Filed
Pursuant to Rule 424(b)(2)
Registration No. 333-269631
PROSPECTUS
SUPPLEMENT
(To
Prospectus dated February 14, 2023)
$3,700,000

BioRestorative
Therapies, Inc.
Common
Stock
___________________________
We
have entered into a Capital on Demand™ Sales Agreement, or the Sales Agreement, with JonesTrading Institutional Services LLC, or
the Agent, relating to the sale of shares of our common stock offered by this prospectus supplement and the accompanying prospectus.
In accordance with the terms of the Sales Agreement, we may offer and sell shares of our common stock having an aggregate offering price
of up to $3,700,000 from time to time through or to the Agent acting as sales agent or principal, at our discretion.
Sales
of our common stock, if any, under this prospectus supplement and the accompanying prospectus will be made in sales deemed to be “at
the market offering” as defined in Rule 415 promulgated under the Securities Act of 1933, as amended, or the Securities Act. There
is no arrangement for funds to be received in any escrow, trust or similar arrangement.
We
will pay the Agent commissions for its services in acting as agent in the sale of our common stock. The Agent will be entitled to compensation
at a commission rate equal to 3.0% of the aggregate gross sales price of the shares sold. In connection with the sale of our common stock
on our behalf, the Agent will be deemed to be an “underwriter” within the meaning of the Securities Act and the compensation
of the Agent will be deemed to be underwriting commissions or discounts. We have also agreed to provide indemnification and contribution
to the Agent with respect to certain liabilities, including liabilities under the Securities Act.
Our
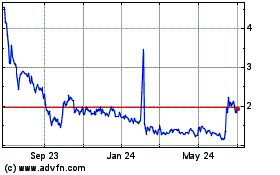
common stock is listed on the Nasdaq Capital Market, or Nasdaq, under the symbol “BRTX.” The last reported sales price of
our shares of common stock on April 13, 2023 was $3.74 per share.
The
aggregate market value of our outstanding common stock held by non-affiliates, or the public float, as of the date hereof pursuant to
General Instruction I.B.6. of Form S-3, was approximately $11,232,551, which was calculated based upon 3,003,356 shares
of our outstanding common stock held by non-affiliates at a price of $3.74 per share, the closing price of our common stock on
April 13, 2023. Pursuant to General Instruction I.B.6 of Form S-3, in no event will we sell our securities in a public primary
offering with a value exceeding more than one-third of our public float in any 12-month period so long as our public float remains below
$75 million. We have not offered any securities pursuant to General Instruction I.B.6 of Form S-3 during the 12 calendar months prior
to and including the date of this prospectus supplement.
___________________________
Investing
in our common stock involves risks. See the section entitled “Risk Factors” beginning on page S-7 of this prospectus supplement
and in the documents we incorporate by reference into this prospectus supplement and the accompanying prospectus.
Neither
the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or passed
upon the adequacy or accuracy of this prospectus supplement and the accompanying prospectus. Any representation to the contrary is a
criminal offense.
___________________________
JonesTrading
The
date of this prospectus supplement is April 14, 2023
TABLE
OF CONTENTS
ABOUT
THIS PROSPECTUS SUPPLEMENT
This
document contains two parts. The first part is this prospectus supplement, which describes the specific terms of this offering and also
supplements and updates information contained in the accompanying prospectus and the documents incorporated by reference into this prospectus
supplement and the accompanying prospectus. The second part is the accompanying prospectus, which provides more general information,
some of which may not apply to this offering. If the information contained in this prospectus supplement differs or varies from the information
contained in the accompanying prospectus, you should rely on the information set forth in this prospectus supplement.
You
should rely only on the information contained or incorporated by reference into this prospectus supplement and the accompanying prospectus.
We have not, and the Agent has not, authorized anyone else to provide you with information that is in addition to or different from that
contained or incorporated by reference into this prospectus supplement and the accompanying prospectus, along with the information contained
in any permitted free writing prospectuses we have authorized for use in connection with this offering.
We
are offering to sell, and seeking offers to buy, shares of our common stock only in jurisdictions where offers and sales are permitted.
The information contained in this prospectus supplement and the accompanying prospectus is accurate only as of the date of this prospectus
supplement or the date of the accompanying prospectus, and the information in the documents incorporated by reference into this prospectus
supplement and the accompanying prospectus is accurate only as of the date of those respective documents, regardless of the time of delivery
of this prospectus supplement and the accompanying prospectus or of any sale of our common stock. Our business, financial condition,
results of operations and prospects may have changed since those dates. It is important for you to read and consider all information
contained or incorporated by reference into this prospectus supplement and the accompanying prospectus in making your investment decision.
You should read both this prospectus supplement and the accompanying prospectus, as well as the documents incorporated by reference into
this prospectus supplement and the accompanying prospectus and the additional information described under “Where You Can Find More
Information” in this prospectus supplement and in the accompanying prospectus, before investing in our common stock.
Unless
the context of this prospectus supplement indicates otherwise, the terms “BioRestorative,” the “Company,” “we,”
“us” or “our” refer to BioRestorative Therapies, Inc. and its subsidiaries.
PROSPECTUS
SUPPLEMENT SUMMARY
This
summary highlights certain information about us and this offering. Because it is a summary, it does not contain all of the information
that you should consider before investing. Before investing in our common stock, you should read this entire prospectus supplement and
the accompanying prospectus carefully, including the “Risk Factors,” and the financial statements and accompanying notes
and other information incorporated by reference into this prospectus supplement and the accompanying prospectus.
Our
Company
We
develop therapeutic products, using cell and tissue protocols, primarily involving adult stem cells. Our two core programs, as described
below, relate to the treatment of disc/spine disease and metabolic disorders:
| |
● |
Disc/Spine
Program (brtxDisc). Our lead cell therapy candidate, BRTX-100, is a product formulated from autologous (or a person’s
own) cultured mesenchymal stem cells, or MSCs, collected from the patient’s bone marrow. We intend that the product will be
used for the non-surgical treatment of painful lumbosacral disc disorders or as a complimentary therapeutic to a surgical procedure.
The BRTX-100 production process utilizes proprietary technology and involves collecting a patient’s bone marrow, isolating
and culturing stem cells from the bone marrow and cryopreserving the cells. In an outpatient procedure, BRTX-100 is to be
injected by a physician into the patient’s damaged disc. The treatment is intended for patients whose pain has not been alleviated
by non-surgical procedures and who potentially face the prospect of surgery. We have received authorization from the Food and Drug
Administration, or the FDA, to commence a Phase 2 clinical trial using BRTX-100 to treat chronic lower back pain arising from
degenerative disc disease. We have commenced such clinical trial through the execution of a CRO agreement with PRC Clinical, the
execution of clinical trial site agreements, patient enrollment, the commencement of patient procedures, the purchase of manufacturing
equipment and the expansion of our laboratory to include capabilities for clinical production. In March 2022, a United States patent
related to our Disc/Spine Program was issued. We have been granted exclusive license rights with regard to the patent. |
| |
|
|
| |
● |
Metabolic
Program (ThermoStem). We are developing a cell-based therapy candidate to target obesity and metabolic disorders using brown
adipose (fat) derived stem cells, or BADSC, to generate brown adipose tissue, or BAT. We refer to this as our ThermoStem Program.
BAT is intended to mimic naturally occurring brown adipose depots that regulate metabolic homeostasis in humans. Initial preclinical
research indicates that increased amounts of brown fat in animals may be responsible for additional caloric burning as well as reduced
glucose and lipid levels. Researchers have found that people with higher levels of brown fat may have a reduced risk for obesity
and diabetes. Patents related to the ThermoStem Program have been issued in the United States and other jurisdictions. |
We
have also licensed an investigational curved needle device designed to deliver cells and/or other therapeutic products or material to
the spine and discs (and other parts of the body). We anticipate that FDA approval or clearance will be necessary for this device prior
to commercialization. We do not intend to utilize this device in connection with our Phase 2 clinical trial with regard to BRTX-100.
