FREMONT, Calif., June 13, 2017 /PRNewswire/ -- Asterias
Biotherapeutics, Inc. (NYSE MKT: AST), a biotechnology company
pioneering the field of regenerative medicine, today announced that
new 9-month follow-up data from the AIS-A 10 million cell cohort in
the company's ongoing SCiStar Phase 1/2a clinical trial shows three
of six (50%) patients have now recovered two levels of motor
function and previously-announced improvements in arm, hand and
finger function at 3-months and 6-months following administration
of AST-OPC1 have been confirmed and further increased at
9-months.
"The new efficacy results show that previously reported
meaningful improvements in arm, hand and finger function in the 10
million cell cohort treated with AST-OPC1 cells have been
maintained and in some patients have been further enhanced
even 9 months following dosing," stated Dr. Edward Wirth III, Chief Medical Officer. "Gains
in motor function, such as the improvements observed in the SCiStar
study to date, have been shown to increase a patient's ability to
function independently following complete cervical spinal cord
injuries. We are increasingly encouraged by these continued
positive results, which are remarkable compared with spontaneous
recovery rates observed in a closely matched untreated patient
population."
Jane S. Lebkowski, Ph.D.,
Asterias' President of R&D and Chief Scientific Officer, will
present the 9-month efficacy and safety data on the AIS-A 10
million cell cohort later today during the International Society
for Stem Cell Research (ISSCR) 2017 Annual Meeting held in
Boston, MA. The full slide
presentation will be available at
http://asteriasbiotherapeutics.com/inv_events_presentations.php.
9-Month Follow-up Results
Improvements in upper extremity motor function are being
measured using the International Standards for Neurological
Classification of Spinal Cord Injury (ISNCSCI) scale, widely used
to quantify functional status of patients with spinal cord
injuries. Both subjects and physicians consistently report that
improvements in upper extremity motor function are the most
desirable functional improvement target in the quadriplegic
population, as they can have a significant impact on functional
independence, quality of life and cost of care from even relatively
modest changes. The SCiStar study is monitoring two separate
ISNCSCI measurements of upper extremity motor function. The upper
extremity motor score (UEMS), is a linear scale used to quantify
motor function at each of five upper extremity muscle groups
driving arm and hand function; these scores are also used to
determine "motor levels", which define the level within the cord
above which a subject has normal function. As suggested by
existing research, patients with severe spinal cord injuries that
show two motor levels of improvement on at least one side may
regain the ability to perform daily activities such as feeding,
dressing and bathing, which significantly reduces the overall level
of daily assistance needed for the patient and associated
healthcare costs.
A total of six patients were enrolled and dosed in the AIS-A 10
million-cell cohort, with five of six patients completing a 9-month
follow-up visit. The results include the following highlights:
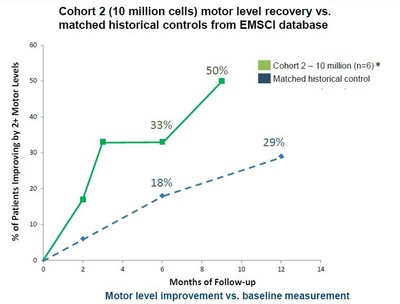
- Motor Level Improvement – Additional motor level
improvement was seen in the AIS-A 10 million cell cohort at 9
months.
-
- Three of six patients (50%) achieved two motor levels of
improvement over baseline on at least one side as of their latest
follow-up visit through 9 months. This compares to two of six
patients (33%) that had improved two motor levels on at least one
side through 3- and 6-months of follow-up.
- In addition, all six patients (100%) achieved at least one
motor level of improvement on at least one side as of their latest
follow-up through 9-months.
- Upper Extremity Motor Score – Additional improvement in
the average UEMS score for this cohort was observed at 9 months.
The average UEMS improvement at 9 months was 11.2 points, compared
to 9.7 points at 6 months.
- Matched Historical Control Data - The 9 month results
show a meaningful improvement in the motor function recovery in the
AIS-A patients receiving 10 million AST-OPC1 cells compared to a
historical control group of 62 closely matched patients from the
EMSCI database. In the historical matched control 29% of patients
recovered two motor levels on at least one side 12 months after
baseline compared to the 50% of the AIS-A patients receiving
AST-OPC1 that have recovered two motor levels on at least one side
9 months after baseline.
- Safety - The trial results to date continue to indicate
a positive safety profile for AST-OPC1. There have been no serious
adverse events related to AST-OPC1 and data from the study indicate
that AST-OPC1 can be safely administered to patients in the
subacute period after severe cervical spinal cord injury.
As previously reported, Asterias will report 12-month efficacy
and safety data from the AIS-A 10 million cell cohort sometime late
in the third quarter 2017 after the 12-month results are collected
for the entire cohort.
Each year in the U.S. more than 17,000 people suffer a severe,
debilitating spinal cord injury. These injuries can be devastating
to quality of life and ability to function independently. Lifetime
healthcare costs for these patients can often approach $5 million. Improvements in arm, hand and finger
functional capabilities in these patients can result in lower
healthcare costs, significant improvements in quality of life,
increased ability to engage in activities of daily living, and
increased
independence.
About the SCiStar Trial
The SCiStar trial is an open-label, single-arm trial testing
three sequential escalating doses of AST-OPC1 administered at up to
20 million AST-OPC1 cells in as many as 35 patients with sub-acute,
C-5 to C-7, motor complete (AIS-A or AIS-B) cervical SCI. These
individuals have essentially lost all movement below their injury
site and experience severe paralysis of the upper and lower limbs.
