Bionano Announces Third OEM Partner has Received China NMPA Approval for DNA Isolation Products for IVD Use of OGM
September 21 2023 - 8:00AM

Bionano Genomics, Inc. (Nasdaq: BNGO) today announced that its
Chinese OEM partner, Ecobono, a distribution company focused on
reproductive health products, has obtained reagent Class I
registration from China's NMPA for Bionano's DNA isolation
products. Ecobono is Bionano’s third Chinese OEM partner to obtain
approval from NMPA for Bionano’s OGM solutions.
NMPA issued an approval to Ecobono for Bionano’s SP-G2 DNA
isolation kit. Ecobono received an IVD Class I label, enabling the
reagents to be used for IVD in China.
“Ecobono is a company that focuses on reproductive clinical and
nucleic acid testing. Currently, structural variation detection in
reproductive clinical research mainly includes karyotype analysis,
fluorescence in situ hybridization (FISH) and other cytogenetic
methods, as well as molecules such as copy number variation
microarray chips. New optical mapping technologies or
third-generation sequencing of ultra-long molecules can unify
cellular and molecular genetic approaches in the clinical field.
Detection with high molecular weight nucleic acids has become the
key to the success of the new methodology. Due to the particularity
of clinical samples, the reagents and instruments used need to be
registered and approved by NMPA. We are very pleased to work with
Bionano on the IVD registration of the Class I reagents. This
approval is the first milestone of our partnership. We look forward
to more potential milestones for the registration of additional
reagents and instruments in the future,” stated Wenli Wang, chief
executive officer of Ecobono.
Erik Holmlin, PhD, president and chief executive officer of
Bionano, commented, “We want to congratulate Ecobono, our third OEM
partner to receive NMPA approval for our reagents. This approval is
an important first step as Ecobono advances its registration
strategy for Bionano products. We believe the expansion of NMPA
approvals can help make OGM available for research into
reproductive health in China, an important market for both Bionano
and Ecobono."
About Bionano
Bionano is a provider of genome analysis solutions that can
enable researchers and clinicians to reveal answers to challenging
questions in biology and medicine. The Company’s mission is to
transform the way the world sees the genome through OGM solutions,
diagnostic services and software. The Company offers OGM solutions
for applications across basic, translational and clinical research.
Through its Lineagen, Inc. d/b/a Bionano
Laboratories business, the Company also provides diagnostic
testing for patients with clinical presentations consistent with
autism spectrum disorder and other neurodevelopmental disabilities.
The Company also offers an industry-leading, platform-agnostic
software solution, which integrates next-generation sequencing and
microarray data designed to provide analysis, visualization,
interpretation and reporting of copy number variants,
single-nucleotide variants and absence of heterozygosity across the
genome in one consolidated view. The Company additionally offers
nucleic acid extraction and purification solutions using
proprietary isotachophoresis (ITP) technology. For more
information,
visit www.bionano.com, www.bionanolaboratories.com or www.purigenbio.com.
Unless specifically identified, Bionano’s OGM products are for
research use only and not for use in diagnostic procedures.
Forward-Looking Statements of Bionano
This press release contains forward-looking statements within
the meaning of the Private Securities Litigation Reform Act of
1995. Words such as “believe,” “can,” “will” and similar
expressions (as well as other words or expressions referencing
future events, conditions or circumstances) convey uncertainty of
future events or outcomes and are intended to identify these
forward-looking statements. Forward-looking statements include
statements regarding our intentions, beliefs, projections, outlook,
analyses or current expectations, concerning, among other things:
the impact and utility of OGM in the analysis of samples for in
vitro fertilization or other reproductive health purposes; the
ability of new optical mapping technologies or third-generation
sequencing of ultra-long molecules to unify cellular and molecular
genetic approaches in the clinical field; the achievement of future
milestones by Ecobono in its partnership and registration strategy
with Bionano; the impact of NMPA approvals or the adoption of our
OGM solutions for reproductive health or other purposes; the
anticipated benefits of Bionano’s platform for our OEM partners in
China and the ultimate success of our OEM partners; and the
expected growth of sales of the Company’s OGM systems in China.
Each of these forward-looking statements involves risks and
uncertainties. Actual results or developments may differ materially
from those projected or implied in these forward-looking
statements. Factors that may cause such a difference include the
risks and uncertainties associated with: the impact of geopolitical
and macroeconomic developments, such as recent and potential future
bank failures, the ongoing Ukraine-Russian conflict, and
related sanctions, and any global pandemics, on our business and
the global economy; execution of our stated strategies and plans,
including those related to our OEM partners in China; general
market conditions; changes in the competitive landscape and the
introduction of competitive technologies or improvements to
existing technologies; changes in our strategic and commercial
plans; our ability to obtain sufficient financing to fund our
strategic plans and commercialization efforts and our ability to
continue as a “going concern”; the ability of medical and research
institutions to obtain funding to support adoption or continued use
of our technologies; failure of our OGM solutions to be adopted for
the analysis of samples for reproductive health applications;
failure of our OGM solutions to provide the anticipated benefits in
the identification of structural variants relevant to reproductive
health; failure of our OEM partners to execute on their commercial
plans; failure of NMPA approvals to drive adoption or use of our
OGM solutions for reproductive health applications; and the risks
and uncertainties associated with our business and financial
condition in general, including the risks and uncertainties
described in our filings with the Securities and Exchange
Commission, including, without limitation, our Annual Report on
Form 10-K for the year ended December 31, 2022 and in
other filings subsequently made by us with the Securities and
Exchange Commission. All forward-looking statements contained in
this press release speak only as of the date on which they were
made and are based on management’s assumptions and estimates as of
such date. We do not undertake any obligation to publicly update
any forward-looking statements, whether as a result of the receipt
of new information, the occurrence of future events or
otherwise.
CONTACTSCompany Contact:Erik
Holmlin, CEOBionano Genomics, Inc.+1 (858)
888-7610eholmlin@bionano.com
Investor Relations:David HolmesGilmartin
Group+1 (858) 888-7625IR@bionano.com
Bionano Genomics (NASDAQ:BNGO)
Historical Stock Chart
From Jun 2024 to Jul 2024
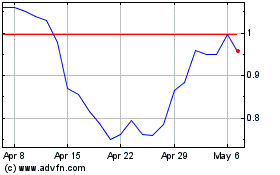
Bionano Genomics (NASDAQ:BNGO)
Historical Stock Chart
From Jul 2023 to Jul 2024