Key
Risks and Uncertainties
Our
business is subject to numerous risks, as more fully described or incorporated by reference in the “Risk Factors” section
beginning on page S-7 of this prospectus supplement. You should read these risks before you invest in our common stock. We may be unable,
for many reasons, including those that are beyond our control, to implement our business strategy. In particular, risks associated with
our business include, among others:
Risks
Related to Our Business Generally
| |
● |
We
have a limited operating history; we have incurred substantial losses since inception; we expect to continue to incur losses for
the near term. |
| |
● |
Even
if we sell all of the securities offered by this prospectus supplement, we will need to obtain a significant amount of financing
to complete our clinical trials and implement our business plan. |
| |
● |
We
will need to enter into agreements in order to implement our business strategy. |
| |
● |
We
depend on our executive officers and on our ability to attract and retain additional qualified personnel. |
Risks
Related to Our Cell Therapy Product Development Efforts
| |
● |
Our
future success is significantly dependent on the timely and successful development and commercialization of BRTX-100, our
lead product candidate for the treatment of chronic lumbar disc disease; if we encounter delays or difficulties in the development
of this product candidate, as well as any other product candidates, our business prospects would be significantly harmed. |
| |
● |
We
may experience delays and other difficulties in enrolling a sufficient number of patients in our clinical trials which could delay
or prevent the receipt of necessary regulatory approvals. |
| |
● |
The
development of our cell therapy product candidates is subject to uncertainty because autologous cell therapy is inherently variable. |
| |
● |
Any
disruption to our access to the media (including cell culture media) and reagents we are using in the clinical development of our
cell therapy product candidates could adversely affect our ability to perform clinical trials and seek future regulatory submissions. |
| |
● |
Our
clinical trials may fail to demonstrate adequately the safety and efficacy of our product candidates, which would prevent or delay
regulatory approval and commercialization. |
| |
● |
Even
if we complete the necessary clinical trials, we cannot predict when, or if, we will obtain regulatory approval to commercialize
a product candidate, and the approval may be for a narrower indication than we seek. |
| |
● |
We
may never obtain FDA approval for any of our product candidates in the United States and, even if we do, we may never obtain approval
for or commercialize any of our product candidates in any foreign jurisdiction, which would limit our ability to realize our full
market potential. |
| |
● |
We
presently lack manufacturing capabilities to produce our product candidates in connection with our Phase 3 clinical trial and at
commercial scale quantities and do not have an alternate manufacturing supply, which could negatively impact our ability to meet
any future demand for the products. |
| |
● |
The
commercial potential and profitability of our products are unknown and subject to significant risk and uncertainty. |
| |
● |
We
may have difficulties in sourcing brown adipose (fat) tissue. |
| |
● |
If
safety problems are encountered by us or others developing new stem cell-based therapies, our stem cell initiatives could be materially
and adversely affected. |
| |
● |
We
are vulnerable to competition and technological change, and also to physicians’ inertia. |
| |
● |
We
have limited experience in the development and marketing of cell therapies and may be unsuccessful in our efforts to establish a
profitable business. |
| |
● |
Our
cell therapy business is based on novel technologies that are inherently expensive, risky and may not be understood by or accepted
in the marketplace, which could adversely affect our future value. |
| |
● |
Our
cell therapy product candidates for which we intend to seek approval as biologic products may face competition sooner than anticipated. |
| |
● |
We
may be subject to significant product liability claims and litigation, including potential exposure from the use of our product candidates
in human subjects, and our insurance may be inadequate to cover claims that may arise. |
| |
● |
Our
internal computer systems, or those that are expected to be used by our clinical investigators, clinical research organizations or
other contractors or consultants, may fail or suffer security breaches, which could result in a material disruption of development
programs for our product candidates. |
| |
● |
Our
inability to obtain reimbursement for our products and services from private and governmental insurers could negatively impact demand
for our products and services. |
Risks
Related to Our Intellectual Property
| |
● |
We
may not be able to protect our proprietary rights. |
| |
● |
Changes
to United States patent law may have a material adverse effect on our intellectual property rights. |
| |
● |
In
certain countries, patent holders may be required to grant compulsory licenses, which would likely have a significant and detrimental
effect on any future revenues in such country. |
Risks
Related to Government Regulation
| |
● |
Even
if we obtain regulatory approval for a product candidate, our products will remain subject to regulatory oversight. |
| |
● |
We
may be subject, directly or indirectly, to federal and state healthcare fraud and abuse laws, false claims laws and health information
privacy and security laws. If we are unable to comply, or have not fully complied, with such laws, we could face substantial penalties. |
| |
● |
The
failure to receive regulatory approvals for our cell therapy product candidates would likely have a material and adverse effect on
our business and prospects. |
| |
● |
If
we are unable to conduct clinical studies in accordance with regulations and accepted standards, we may be delayed in receiving,
or may never receive, regulatory approvals of our product candidates from the FDA and other regulatory authorities. |
| |
● |
Health
care companies have been the subjects of federal and state investigations, and we could become subject to investigations in the future. |
| |
● |
It
is uncertain to what extent the government, private health insurers and third-party payors will approve coverage or provide reimbursement
for the therapies and products to which our services relate. Availability for such reimbursement may be further limited by reductions
in Medicare, Medicaid and other federal healthcare program funding in the United States. |
| |
● |
Competitor
companies or hospitals in the European Union, or EU, may be able to take advantage of EU rules permitting sales of unlicensed medicines
for individual patients to sell competing products without a marketing authorization. |
Corporate
Information
We
are a Nevada corporation. Our headquarters are located at 40 Marcus Drive, Suite One, Melville, New York 11747. Our telephone number
is (631) 760-8100. We maintain certain information on our website at www.biorestorative.com. The information on our website is not (and
should not be considered) part of this prospectus supplement and is not incorporated into this prospectus supplement by reference. We
have included our website address solely as an inactive textual reference. Investors should not rely on any such information in deciding
whether to purchase our common stock.
THE
OFFERING
| Common
stock offered |
|
Shares
of our common stock having an aggregate offering price of up to $3,700,000. |
| |
|
|
| Manner
of offering |
|
“At
the market offering” pursuant to the Sales Agreement that may be made from time to time through or to the Agent (i.e., JonesTrading
Institutional Services LLC), as sales agent or principal. See “Plan of Distribution” on page S-10 of this prospectus
supplement. |
| |
|
|
| Use
of proceeds |
|
We
intend to use the net proceeds from this offering in connection with our clinical trials with respect to BRTX-100, pre-clinical
research and development with respect to our ThermoStem Program and for general corporate purposes and working capital. We
may also use a portion of the net proceeds from this offering to acquire or invest in complementary businesses, technologies, product
candidates or other intellectual property, although we have no present commitments or agreements to do so. See “Use of Proceeds”
on page S-9 of this prospectus supplement. |
| |
|
|
| Risk
factors |
|
See
“Risk Factors” beginning on page S-7 of this prospectus supplement, and under similar headings in other documents incorporated
by reference into this prospectus supplement and the accompanying prospectus, for a discussion of factors you should consider carefully
before deciding to invest in our common stock. |
| |
|
|
| Nasdaq
Capital Market symbol |
|
“BRTX” |
RISK
FACTORS
Investing
in our common stock involves a high degree of risk. Before making an investment decision, you should carefully consider the risks described
below and in our most recent Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and in our other
Securities and Exchange Commission, or SEC, filings incorporated by reference into this prospectus supplement and the accompanying prospectus,
any amendment or update thereto reflected in our subsequent filings with the SEC, and all of the other information in this prospectus
supplement and the accompanying prospectus, including our financial statements and related notes incorporated by reference into this
prospectus supplement and the accompanying prospectus. If any of these risks is realized, our business, financial condition, results
of operations and prospects could be materially and adversely affected. In that event, the trading price of our common stock could decline
and you could lose part or all of your investment. Additional risks and uncertainties that are not yet identified or that we think are
immaterial may also materially harm our business, operating results and financial condition and could result in a complete loss of your
investment.
Risks
Related to this Offering
We
will have broad discretion in how we use the net proceeds of this offering. We may not use these proceeds effectively, which could affect
our results of operations and cause our stock price to decline.
Although
we currently intend to use the net proceeds from this offering in the manner described in the section entitled “Use of Proceeds”
in this prospectus supplement, we will have considerable discretion in the application of the net proceeds of this offering. We may use
the net proceeds for purposes that do not yield a significant return or any return at all for our stockholders. In addition, pending
their use, we may invest the net proceeds from this offering in a manner that does not produce income or that loses value. If we do not
invest or apply the net proceeds from this offering in ways that enhance stockholder value, we may fail to achieve expected financial
results, which could cause our stock price to decline.
It
is not possible to predict the actual number of shares we will sell under the Sales Agreement, or the gross proceeds resulting from those
sales.
Subject
to certain limitations in the Sales Agreement and compliance with applicable law, we have the discretion to deliver a placement notice
to the Agent at any time throughout the term of the Sales Agreement. The number of shares that are sold through the Agent after delivering
a placement notice will fluctuate based on a number of factors, including the market price of the common stock during the sales period,
the limits we set with the Agent in any applicable placement notice, and the demand for our common stock during the sales period. Because
the price per share of each share sold will fluctuate during the sales period, it is not currently possible to predict the number of
shares that will be sold or the gross proceeds to be raised in connection with those sales, if any.
The
common stock offered hereby will be sold in “at the market offerings,” and investors who buy shares at different
times will likely pay different prices.
Investors
who purchase shares in this offering at different times will likely pay different prices, and so they may experience different outcomes
in their investment results. We will have discretion, subject to market demand, to vary the timing, prices, and numbers of shares sold
in this offering. In addition, there is no minimum or maximum sales price for shares to be sold in this offering. Investors may experience
a decline in the value of the shares they purchase in this offering as a result of sales made at prices lower than the prices they paid.
FORWARD-LOOKING
STATEMENTS
This
prospectus supplement, the accompanying prospectus, the documents we have filed with the SEC that are incorporated by reference and any
free writing prospectus that we have authorized for use in connection with this offering contain “forward-looking statements”
within the meaning of Section 27A of the Securities Act and Section 21E of the Exchange Act. Forward-looking statements are made based
on our management’s expectations and beliefs concerning future events impacting our company and are subject to uncertainties and
factors relating to our operations and economic environment, all of which are difficult to predict and many of which are beyond our control.
You can identify these statements from our use of the words “estimate,” “project,” “believe,” “intend,”
“anticipate,” “expect,” “target,” “plan,” “may” and similar expressions.
These forward-looking statements may include, among other things:
| |
● |
statements
relating to projected growth and management’s long-term performance goals; |
| |
● |
statements
relating to the anticipated effects on results of operations or our financial condition from expected developments or events; |
| |
● |
statements
relating to our business and growth strategies; and |
| |
● |
any
other statements which are not historical facts. |
Forward-looking
statements involve known and unknown risks, uncertainties and other important factors that could cause our actual results, performance
or achievements, or industry results, to differ materially from our expectations of future results, performance or achievements expressed
or implied by these forward-looking statements. These forward-looking statements may not be realized due to a variety of factors, including
without limitation:
| |
● |
our
current and anticipated cash needs and our need for additional financing; |
| |
● |
federal,
state and foreign regulatory requirements; |
| |
● |
our
ability to conduct clinical trials with respect to our products and services; |
| |
● |
our
ability to develop and commercialize our products and services; |
| |
● |
our
ability to enter into agreements to implement our business strategy; |
| |
● |
the
acceptance of our products and services by patients and the medical community; |
| |
● |
our
ability to secure necessary media and reagents, as well as devices, materials and systems, for our clinical trials and
commercial production; |
| |
● |
our
manufacturing capabilities to produce our products; |
| |
● |
our
ability to obtain brown adipose (fat) tissue in connection with our ThermoStem Program; |
| |
● |
our
ability to protect our intellectual property; |
| |
● |
our
ability to obtain and maintain an adequate level of product liability insurance; |
| |
● |
our
ability to obtain third party reimbursement for our products and services from private and governmental insurers; |
| |
● |
the
effects of competition in our market areas; |
| |
● |
our
reliance on certain key personnel; |
| |
● |
further
sales or other dilution of our equity, which may adversely affect the market price of our common stock; and |
| |
● |
other
factors and risks referred to under “Risk Factors” beginning on page S-7 of this prospectus supplement. |
You
should not place undue reliance on any forward-looking statement. We undertake no obligation to update any forward-looking statement
to reflect events or circumstances after the date of this prospectus supplement or to reflect the occurrence of unanticipated events.