AIS-A patients have lost all motor and sensory function below their
injury site, while AIS-B patients have lost all motor function but
may retain some minimal sensory function below their injury site.
AST-OPC1 is being administered 14 to 30 days post-injury. Patients
will be followed by neurological exams and imaging procedures to
assess the safety and activity of the product.
The study is being conducted at six centers in the U.S. and the
company plans to increase this to up to 12 sites to accommodate the
expanded patient enrollment. Clinical sites involved in the study
include the Medical College of
Wisconsin in Milwaukee,
Shepherd Medical Center in Atlanta, University of
Southern California (USC)
jointly with Rancho Los Amigos National Rehabilitation Center in
Los Angeles, Indiana University, Rush
University Medical Center in Chicago and Santa Clara Valley Medical Center
in San Jose jointly with
Stanford University.
Asterias has received a Strategic Partnerships Award grant from
the California Institute for Regenerative Medicine, which provides
$14.3 million of non-dilutive funding
for the Phase 1/2a clinical trial and other product development
activities for AST-OPC1.
Additional information on the Phase 1/2a trial, including trial
sites, can be found at www.clinicaltrials.gov, using Identifier
NCT02302157, and at the SCiStar Study Website
(www.SCiStar-study.com).
About AST-OPC1
AST-OPC1, an oligodendrocyte progenitor population derived from
human embryonic stem cells, has been shown in animals and in vitro
to have three potentially reparative functions that address the
complex pathologies observed at the injury site of a spinal cord
injury. These activities of AST-OPC1 include production of
neurotrophic factors, stimulation of vascularization, and induction
of remyelination of denuded axons, all of which are critical for
survival, regrowth and conduction of nerve impulses through axons
at the injury site. In preclinical animal testing, AST-OPC1
administration led to remyelination of axons, improved hindlimb and
forelimb locomotor function, dramatic reductions in injury-related
cavitation and significant preservation of myelinated axons
traversing the injury site.
In a previous Phase 1 clinical trial, five patients with
neurologically complete, thoracic spinal cord injury were
administered two million AST-OPC1 cells at the spinal cord injury
site 7-14 days post-injury. They also received low levels of
immunosuppression for the next 60 days. Delivery of AST-OPC1 was
successful in all five subjects with no serious adverse events
associated with AST-OPC1. No evidence of rejection of AST-OPC1 was
observed in detailed immune response monitoring of all patients. In
four of the five patients, serial MRI scans indicated that reduced
spinal cord cavitation may have occurred. Based on the results of
this study, Asterias received clearance from FDA to progress
testing of AST-OPC1 to patients with cervical spine injuries, which
represents the first targeted population for registration
trials.
About Asterias Biotherapeutics
Asterias Biotherapeutics, Inc. is a biotechnology company
pioneering the field of regenerative medicine. The company's
proprietary cell therapy programs are based on its pluripotent stem
cell and immunotherapy platform technologies. Asterias is presently
focused on advancing three clinical-stage programs which have the
potential to address areas of very high unmet medical need in the
fields of neurology and oncology. AST-OPC1 (oligodendrocyte
progenitor cells) is currently in a Phase 1/2a dose escalation
clinical trial in spinal cord injury. AST-VAC1 (antigen-presenting
autologous dendritic cells) is undergoing continuing development by
Asterias based on promising efficacy and safety data from a Phase 2
study in Acute Myeloid Leukemia (AML), with current efforts focused
on streamlining and modernizing the manufacturing process. AST-VAC2
(antigen-presenting allogeneic dendritic cells) represents a second
generation, allogeneic cancer immunotherapy. The company's research
partner, Cancer Research UK, plans to begin a Phase 1/2a clinical
trial of AST-VAC2 in non-small cell lung cancer in 2017. Additional
information about Asterias can be found at
www.asteriasbiotherapeutics.com.
FORWARD-LOOKING STATEMENTS
Statements pertaining to future financial and/or operating
and/or clinical research results, future growth in research,
technology, clinical development, and potential opportunities for
Asterias, along with other statements about the future
expectations, beliefs, goals, plans, or prospects expressed by
management constitute forward-looking statements. Any statements
that are not historical fact (including, but not limited to
statements that contain words such as "will," "believes," "plans,"
"anticipates," "expects," "estimates") should also be considered to
be forward-looking statements. Forward-looking statements involve
risks and uncertainties, including, without limitation, risks
inherent in the development and/or commercialization of potential
products, uncertainty in the results of clinical trials or
regulatory approvals, need and ability to obtain future capital,
and maintenance of intellectual property rights. Actual results may
differ materially from the results anticipated in these
forward-looking statements and as such should be evaluated together
with the many uncertainties that affect the businesses of Asterias,
particularly those mentioned in the cautionary statements found in
Asterias' filings with the Securities and Exchange Commission.
Asterias disclaims any intent or obligation to update these
forward-looking statements.
To view the original version on PR Newswire,
visit:http://www.prnewswire.com/news-releases/patients-with-complete-paralysis-show-additional-recovery-of-arm-hand-and-finger-function-at-9-months-after-treatment-with-asterias-ast-opc1-300472759.html
SOURCE Asterias Biotherapeutics, Inc.