week of December 2023. The estimated study completion date (last participant,
last visit) is June 2024 with headline results from the clinical trial expected to be reported in 2H 2024.
Infectious diarrhea is the most common illness
reported by travelers visiting developing countries and among US troops deployed overseas. The morbidity and associated discomfort stemming
from diarrhea decreases daily performance, affects judgment, decreases morale and declines operational readiness. The first line of treatment
for infectious diarrhea is the prescription of antibiotics. Unfortunately, in the last decade, several enteric pathogens have demonstrated
increasing resistance to commonly prescribed antibiotics. In addition, traveler’s diarrhea is now recognized by the medical community
to result in post-infectious sequelae, including post-infectious irritable bowel syndrome (IBS) and several post-infectious autoimmune
diseases. A preventative treatment that defends against infectious enteric diseases is a high priority objective for the US Military.
This release has been authorised by the directors
of Immuron Limited.
- - - END - - -


|
COMPANY CONTACT:
Steven Lydeamore
Chief Executive Officer
Ph: +61 (0)3 9824 5254
info@immuron.com |
|
|
About Travelan®
Travelan® is an
orally administered passive immunotherapy that prophylactically reduces the likelihood of contracting travelers’ diarrhea, a
digestive tract disorder that is commonly caused by pathogenic bacteria and the toxins they produce. Travelan® is a highly
purified tabletised preparation of hyper immune bovine antibodies and other factors, which when taken with meals bind to diarrhea-causing
bacteria and prevent colonization and the pathology associated with travelers’ diarrhea. In Australia, Travelan® is a listed
medicine on the Australian Register for Therapeutic Goods (AUST L 106709) and is indicated to reduce the risk of Travelers’ Diarrhea,
reduce the risk of minor gastro-intestinal disorders and is antimicrobial. In Canada, Travelan® is a licensed natural health product
(NPN 80046016) and is indicated to reduce the risk of Travelers’ Diarrhea. In the U.S., Travelan® is sold as a dietary supplement
for digestive tract protection.
About Travelers’ diarrhea
Travelers’ diarrhea
is a gastrointestinal infection with symptoms that include loose, watery (and occasionally bloody) stools, abdominal cramping, bloating,
and fever, Enteropathogenic bacteria are responsible for most cases, with enterotoxigenic Escherichia coli (ETEC) playing a dominant
causative role. Campylobacter spp. are also responsible for a significant proportion of cases. The more serious infections with Salmonella
spp. the bacillary dysentery organisms belonging to Shigella spp. and Vibrio spp. (the causative agent of cholera) are often confused
with travelers’ diarrhea as they may be contracted while travelling and initial symptoms are often indistinguishable.
About Immuron
Immuron Limited (ASX:
IMC, NASDAQ: IMRN), is an Australian biopharmaceutical company focused on developing and commercializing orally delivered targeted polyclonal
antibodies for the treatment of inflammatory mediated and infectious diseases.
For more information
visit: http://www.immuron.com
FORWARD-LOOKING STATEMENTS:
This press release may contain “forward-looking
statements” within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934,
each as amended. Such statements include, but are not limited to, any statements relating to our growth strategy and product development
programs and any other statements that are not historical facts. Forward-looking statements are based on management’s current expectations
and are subject to risks and uncertainties that could negatively affect our business, operating results, financial condition and stock
value. Factors that could cause actual results to differ materially from those currently anticipated include: risks relating to our growth
strategy; our ability to obtain, perform under and maintain financing and strategic agreements and relationships; risks relating to the
results of research and development activities; risks relating to the timing of starting and completing clinical trials; uncertainties
relating to preclinical and clinical testing; our dependence on third-party suppliers; our ability to attract, integrate and retain key
personnel; the early stage of products under development; our need for substantial additional funds; government regulation; patent and
intellectual property matters; competition; as well as other risks described in our SEC filings. We expressly disclaim any obligation
or undertaking to release publicly any updates or revisions to any forward-looking statements contained herein to reflect any change in
our expectations or any changes in events, conditions or circumstances on which any such statement is based, except as required by law.

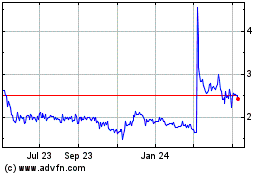
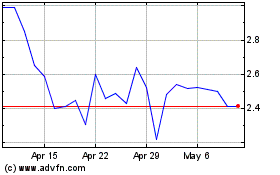
Immuron (NASDAQ:IMRN)
Historical Stock Chart
From Jun 2024 to Jul 2024
Immuron (NASDAQ:IMRN)
Historical Stock Chart
From Jul 2023 to Jul 2024