You
should rely only on the information contained in, or incorporated by reference into, this prospectus supplement, the accompanying prospectus
and any free writing prospectus that we have authorized for use in connection with this offering. We have not authorized anyone to provide
you with different information. The securities offered under this prospectus supplement are not being offered in any state where the
offer is not permitted. You should not assume that the information contained in this prospectus supplement or the accompanying prospectus
and any free writing prospectus that we have authorized for use in connection with this offering is accurate as of any date other than
the date on the front of this prospectus supplement or the accompanying prospectus, as applicable, or that any information incorporated
by reference into this prospectus supplement or the accompanying prospectus is accurate as of any date other than the date of the document
so incorporated by reference. Unless required by law or regulation, we undertake no obligation to update or revise any forward-looking
statements to reflect new information or future events or developments. Thus, you should not assume that our silence over time means
that actual events are bearing out as expressed or implied in such forward-looking statements.
USE
OF PROCEEDS
We
may issue and sell shares of common stock having aggregate sales proceeds of up to $3,700,000 from time to time in this offering.
Because there is no minimum offering amount required as a condition to close this offering, the actual total public offering amount,
commissions and net proceeds to us, if any, are not determinable at this time. There can be no assurance that we will sell any shares
in this offering, or that in the future we will sell any additional shares that remain available for sale under or fully utilize the
Sales Agreement with the Agent as a source of financing.
We
intend to use the net proceeds from this offering in connection with our clinical trials with respect to BRTX-100, pre-clinical
research and development with respect to our ThermoStem Program and for general corporate purposes and working capital. We may
also use a portion of the net proceeds from this offering to acquire or invest in complementary businesses, technologies, product candidates
or other intellectual property, although we have no present commitments or agreements to do so.
The
amounts and timing of these expenditures will depend on a number of factors, such as the timing and progress of our research and development
efforts, regulatory actions affecting our product candidates and our business, technological advances and the competitive environment
for our product candidates. As of the date of this prospectus supplement, we cannot specify with certainty all of the particular uses
for the net proceeds to us from this offering. Accordingly, we will retain broad discretion over the use of these proceeds.
PLAN
OF DISTRIBUTION
We
have entered into a Capital on Demand™ Sales Agreement, or the Sales Agreement, with JonesTrading, under which we may issue and
sell shares of our common stock from time to time through or to the Agent acting as our sales agent or principal. Sales of our common
stock, if any, will be made at market prices by any method that is deemed to be an “at the market offering” as defined in
Rule 415 promulgated under the Securities Act. We may instruct the Agent not to sell our common stock if the sales cannot be effected
at or above the price designated by us from time to time. We or the Agent may suspend the offering of our common stock upon notice and
subject to other conditions. As agent, the Agent will not engage in any transactions that stabilize the price of our common stock.
Each
time we wish to issue and sell common stock under the Sales Agreement, we will notify the Agent of the number or dollar value of shares
to be sold, the dates on which such sales are anticipated to be made, any minimum price below which sales may not be made and other sales
parameters as we deem appropriate. Once we have so instructed the Agent, unless the Agent declines to accept the terms of the notice,
the Agent has agreed to use commercially reasonable efforts consistent with its normal trading and sales practices to sell such shares
up to the amount specified on such terms. The obligation of the Agent under the Sales Agreement to sell shares of our common stock is
subject to a number of conditions that we must meet.
We
will pay the Agent a commission equal to 3.0% of the gross proceeds we receive from the sales of our common stock under the Sales Agreement.
Because there is no minimum offering amount required as a condition to closing this offering, the actual total public offering amount,
the Agent’s commission and proceeds to us, if any, are not determinable at this time. In addition, we have agreed to reimburse
the Agent for certain reasonable documented out-of-pocket expenses, including fees and disbursements of its counsel, in an amount not
to exceed $50,000 in the aggregate and ongoing diligence expenses in an amount not to exceed $10,000 in the aggregate per calendar year,
as provided in the Sales Agreement. We estimate that the total expenses of the offering payable by us, excluding commissions payable
to the Agent under the Sales Agreement, will be approximately $100,000.
Settlement
for sales of our common stock will occur on the second trading day following the date on which any sales are made, in return for payment
of the net proceeds to us. There is no arrangement for funds to be received in an escrow, trust or similar arrangement.
In
connection with the sale of the common stock on our behalf, the Agent will be deemed to be an “underwriter” within the meaning
of the Securities Act, and the compensation of the Agent will be deemed to be underwriting commission or discount. We have agreed to
provide indemnification and contribution to the Agent against certain liabilities, including liabilities under the Securities Act.
The
offering pursuant to the Sales Agreement will terminate upon the earlier of (i) the issuance and sale of all shares of our common stock
subject to the Sales Agreement, or (ii) the termination of the Sales Agreement as permitted therein.
The
Agent and its respective affiliates may in the future provide various investment banking and other financial services for us and our
affiliates, for which services they may in the future receive customary fees. To the extent required by Regulation M, the Agent will
not engage in any market making activities involving our common stock while the offering is ongoing under this prospectus supplement.
This
summary of the material provisions of the Sales Agreement does not purport to be a complete statement of its terms and conditions. We
are filing a copy of the Sales Agreement on a Form 8-K filed on or about the date of this prospectus supplement.
LEGAL
MATTERS
Except
as otherwise provided in any prospectus supplement, the validity of the issuance of the securities to be offered by this prospectus supplement
will be passed upon for us by Certilman Balin Adler & Hyman, LLP, East Meadow, New York. As of April 13, 2023, Certilman Balin Adler
& Hyman, LLP owned 41 shares of our common stock. Duane Morris LLP, New York, New York, is acting as counsel to the Agent in connection
with this offering.
EXPERTS
The
financial statements as of and for the year ended December 31, 2022, incorporated by reference into this prospectus supplement and in
the registration statement, have been so incorporated in reliance on the report of Marcum LLP, an independent registered public accounting
firm, incorporated herein by reference, given on the authority of said firm as experts in auditing and accounting.
The
financial statements as of and for the year ended December 31, 2021, incorporated by reference into this prospectus supplement and in
the registration statement, have been so incorporated in reliance on the report of Friedman LLP, an independent registered public accounting
firm, incorporated herein by reference, given on the authority of said firm as experts in auditing and accounting.
WHERE
YOU CAN FIND MORE INFORMATION
We
have filed a registration statement on Form S-3 with the SEC under the Securities Act. This prospectus supplement and the accompanying
prospectus is part of the registration statement but the registration statement includes and incorporates by reference additional information
and exhibits. We file annual, quarterly and current reports, proxy statements and other information with the SEC. The SEC maintains a
website that contains reports, proxy and information statements and other information regarding companies, such as ours, that file documents
electronically with the SEC. The address of that site on the worldwide web is http://www.sec.gov. The information on the SEC’s
website is not part of this prospectus supplement and the accompanying prospectus, and any references to this website or any other website
are inactive textual references only.
INCORPORATION
BY REFERENCE
The
SEC permits us to “incorporate by reference” the information contained in documents we file with the SEC, which means that
we can disclose important information to you by referring you to those documents rather than by including them in this prospectus supplement
and the accompanying prospectus. Information that is incorporated by reference is considered to be part of this prospectus supplement
and the accompanying prospectus and you should read it with the same care that you read this prospectus supplement and the accompanying
prospectus. Later information that we file with the SEC will automatically update and supersede the information that is either contained
in, or incorporated by reference into, this prospectus supplement and the accompanying prospectus, and will be considered to be a part
of this prospectus supplement and the accompanying prospectus from the date those documents are filed. We have filed with the SEC, and
incorporate by reference into this prospectus supplement and the accompanying prospectus (Commission File No. 001-37603):
| |
● |
our
Annual Report on Form 10-K for the year ended December 31, 2022, filed with the SEC on March 27, 2023; |
| |
|
|
| |
● |
our
Current Report on Form 8-K filed with the SEC on January 5, 2023; |
| |
|
|
| |
● |
our
Current Report on Form 8-K filed with the SEC on April 6, 2023; |
| |
|
|
| |
● |
the
description of our common stock contained in our registration statement on Form 8-A filed with the SEC on November 4, 2021, and any
amendment or report filed with the SEC for the purpose of updating the description. |
We
also incorporate by reference into this prospectus supplement all documents (other than Current Reports furnished under Item 2.02 or
Item 7.01 of Form 8-K and exhibits filed on such form that are related to such items) that are subsequently filed by us with the SEC
pursuant to Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act prior to the termination of the offering of the securities made by
this prospectus supplement. These documents include periodic reports, such as Annual Reports on Form 10-K, Quarterly Reports on Form
10-Q and Current Reports on Form 8-K, as well as proxy statements.
Any
statement contained in this prospectus or in a document incorporated or deemed to be incorporated by reference into this prospectus supplement
will be deemed to be modified or superseded to the extent that a statement contained in this prospectus or any subsequently filed document
that is deemed to be incorporated by reference into this prospectus supplement modifies or supersedes the statement.
We
will furnish without charge to you, on written or oral request, a copy of any or all of the documents incorporated by reference, excluding
exhibits to these documents unless the exhibits are specifically incorporated by reference into such documents. You should direct any
requests for documents to:
BioRestorative
Therapies, Inc.
40
Marcus Drive, Suite One
Melville,
New York 11747
(631)
760-8100
Attention:
Secretary
PROSPECTUS
BIORESTORATIVE
THERAPIES, INC.
$75,000,000
Common
Stock
Preferred
Stock
Warrants
Debt
Securities
Units
We
may offer, issue and sell, from time to time, in one or more offerings, the securities described in this prospectus. The aggregate initial
offering price of all securities sold under this prospectus by us will not exceed $75,000,000.
This
prospectus describes the general terms of our securities and the general manner in which our securities will be offered by us. We will
provide the specific terms of these offerings in supplements to this prospectus. We may authorize one or more free writing prospectuses
to be provided to you in connection with these offerings. The applicable prospectus supplement and any related free writing prospectus
may also add, update or change information contained or incorporated by reference in this prospectus. You should carefully read this
prospectus, the applicable prospectus supplement and any related free writing prospectus, as well as any documents incorporated by reference,
before buying any of the securities being offered.
We
may offer securities in amounts, at prices and on terms determined at the time of offering. Our securities may be sold directly to you,
through agents, or through underwriters and dealers. If agents, underwriters or dealers are used to sell our securities, we will name
them and describe their compensation in a prospectus supplement. No securities may be sold without delivery of this prospectus and the
applicable prospectus supplement describing the method and terms of the offering of such securities.
Our
common stock is listed on the Nasdaq Capital Market under the symbol “BRTX.”
As
of February 6, 2023, the aggregate market value of our outstanding common stock held by non-affiliates, or the public float, was $8,900,824,
based on 3,679,847 shares of common stock outstanding, of which 2,889,878 shares were held by non-affiliates, and a price of $3.08 per
share, which was the last reported sale price of our common stock on the Nasdaq Capital Market on February 6, 2023. Pursuant to General
Instruction I.B.6 of Form S-3, in no event will we sell shares pursuant to this prospectus with a value of more than one-third of the
aggregate market value of our common stock held by non-affiliates in any 12-month period, so long as the aggregate market value of our
common stock held by non-affiliates is less than $75,000,000. During the 12 calendar months prior to, and including, the date of this
prospectus, we have not sold securities pursuant to General Instruction I.B.6 of Form S-3.
Investing
in our securities involves a high degree of risk. You should review carefully the risks and uncertainties described under the heading
“Risk Factors” on page 3 and contained in the applicable prospectus supplement and any related free writing prospectus, and
under similar headings in the other documents that are incorporated by reference in this prospectus.
Neither
the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined
if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
This
prospectus may not be used to consummate sales of securities unless accompanied by a prospectus supplement.
The
date of this prospectus is February 14, 2023.
TABLE
OF CONTENTS
ABOUT
THIS PROSPECTUS
This
prospectus is part of a registration statement that we filed with the Securities and Exchange Commission, referred to as the SEC, using
a “shelf” registration process. Under this shelf registration process, we may from time to time offer and sell any of the
securities or any combination of the securities described in this prospectus in one or more offerings up to a total dollar amount not
to exceed $75,000,000.
This
prospectus provides you with a general description of the securities we may offer. Each time we sell securities, we will provide a prospectus
supplement that will contain specific information about the terms of that offering. Such prospectus supplement may also add, update or
change information contained in this prospectus. If there is any inconsistency between the information in this prospectus and the applicable
prospectus supplement, you should rely on the information in the prospectus supplement. You should read the information in this prospectus,
the applicable prospectus supplement, any free writing prospectus that we authorize for use in connection with this offering and the
additional information incorporated by reference herein as provided for under the heading “Incorporation of Certain Information
by Reference.”
You
should rely only on the information contained or incorporated by reference in this prospectus, any applicable prospectus supplement and
any free writing prospectuses that we authorize for use in connection with this offering. We have not authorized anyone to provide you
with different or additional information. If anyone provides you with different or additional information, you should not rely on it.
We are not making an offer to sell the securities in any jurisdiction where the offer or sale is not permitted or in which the person
making such offer or solicitation is not qualified to do so or to any person to whom it is unlawful to make such offer or solicitation.
You should not assume that the information contained or incorporated by reference in this prospectus, any prospectus supplement, or any
free writing prospectus that we authorize for use in connection with this offering is accurate or complete as of any date other than
the dates of the applicable documents. Our business, financial condition, liquidity, results of operations and prospects may have changed
since those dates.
This
prospectus contains summaries of certain provisions contained in some of the documents described herein, but reference is made to the
actual documents for complete information. All of the summaries are qualified in their entirety by the actual documents. Copies of some
of the documents referred to herein have been filed, will be filed or will be incorporated by reference as exhibits to the registration
statement of which this prospectus is a part, and you may obtain copies of those documents as described below under the heading “Where
You Can Obtain More Information.” As permitted by the rules and regulations of the SEC, the registration statement that contains
this prospectus includes additional information not contained in this prospectus. You may read the registration statement and the other
reports we file with the SEC at the SEC’s web site or at the SEC’s offices described below under the heading “Where
You Can Obtain More Information.”
Unless
the context of this prospectus indicates otherwise, the terms “BioRestorative,” the “Company,” “we,”
“us” or “our” refer to BioRestorative Therapies, Inc. and its subsidiaries.
WHERE
YOU CAN OBTAIN MORE INFORMATION
We
are subject to the information requirements of the Securities Exchange Act of 1934, as amended, referred to as the Exchange Act, which
means that we are required to file annual, quarterly and current reports, proxy statements and other information with the SEC, all of
which are available at the Public Reference Room of the SEC at 100 F Street, NE, Washington D.C. 20549. You may also obtain copies of
these reports, proxy statements and other information from the Public Reference Room of the SEC, at prescribed rates, by calling 1-800-SEC-0330.
The SEC maintains an Internet website at http://www.sec.gov where you can access reports, proxy statements, information and registration
statements, and other information regarding us that we file electronically with the SEC. In addition, we make available, without charge,
through our website, www.biorestorative.com, electronic copies of various filings with the SEC, including copies of Annual Reports on
Form 10-K. Information on our website should not be considered a part of this prospectus, and we do not intend to incorporate in this
prospectus any information contained on our website.
INCORPORATION
OF CERTAIN INFORMATION BY REFERENCE
The
SEC allows us to “incorporate by reference” the information we file with it, which means that we can disclose important information
to you by referring to those documents filed separately with the SEC. The information we incorporate by reference is an important part
of this prospectus. We incorporate by reference the documents listed below, except to the extent that any information contained in those
documents is deemed “furnished” in accordance with SEC rules.
| |
● |
Our
Annual Report on Form 10-K for the year ended December 31, 2021; |
| |
|
|
| |
● |
Our
Quarterly Report on Form 10-Q for the quarter ended March 31, 2022; |
| |
|
|
| |
● |
Our
Quarterly Report on Form 10-Q for the quarter ended June 30, 2022; |
| |
|
|
| |
● |
Our
Quarterly Report on Form 10-Q for the quarter ended September 30, 2022; |
| |
|
|
| |
● |
Our
Current Report on Form 8-K filed on September 13, 2022; |
| |
|
|
| |
● |
Our
Current Report on Form 8-K filed on November 9, 2022; |
| |
|
|
| |
● |
Our
Current Report on Form 8-K filed on January 5, 2023; |
| |
|
|
| |
● |
The
description of our common stock contained in our registration statement on Form 8-A filed with the SEC on November 4, 2021 (File
No. 001-37603), and any amendment or report filed with the SEC for the purpose of updating the description. |
We
also incorporate by reference additional documents that we will file with the SEC pursuant to Section 13(a), 13(c), 14 or 15(d) of the
Exchange Act prior to the termination of the offering under this prospectus. Those documents include periodic reports such as Annual
Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K, as well as proxy statements on Schedule DEF 14A.
Any
statement contained in a document that is incorporated by reference will be modified or superseded for all purposes to the extent that
a statement contained in this prospectus, any prospectus supplement or any free writing prospectus that we authorize for use in connection
with this offering modifies or is contrary to that previous statement. Any statement so modified or superseded will not be deemed a part
of this prospectus or any prospectus supplement except as so modified or superseded.
Documents
which we incorporate by reference are available from us without charge, excluding all exhibits, unless we have specifically incorporated
by reference an exhibit in this prospectus. You may obtain documents incorporated by reference in this prospectus by requesting them
in writing or by telephone from us at:
BioRestorative
Therapies, Inc.
40
Marcus Drive, Suite One
Melville,
New York 11747
(631)
760-8100
Attention:
Secretary
RISK
FACTORS
An
investment in our securities involves a high degree of risk. Prior to making a decision about investing in our securities, you should
consider carefully the specific risk factors discussed in the section entitled “Management’s Discussion and Analysis of Financial
Conditions and Results of Operations - Factors That May Affect Future Results and Financial Condition” contained in our most recent
Annual Report on Form 10-K for the year ended December 31, 2021, as filed with the SEC, and which is incorporated in this prospectus
by reference in its entirety, as well as any amendment or updates to our risk factors reflected in subsequent filings with the SEC, including
any prospectus supplement hereto. These risks and uncertainties are not the only risks and uncertainties we face. Additional risks and
uncertainties not presently known to us, or that we currently view as immaterial, may also impair our business. If any of the risks or
uncertainties described in our SEC filings or any additional risks and uncertainties actually occur, our business, financial condition,
results of operations and cash flow could be materially and adversely affected. In that case, the trading price of our securities could
decline and you might lose all or part of your investment. Please also refer to the section below entitled “Special Cautionary
Note Regarding Forward-Looking Statements.”
SPECIAL
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
Some
of the statements in or incorporated by reference into this prospectus contain “forward-looking statements.” Forward-looking
statements are made based on our management’s expectations and beliefs concerning future events impacting our company and are subject
to uncertainties and factors relating to our operations and economic environment, all of which are difficult to predict and many of which
are beyond our control. You can identify these statements from our use of the words “estimate,” “project,” “believe,”
“intend,” “anticipate,” “expect,” “target,” “plan,” “may” and
similar expressions. These forward-looking statements may include, among other things:
| |
● |
statements
relating to projected growth and management’s long-term performance goals; |
| |
● |
statements
relating to the anticipated effects on results of operations or our financial condition from expected developments or events; |
| |
● |
statements
relating to our business and growth strategies; and |
| |
● |
any
other statements which are not historical facts. |
Forward-looking
statements involve known and unknown risks, uncertainties and other important factors that could cause our actual results, performance
or achievements, or industry results, to differ materially from our expectations of future results, performance or achievements expressed
or implied by these forward-looking statements. These forward-looking statements may not be realized due to a variety of factors, including
without limitation:
| |
● |
our
current and anticipated cash needs and our need for additional financing; |
| |
● |
federal,
state and foreign regulatory requirements; |
| |
● |
our
ability to conduct clinical trials with respect to our products and services; |
| |
● |
our
ability to develop and commercialize our products and services; |
| |
● |
our
ability to enter into agreements to implement our business strategy; |
| |
● |
the
acceptance of our products and services by patients and the medical community; |
| |
● |
our
ability to secure necessary media and reagents, as well as devices, materials and systems, for our clinical trials and commercial
production; |
| |
● |
our
manufacturing capabilities to produce our products; |
| |
● |
our
ability to obtain brown adipose (fat) tissue in connection with our ThermoStem Program; |
| |
● |
our
ability to protect our intellectual property; |
| |
● |
our
ability to obtain and maintain an adequate level of product liability insurance; |
| |
● |
our
ability to obtain third party reimbursement for our products and services from private and governmental insurers; |
| |
● |
the
effects of competition in our market areas; |
| |
● |
our
reliance on certain key personnel; |
| |
● |
further
sales or other dilution of our equity, which may adversely affect the market price of our securities; and |
| |
● |
other
factors and risks referred to under “Risk Factors” on page 3 of this prospectus. |
You
should not place undue reliance on any forward-looking statement. We undertake no obligation to update any forward-looking statement
to reflect events or circumstances after the date of this prospectus or to reflect the occurrence of unanticipated events.
THE
COMPANY
We
develop therapeutic products, using cell and tissue protocols, primarily involving adult stem cells. Our two core programs, as described
below, relate to the treatment of disc/spine disease and metabolic disorders:
| |
● |
Disc/Spine
Program (brtxDisc). Our lead cell therapy candidate, BRTX-100, is a product formulated from autologous (or a person’s
own) cultured mesenchymal stem cells collected from the patient’s bone marrow. We intend that the product will be used for
the non-surgical treatment of painful lumbosacral disc disorders or as a complimentary therapeutic to a surgical procedure. The BRTX-100
production process utilizes proprietary technology and involves collecting a patient’s bone marrow, isolating and culturing
stem cells from the bone marrow and cryopreserving the cells. In an outpatient procedure, BRTX-100 is to be injected by a
physician into the patient’s damaged disc. The treatment is intended for patients whose pain has not been alleviated by non-surgical
procedures and who potentially face the prospect of surgery. We have received authorization from the FDA to commence a Phase 2 clinical
trial using BRTX-100 to treat chronic lower back pain arising from degenerative disc disease. We have commenced such clinical
trial through the execution of a CRO agreement with PRC Clinical, the execution of clinical trial site agreements, patient enrollment,
the commencement of patient procedures, the purchase of manufacturing equipment and the expansion of our laboratory to include capabilities
for clinical production. In March 2022, a United States patent related to BRTX-100 was issued. We have been granted excusive
license rights with regard to the patent. |
| |
|
|
|
● |
Metabolic
Program (ThermoStem). We are developing a cell-based therapy candidate to target obesity and metabolic disorders using brown
adipose (fat) derived stem cells to generate brown adipose tissue, or BAT. We refer to this as our ThermoStem Program. BAT
is intended to mimic naturally occurring brown adipose depots that regulate metabolic homeostasis in humans. Initial preclinical
research indicates that increased amounts of brown fat in animals may be responsible for additional caloric burning as well as reduced
glucose and lipid levels. Researchers have found that people with higher levels of brown fat may have a reduced risk for obesity
and diabetes. Patents related to the ThermoStem Program have been issued in the United States and other jurisdictions. |
We
have also licensed an investigational curved needle device designed to deliver cells and/or other therapeutic products or material to
the spine and discs (and other parts of the body). We anticipate that FDA approval or clearance will be necessary for this device prior
to commercialization. We do not intend to utilize this device in connection with our Phase 2 clinical trial with regard to BRTX-100.
The
patents and patent applications for the Disc/Spine Program, the ThermoStem Program and the curved needle device are listed
under “Business - Technology; Research and Development” in Part I of our Annual Report on Form 10-K for the year ended December
31, 2021 incorporated herein by reference.
We
are a Nevada corporation. Our headquarters are located at 40 Marcus Drive, Suite One, Melville, New York 11747. Our telephone number
is (631) 760-8100. We maintain certain information on our website at www.biorestorative.com. The information on our website is not (and
should not be considered) part of this prospectus and is not incorporated into this prospectus by reference.
USE
OF PROCEEDS
Unless
we indicate a different use in an accompanying prospectus supplement, the net proceeds from our sale of the offered securities will be
used for general corporate purposes and working capital, including in connection with our clinical trials with respect to BRTX-100,
pre-clinical research and development with respect to our ThermoStem Program and other business initiatives.
The
applicable prospectus supplement will provide more details on the use of proceeds of any specific offering.
DESCRIPTION
OF SECURITIES WE MAY OFFER
This
prospectus contains summary descriptions of the common stock, preferred stock, warrants, debt securities and units that we may offer
from time to time. These summary descriptions do not purport to be complete and are subject to, and qualified in their entirety by reference
to, the more complete descriptions thereof set forth in our amended and restated articles of incorporation, which we refer to as our
charter, and our bylaws, each as amended to date. The applicable prospectus supplement may add, update or change the terms and conditions
of the securities as described in this prospectus.
Authorization
Our
authorized capital stock consists of 95,000,000 shares of capital stock. We are authorized to issue 75,000,000 shares of common stock,
par value $0.0001 per share, and 20,000,000 shares of preferred stock, par value $0.01 per share.
As
of February 6, 2023, there were 3,679,847 shares of common stock issued and outstanding and 1,543,158 shares of Series B preferred stock
issued and outstanding.
Common
Stock
Dividend
Rights. Subject to preferences that may be applicable to any shares of our preferred stock that may be issued, the holders of our
common stock are entitled to share ratably in such dividends as may be declared by our Board of Directors out of funds legally available
therefor.
Voting
Rights. Each share of our common stock entitles its holder to one vote in the election of directors as well as all other matters
to be voted on by stockholders.
No
Preemptive Rights. Holders of our common stock do not have any preemptive rights to subscribe for additional shares on a pro rata
basis or otherwise when additional shares are offered for sale by us.
Liquidation
Rights. Subject to preferences that may be applicable to any shares of our preferred stock that may be issued, in the event of our
liquidation, dissolution or winding up, the holders of our common stock would be entitled to receive, pro rata, after payment of all
of our debts and liabilities, all of our remaining assets available for distribution.
Other
Rights. Holders of our common stock have no preferences or conversion or exchange rights. Shares of our common stock will not be
liable for further calls or assessments by us and are not subject to redemption.
Preferred
Stock
Our
board of directors has the authority, without further action by our stockholders, to issue from time to time the authorized preferred
stock in one or more series, and to fix the number of shares, designations, preferences, powers, and other rights and qualifications,
limitations or restrictions as our board of directors may authorize, including:
| |
● |
the
distinctive designation of each series and the number of shares that will constitute the series; |
| |
|
|
| |
● |
the
voting rights, if any, of shares of the series and the terms and conditions of the voting rights; |
| |
|
|
| |
● |
the
dividend rate on the shares of the series, the dates on which dividends are payable, any restriction, limitation or condition upon
the payment of dividends, whether dividends will be cumulative, and the dates from and after which dividends shall accumulate; |
| |
● |
the
prices at which, and the terms and conditions on which, the shares of the series may be redeemed, if the shares are redeemable; |
| |
|
|
| |
● |
the
terms and conditions of a sinking or purchase fund for the purchase or redemption of shares of the series, if such a fund is provided; |
| |
|
|
| |
● |
any
preferential amount payable upon shares of the series in the event of our liquidation, dissolution or winding up, or upon the distribution
of any of our assets; and |
| |
|
|
| |
● |
the
prices or rates of conversion or exchange at which, and the terms and conditions on which, the shares of the series may be converted
or exchanged into other securities, if the shares are convertible or exchangeable. |
Depending
upon the rights prescribed for a series of preferred stock, the issuance of preferred stock could have an adverse effect on the voting
power of the holders of common stock and could adversely affect holders of common stock by delaying or preventing a change in control,
making removal of our present management more difficult or imposing restrictions upon the payment of dividends and other distributions
to the holders of common stock.
The
particular terms of any series of preferred stock, and the transfer agent and registrar for that series, will be described in a prospectus
supplement.
Warrants
We
may offer to sell warrants from time to time. If we do so, we will describe the specific terms of the warrants in a prospectus supplement.
In particular, we may issue warrants for the purchase of common stock, preferred stock and/or debt securities in one or more series.
We may also issue warrants independently or together with other securities and the warrants may be attached to or separate from those
securities.
We
will evidence each series of warrants by warrant certificates that we will issue under a separate agreement. We will enter into the warrant
agreement with a warrant agent. We will indicate the name and address of the warrant agent in the applicable prospectus supplement relating
to a particular series of warrants.
We
will describe in the applicable prospectus supplement the terms of the series of warrants, including:
| |
● |
the
offering price and aggregate number of warrants offered; |
| |
|
|
| |
● |
the
currency for which the warrants may be purchased; |
| |
|
|
| |
● |
if
applicable, the designation and terms of the securities with which the warrants are issued and the number of warrants issued with
each such security or each principal amount of such security; |
| |
● |
if
applicable, the date on and after which the warrants and the related securities will be separately transferable; |
| |
|
|
| |
● |
in
the case of warrants to purchase debt securities, the principal amount of debt securities purchasable upon exercise of one warrant
and the price at, and currency in which, this principal amount of debt securities may be purchased upon such exercise; |
| |
|
|
| |
● |
in
the case of warrants to purchase common stock or preferred stock, the number of shares of common stock or preferred stock, as the
case may be, purchasable upon the exercise of one warrant and the price at which these shares may be purchased upon such exercise; |
| |
|
|
| |
● |
the
effect of any merger, consolidation, sale or other disposition of our business on the warrant agreement and the warrants; |
| |
|
|
| |
● |
the
terms of any rights to redeem or call the warrants; |
| |
|
|
| |
● |
any
provisions for changes to or adjustments in the exercise price or number of securities issuable upon exercise of the warrants; |
| |
|
|
| |
● |
the
dates on which the right to exercise the warrants will commence and expire; |
| |
|
|
| |
● |
the
manner in which the warrant agreement and warrants may be modified; |
| |
|
|
| |
● |
certain
United States federal income tax consequences of holding or exercising the warrants; |
| |
|
|
| |
● |
the
terms of the securities issuable upon exercise of the warrants; and |
| |
|
|
| |
● |
any
other specific material terms, preferences, rights or limitations of or restrictions on the warrants. |
Debt
Securities
The
following is a description of the material features, terms and provisions of debt securities that we may offer. This summary does not
purport to be exhaustive and may not contain all the information that is important to you. Therefore, you should read the applicable
prospectus supplement relating to those debt securities and any other offering materials that we may provide.
We
may issue debt securities from time to time in one or more series. We may issue senior debt securities or subordinated debt securities
under separate indentures, which may be supplemented or amended from time to time. Senior debt securities would be issued under a senior
indenture and subordinated debt securities would be issued under a subordinated indenture. The senior debt indenture and subordinated
debt indenture are referred to individually in this prospectus as the indenture, and collectively as the indentures.
The
particular terms of a series of debt securities will be described in a prospectus supplement relating to such series of debt securities.
The indentures will be subject to and governed by the Trust Indenture Act of 1939, as amended, and may be supplemented or amended from
time to time following their execution. Unless otherwise stated in the applicable prospectus supplement, we will not be limited in the
amount of debt securities that we may issue and neither the senior debt securities nor the subordinated debt securities will be secured
by any of our property or assets. Thus, by owning debt securities, you are one of our unsecured creditors.
The
indentures, and any supplemental indentures, will contain the full legal text of the matters described in this section of the prospectus.
Because this section is a summary, it does not describe every aspect of the debt securities or any applicable indentures or supplemental
indenture. This summary is therefore subject to and is qualified in its entirety by reference to all the provisions of any applicable
indenture or supplemental indenture, including any definitions of terms used in such indenture. Your rights will be defined by the terms
of any applicable indenture or supplemental indenture, not the summary provided herein. This summary is also subject to and qualified
by reference to the description of the particular terms of a particular series of debt securities described in the applicable prospectus
supplement or supplements.
The
debt securities may be denominated and payable in U.S. dollars. We may also issue debt securities, from time to time, with the principal
amount, interest or other amounts payable on any relevant payment date to be determined by reference to one or more currency exchange
rates, securities or baskets of securities, commodity prices, indices or any other financial, economic or other measure or instrument,
including the occurrence or non-occurrence of any event or circumstance. In addition, we may issue debt securities as part of any units
issued by us. All references in this prospectus or any prospectus supplement to other amounts will include premiums, if any, other cash
amounts payable under the applicable indenture, and the delivery of securities or baskets of securities under the terms of the debt securities.
Debt securities may bear interest at a fixed rate, which may be zero, or a floating rate.
We
will set forth in the applicable prospectus supplement the terms, if any, on which a series of debt securities may be convertible into
or exchangeable for our preferred stock, common stock or other securities. We will include provisions as to whether conversion or exchange
is mandatory, at the option of the holder or at our option. We may include provisions pursuant to which the number of shares of our preferred
stock, common stock or other securities that holders of the series of debt securities receive would be subject to adjustment.
The
applicable prospectus supplement will describe the debt securities and the price or prices at which we will offer the debt securities.
The description will, to the extent applicable, include:
| |
● |
the
title and form of the debt securities; |
| |
|
|
| |
● |
the
ranking of the debt securities as compared to other debt; |
| |
|
|
| |
● |
the
aggregate principal amount of the debt securities or the series of which they are a part; |
| |
● |
the
person or persons to whom any principal or interest on a debt security of the series will be paid; |
| |
● |
the
date or dates on which we must repay the principal; |
| |
● |
the
rate or rates at which the debt securities will bear interest; |
| |
● |
the
date or dates from which interest will accrue, and the dates on which we must pay interest; |
| |
● |
the
place or places where we must pay the principal and any premium or interest on the debt securities; |
| |
● |
the
terms and conditions on which the debt securities may be convertible into other securities; |
| |
● |
whether
the debt securities are entitled to the benefit of any sinking fund; |
| |
● |
the
identity of the trustee; |
| |
● |
the
terms and conditions on which we may redeem any debt security, if at all; |
| |
● |
any
obligation to redeem or purchase any debt securities, and the terms and conditions on which we must do so; |
| |
● |
the
denominations in which we may issue the debt securities; |
| |
● |
the
manner in which we will determine the amount of principal of or any premium or interest on the debt securities; |
| |
● |
the
currency in which we will pay the principal of and any premium or interest on the debt securities; |
| |
● |
the
principal amount of the debt securities that we will pay upon declaration of acceleration of their maturity; |
| |
● |
the
amount that will be deemed to be the principal amount for any purpose, including the principal amount that will be due and payable
upon any maturity or that will be deemed to be outstanding as of any date; |
| |
● |
whether
the debt securities are defeasible and the terms of such defeasance; and |
| |
● |
any
addition to or change in the events of default applicable to the debt securities and any change in the right of the trustee or the
holders to declare the principal amount of any of the debt securities due and payable. |
Some
of the debt securities may be issued as original issue discount debt securities. Original issue discount securities bear no interest
or bear interest at below market rates and will be sold at a discount below their stated principal amount. A prospectus supplement relating
to an issue of original issue discount securities will contain information relating to United States federal income tax, accounting,
and other special considerations applicable to original issue discount securities.
Units
We
may issue units comprised of one or more of the other securities described in this prospectus or any prospectus supplement in any combination.
Each unit will be issued so that the holder of the unit is also the holder, with the rights and obligations of a holder, of each security
included in the unit. The unit agreement under which a unit is issued may provide that the securities included in the unit may not be
held or transferred separately, at any time or at any times before a specified date or upon the occurrence of a specified event or occurrence.
The
applicable prospectus supplement will describe:
| |
● |
the
designation and the terms of the units and of the securities comprising the units, including whether and under what circumstances
those securities may be held or transferred separately; |
| |
|
|
| |
● |
any
unit agreement under which the units will be issued; |
| |
|
|
| |
● |
any
provisions for the issuance, payment, settlement, transfer or exchange of the units or of the securities comprising the units; and |
| |
|
|
| |
● |
whether
the units will be issued in fully registered or global form. |
Certain
Provisions Having Potential Anti-Takeover Effects
General.
The following is a summary of the material provisions of the Nevada Revised Statues, which we refer to as the NRS, and our charter
and bylaws that address matters of corporate governance and the rights of stockholders. Certain of these provisions may delay or prevent
takeover attempts not first approved by our Board of Directors (including takeovers which certain stockholders may deem to be in their
best interests). These provisions also could delay or frustrate the removal of incumbent directors or the assumption of control by stockholders.
The primary purpose of these provisions is to encourage negotiations with our management by persons interested in acquiring control of
our company. All references to the charter and bylaws are to our charter and bylaws in effect on the date of this prospectus.
Combinations
with Interested Stockholder. Sections 78.411-78.444, inclusive, of the NRS contain provisions governing combinations with an interested
stockholder. For purposes of the NRS, “combinations” include: (i) any merger or consolidation with any interested stockholder,
(ii) any sale, lease, exchange, mortgage, pledge, transfer or other disposition to any interested stockholder of corporate assets with
an aggregate market value equal to more than 5% of the aggregate market value of the corporation’s consolidated assets, more than
5% of the aggregate market value of outstanding shares of the corporation or more than 10% of the earning power or net income of the
corporation, (iii) the issuance to any interested stockholder of voting shares (except pursuant to a share dividend or similar proportionate
distribution) with an aggregate market value equal to 5% or more of the aggregate market value of all the outstanding shares of the corporation,
(iv) the adoption of the dissolution of the corporation if proposed by or on behalf of any interested stockholder, (v) any reclassification
of securities, recapitalization or corporate reorganization that will have the effect of increasing the proportionate share of the corporation’s
outstanding voting shares held by any interested stockholder and (vi) any receipt by the interested stockholder of the benefit (except
proportionately as a stockholder) of any loan, advance, guarantee, pledge or other financial assistance. For purposes of the NRS, an
“interested stockholder” is defined to include any beneficial owner of more than 10% of any class of the voting securities
of a Nevada corporation and any person who is an affiliate or associate of the corporation and was at any time during the preceding two
years the beneficial owner of more than 10% of any class of the voting securities of the Nevada corporation.
Subject
to certain exceptions, the provisions of the NRS statute governing combinations with interested stockholders provide that a Nevada corporation
may not engage in a combination with an interested stockholder for two years after the date that the person first became an interested
stockholder unless the combination or the transaction by which the person first became an interested stockholder is approved by the Board
of Directors before the person first became an interested stockholder, or unless the combination is approved by the Board of Directors
and sixty percent of the corporation’s voting power not beneficially owned by the interested stockholder, its affiliates and associates.
Control
Share Acquisitions. The NRS also contains a “control share acquisitions statute.” If applicable to a Nevada corporation
this statute restricts the voting rights of certain stockholders referred to as “acquiring persons,” that acquire or offer
to acquire ownership of a “controlling interest” in the outstanding voting stock of an “issuing corporation.”
For purposes of these provisions, a “controlling interest” means with certain exceptions the ownership of outstanding voting
stock sufficient to enable the acquiring person to exercise one-fifth or more but less than one-third, one-third or more but less than
a majority, or a majority or more of all voting power in the election of directors; “issuing corporation” means a Nevada
corporation that has 200 or more stockholders of record, at least 100 of whom have addresses in Nevada appearing on the stock ledger
of the corporation at all times during the 90 days immediately preceding such date, and which does business in Nevada directly or through
an affiliated corporation. The voting rights of an acquiring person in the affected shares will be restored only if such restoration
is approved by the holders of a majority of the voting power of the corporation. The NRS allows a corporation to “opt-out”
of the control share acquisitions statute by providing in such corporation’s articles of incorporation or bylaws that the control
share acquisitions statute does not apply to the corporation or to an acquisition of a controlling interest specifically by types of
existing or future stockholders, whether or not identified. The Company has not opted out of this statute.
Authorized
But Unissued Shares. Nevada law does not require stockholder approval for any issuance of authorized shares. Authorized but unissued
shares may be used for a variety of corporate purposes, including future public or private offerings to raise additional capital or to
facilitate corporate acquisitions. One of the effects of the existence of authorized but unissued shares may be to enable our Board of
Directors to issue shares to persons friendly to current management, which issuance could render more difficult or discourage an attempt
to obtain control of us by means of a merger, tender offer, proxy contest or otherwise, and thereby protect the continuity of our management
and possibly deprive the stockholders of opportunities to sell their shares of common stock at prices higher than prevailing market prices.
Preferred
Stock. Under the terms of our charter, our Board of Directors is authorized to issue shares of preferred stock in one or more series
without stockholder approval. Our Board of Directors has the discretion to determine the rights, preferences, privileges and restrictions,
including voting rights, dividend rights, conversion rights, redemption privileges and liquidation preferences, of each series of preferred
stock. The purpose of authorizing our Board of Directors to issue preferred stock and determine its rights and preferences is to provide
flexibility and eliminate delays associated with a stockholder vote on specific issues. However, the ability of our Board of Directors
to issue preferred stock and determine its rights and preferences may have the effect of delaying or preventing a change in control,
as described above under “Description of Securities We May Offer — Preferred Stock.”
Classified
Board. We have a classified Board of Directors consisting of three classes of directors. A classified board is one in which a certain
number, but not all, of the directors are elected on a rotating basis each year. This method of electing directors makes changes in the
composition of our Board more difficult, and thus a potential change in control may be a lengthier process. The existence of our classified
Board reduces the possibility that a third party could effect an unsolicited change in control of our Board. Since our classified Board
will increase the amount of time required for a takeover bidder to obtain control of us without the cooperation of the Board, even if
the takeover bidder were to acquire a majority of our outstanding common stock, the existence of our classified Board could tend to discourage
certain tender offers which stockholders might feel would be in their best interests. Our classified Board will likely allow management,
if confronted by a proposal from a third party who has acquired a block of our common stock, sufficient time to review the proposal and
appropriate alternatives to the proposal and to attempt to negotiate a better transaction, if possible, for our stockholders.
Special
Meetings of Stockholders. Our bylaws provide that special meetings of stockholders may be called only by our Board of Directors or
the Chairman of the Board.
Filling
Vacancies. Vacancies occurring in our Board of Directors and newly created directorships resulting from an increase in the authorized
number of directors may be filled by a majority of the remaining directors, even if less than a quorum.
Removal
of Directors by Stockholders. Under the terms of our charter, stockholders may remove directors with or without cause with the affirmative
vote of holders of 75% of the voting power of all of the then-outstanding shares of our capital stock then entitled to vote at an election
of directors, voting together as a single class.
Amendment
of Bylaws. Our bylaws may be amended by our Board of Directors or by the holders of at least 75% of the voting power of our company.
Amendment
of Certain Charter Provisions. Under the terms of our charter, amending certain charter provisions requires the affirmative vote
of the holders of at least 75% of the voting power of all of the then-outstanding shares of our capital stock entitled to vote thereon,
voting together as a single class. The provisions subject to such heightened requirement include those relating to stockholder action
by written consent, the calling of special meetings, board classification, the filling of board vacancies, the removal of directors and
the ability to amend our bylaws, among others.
Advance
Notification of Stockholder Nominations and Proposals. Our bylaws establish advance notice procedures with respect to the nomination
of persons for election as directors, other than nominations made by or at the direction of our Board of Directors, and stockholder proposals
for business.
Stockholder
Nominees; Stockholder Proposals.
In
order for a stockholder to nominate a candidate for director at, or bring any business before, an annual meeting of stockholders, under
our bylaws, timely notice of the nomination or business must be received by us in advance of the meeting. To be timely, a stockholder’s
notice must be delivered to or mailed and received by our Secretary at our principal executive offices not less than 90 days nor more
than 120 days prior to the one-year anniversary of the date on which we first mailed the proxy materials for the preceding year’s
annual meeting of stockholders; provided, however, that if the meeting is convened more than 30 days prior to or delayed more than 60
days after the anniversary of the preceding year’s annual meeting, to be timely a stockholder’s notice must be received not
earlier than the close of business on the 120th day prior to the date of such annual meeting and not later than the close of business
on the later of the 90th day prior to the date of such annual meeting or, if the first public announcement of the date of such annual
meeting is less than 100 days prior to the date of such annual meeting, the 10th day following the day on which public announcement of
the date of such meeting is first made by us.
The
stockholder sending the notice of nomination or proposed business must describe various matters, including the following:
| |
● |
as
to each person whom the stockholder proposes to nominate for election as a director, all information relating to such person as would
be required to be disclosed in solicitations of proxies for election of such nominee as a director pursuant to Regulation 14A under
the Exchange Act; |
| |
|
|
| |
● |
(i)
the name and address of such stockholder, as they appear on our books and of such beneficial owner or Control Person (as defined
in our bylaws), if any, (ii) the number of our shares which are, directly or indirectly, owned beneficially and of record by such
stockholder and such beneficial owner or Control Person, if any (iii) a representation that the stockholder intends to appear at
the meeting in person or by proxy to submit the business specified in such notice, (iv) if the notice relates to any business other
than a nomination of director(s), a brief description of the business desired to be brought before the meeting, including the complete
text of any resolutions proposed for consideration, and the reasons for conducting such business at the meeting, (v) any direct or
indirect personal or other interest of the stockholder in the business to be submitted, (vi) a description of any agreement, arrangement
or understanding (including any derivative or short positions, profit interests, options, hedging transactions, and borrowed or loaned
shares) that has been entered into as of the date of the stockholder’s notice by, or on behalf of, such stockholder or beneficial
owner and by any Control Person or any other person acting in concert with any of the foregoing, the effect or intent of which is
to mitigate loss, manage risk or benefit from changes in the share price of any class of our stock, or maintain, increase or decrease
the voting power of the stockholder or beneficial owner with respect to shares of our stock, and a representation that the stockholder
will notify us in writing within five business days after the record date for such meeting of any such agreement, arrangement or
understanding in effect as of the record date for the meeting, (vii) a representation whether the stockholder or the beneficial owner,
if any, and any Control Person will engage in a solicitation with respect to the nomination or business and, if so, the name of each
participant (as defined in Item 4 of Schedule 14A under the Securities Exchange Act of 1934) in such solicitation and whether such
person intends or is part of a group which intends to deliver a proxy statement and/or form of proxy to holders of at least the percentage
of our outstanding stock required to approve or adopt the business to be proposed (in person or by proxy) by the stockholder and
(viii) any other information relating to such stockholder, beneficial owner or Control Person, if any, that would be required to
be disclosed in a proxy statement and form of proxy or other filings required to be made in connection with solicitations of proxies
for, as applicable, the proposal and/or for the election of directors in a contested election pursuant to Section 14 of the Securities
Exchange Act of 1934 and the rules and regulations promulgated thereunder. |
These
provisions are intended to enhance the likelihood of continuity and stability in the composition of the Board and in policies formulated
by the Board and to discourage certain types of transactions that may involve an actual or threatened change of control of our company.
These provisions are designed to reduce our vulnerability to an unsolicited proposal for a takeover that does not contemplate the acquisition
of all of our outstanding shares or an unsolicited proposal for the restructuring or sale of all or part of our company.
Limitations
on Director Liability
Our
charter provides that our directors shall generally not be liable to us or any of our stockholders for damages for breach of duty as
a director. This provision will eliminate such liability except for (i) any breach of the director’s duty of loyalty to us or to
our stockholders, (ii) acts and omissions not in good faith or which involve intentional misconduct or a knowing violation of law, (iii)
liability for unlawful payment of dividends or unlawful stock purchases or redemptions in violation of the NRS, and (iv) any transaction
from which the director derived an improper personal benefit.
Indemnification
of Directors and Officers
Section
78.7502(1) of the NRS provides that a corporation may, and our charter and bylaws provide that we shall, indemnify any person who was
or is a party or is threatened to be made a party to any threatened, pending or completed action, suit or proceeding, whether civil,
criminal, administrative or investigative, or an Action, by reason of the fact that he is or was our director, officer, employee or agent
or is or was serving at our request in such capacity in another corporation, partnership, joint venture, trust or other enterprise, or
the Indemnified Party, against expenses (including attorney’s fees), judgments, fines and amounts paid in settlement actually and
reasonably incurred by him in connection with such action, suit or proceeding if he acted in good faith and in a manner he reasonably
believed to be in or not opposed to our best interests, and, with respect to any criminal action or proceeding, had no reasonable cause
to believe his conduct was unlawful; provided, however, no indemnification shall be made in respect of any Actions by or in our right
if the Indemnified Party shall have been adjudged by a court of competent jurisdiction, after exhaustion of any appeals, to be liable
to us, unless and only to the extent that the court shall determine that, despite the adjudication of liability but in view of all circumstances,
such person is fairly and reasonably entitled to indemnity.
Under
the NRS, the directors have a fiduciary duty to us that is not eliminated by this provision of our charter and, in appropriate circumstances,
equitable remedies such as injunctive or other forms of non-monetary relief will remain available. In addition, each director will continue
to be subject to liability under the NRS for breach of the director’s duty of loyalty to us for acts or omissions which are found
by a court of competent jurisdiction to not be in good faith or involve intentional misconduct, for knowing violations of law, for actions
leading to improper personal benefit to the director, and for payment of dividends or approval of stock repurchases or redemptions that
are prohibited by the NRS. This provision also does not affect the directors’ responsibilities under any other laws, such as the
federal securities laws or state or federal environmental laws.
Furthermore,
Section 78.7502(3) of the NRS provides that determination of an Indemnified Party’s eligibility for indemnification by us shall
be made on a case-by-case basis by: (i) the stockholders; (ii) the board of directors by a majority vote of a quorum consisting of directors
who were not parties to the action, suit or proceeding; (iii) independent legal counsel’s written opinion if: (1) a majority vote
of a quorum consisting of directors who were not parties to the action, suit or proceeding so orders; or (2) a quorum consisting of directors
who were not parties to the action, suit or proceeding cannot be obtained.
Lastly,
Section 78.752(1) of the NRS empowers a corporation to purchase and maintain insurance or make other financial arrangements with respect
to liability arising out of the actions or omissions of directors, officers, employees or agents in their capacity or status as such,
whether or not the corporation has the authority to indemnify him against such liability.
Our
charter provides that, to the fullest extent permitted by the NRS, no director or officer shall be personally liable to us or to our
stockholders for monetary damages for breach of fiduciary duty as a director or an officer, except to the extent that such exemption
from liability or limitation thereof is not permitted under the NRS currently in effect or as the same may be amended. If the NRS is
amended to further eliminate or limit or authorize corporate action to further eliminate or limit the liability of directors or officers,
the liability of our directors and officers shall be eliminated or limited to the fullest extent permitted by the NRS, as so amended
from time to time. No repeal or modification of this provision of our charter will apply to or have any effect on the liability or alleged
liability of any of our directors or officers for or with respect to any acts or omissions of such directors or officers occurring prior
to such repeal or modification.
Our
bylaws provide that we will indemnify and hold harmless any person who was or is a party or is
threatened to be made a party to any threatened, pending or completed action, suit or proceeding, whether civil, criminal, administrative
or investigative, in such manner, under such circumstances and to the fullest extent permitted by our charter and the NRS.
Insofar
as indemnification for liabilities arising under the Securities Act may be permitted to our directors, officers and controlling persons
under the provisions discussed above or otherwise, we have been advised that, in the opinion of the SEC, such indemnification is against
public policy as expressed in the Securities Act and is, therefore, unenforceable.
Transfer
Agent
The
transfer agent for our common stock is Transhare Corporation.
PLAN
OF DISTRIBUTION
We
may use this prospectus, any applicable prospectus supplement and any related free writing prospectus that we authorize for use in connection
with this offering to sell our securities from time to time pursuant to underwritten public offerings, negotiated transactions, block
trades or a combination of these methods. We may sell our securities (1) through one or more underwriters or dealers, (2) through one
or more agents, and/or (3) directly to one or more purchasers. We may distribute our securities from time to time in one or more transactions
at:
| |
● |
a
fixed price or prices, which may be changed; |
| |
|
|
| |
● |
market
prices prevailing at the time of sale; |
| |
|
|
| |
● |
prices
related to the prevailing market prices; |
| |
|
|
| |
● |
varying
prices determined at the time of sale; or |
| |
|
|
| |
● |
negotiated
prices. |
We
may solicit directly offers to purchase the securities being offered by this prospectus. We may also designate agents to solicit offers
to purchase our securities from time to time. We will name in a prospectus supplement any underwriter, dealer or agent involved in the
offer or sale of our securities and describe any commissions payable by us to these agents in the applicable prospectus supplement. Unless
otherwise indicated in the applicable prospectus supplement, these agents will be acting on a best efforts basis for the period of their
appointment. The agents may be customers of, or may engage in transactions with or perform services for, us in the ordinary course of
business.
We
may also sell equity securities covered by this registration statement in an “at the market” offering as defined in Rule
415(a)(4) under the Securities Act. Such offering may be made into an existing trading market for such securities in transactions at
other than a fixed price on or through the facilities of the Nasdaq Capital Market or any other securities exchange or quotation or trading
service on which such securities may be listed, quoted or traded at the time of sale. Such at the market offerings, if any, may be conducted
by underwriters acting as principal or agent.
If
we utilize a dealer in the sale of the securities being offered by this prospectus, we will sell our securities to the dealer, as principal.
The dealer may then resell our securities to the public at varying prices to be determined by the dealer at the time of resale.
If
we utilize an underwriter in the sale of our securities being offered by this prospectus, we will execute an underwriting agreement with
the underwriter at the time of sale and we will provide the name of any underwriter in the prospectus supplement which the underwriter
will use to make resales of our securities to the public. In connection with the sale of our securities, we, or the purchasers of our
securities for whom the underwriter may act as agent, may compensate the underwriter in the form of underwriting discounts or commissions.
The underwriter may sell our securities to or through dealers, and the underwriter may compensate those dealers in the form of discounts,
concessions or commissions.
With
respect to underwritten public offerings, negotiated transactions and block trades, we will provide in the applicable prospectus supplement
any compensation we pay to underwriters, dealers or agents in connection with the offering of our securities, and any discounts, concessions
or commissions allowed by underwriters to participating dealers. Underwriters, dealers and agents participating in the distribution of
our securities may be deemed to be underwriters within the meaning of the Securities Act and any discounts and commissions received by
them and any profit realized by them on resale of our securities may be deemed to be underwriting discounts and commissions. We may enter
into agreements to indemnify underwriters, dealers and agents against civil liabilities, including liabilities under the Securities Act,
or to contribute to payments they may be required to make in respect thereof.
Our
common stock is listed for trading on the Nasdaq Capital Market. We may elect to list any other security we offer on an exchange, but
we are not obligated to do so. To facilitate the offering of our securities, certain persons participating in the offering may engage
in transactions that stabilize, maintain or otherwise affect the price of our securities. This may include over-allotments or short sales
of our securities, which involve the sale by persons participating in the offering of more securities than we sold to them. In these
circumstances, these persons would cover such over-allotments or short positions by making purchases in the open market or by exercising
their over-allotment option. In addition, these persons may stabilize or maintain the price of our securities by bidding for or purchasing
our securities in the open market or by imposing penalty bids, whereby selling concessions allowed to dealers participating in the offering
may be reclaimed if securities sold by them are repurchased in connection with stabilization transactions. The effect of these transactions
may be to stabilize or maintain the market price of our securities at a level above that which might otherwise prevail in the open market.
These transactions may be discontinued at any time.
The
underwriters, dealers and agents may engage in other transactions with us, or perform other services for us, in the ordinary course of
their business.
In
order to comply with the securities laws of certain states, if applicable, the securities offered by this prospectus may be sold in these
jurisdictions only through registered or licensed brokers or dealers. In addition, in certain states, the securities offered by this
prospectus may not be sold unless such securities have been registered or qualified for sale in these states or an exemption from registration
or qualification is available.
LEGAL
MATTERS
Except
as otherwise provided in any prospectus supplement, the validity of the issuance of the securities to be offered by this prospectus will
be passed upon for us by Certilman Balin Adler & Hyman, LLP, East Meadow, New York. As of February 6, 2023, members of Certilman
Balin Adler & Hyman, LLP owned 41 shares of our common stock. If legal matters in connection with offerings made pursuant to this
prospectus are passed upon by counsel for underwriters, dealers or agents, if any, such counsel will be named in the prospectus supplement
relating to such offering.
EXPERTS
Our
consolidated financial statements as of December 31, 2021 and 2020 and for the years then ended appearing in our Annual Report on Form
10-K for the year ended December 31, 2021 have been incorporated by reference in this prospectus in reliance upon the report of Friedman
LLP, an independent registered public accounting firm, incorporated by reference herein, and upon the authority of said firm as experts
in accounting and auditing.
$3,700,000

Common
Stock
____________________________
PROSPECTUS
SUPPLEMENT
____________________________
JonesTrading
____________________________
April
14, 2023
BioRestorative Therapies (NASDAQ:BRTX)
Historical Stock Chart
From Jun 2024 to Jul 2024
BioRestorative Therapies (NASDAQ:BRTX)
Historical Stock Chart
From Jul 2023 to Jul 2024