Marinus Appoints Seth H.Z. Fischer to its Board of Directors
September 07 2016 - 7:30AM

Marinus Pharmaceuticals, Inc. (Nasdaq:MRNS), a biopharmaceutical
company dedicated to the development of innovative therapeutics to
treat epilepsy and neuropsychiatric disorders, today announced the
appointment of Seth H.Z. Fischer to its Board of Directors.
“Seth brings over 30 years of pharmaceutical
operations and commercialization experience,” stated Christopher M.
Cashman, chairman and chief executive officer of Marinus
Pharmaceuticals. “Seth has successfully advanced and commercialized
a wide range of therapeutics, including Topamax® in epilepsy and
migraines. We will look to leverage his significant knowledge as we
advance ganaxolone in status epilepticus and rare pediatric genetic
indications. I join my fellow Board members in welcoming Seth to
our Board of Directors.”
Mr. Fischer is presently CEO and Director of
VIVUS, Inc., a publicly traded biopharmaceutical company
commercializing and developing innovative, next-generation
therapies to address unmet needs in obesity and sexual health.
Prior to VIVUS, Mr. Fischer served in various positions at Johnson
& Johnson, most recently as Company Group Chairman, Johnson
& Johnson and Worldwide Franchise Chairman of Cordis
Corporation. Prior to that he served as Company Group
Chairman, North America Pharmaceuticals, which included
responsibilities for Ortho-McNeil Pharmaceuticals, Janssen and
Scios and prior to that, Mr. Fischer served as President of
Ortho-McNeil Pharmaceuticals.
“Ganaxolone is a promising CNS-selective GABAA
modulator that has the potential to make a meaningful impact on the
lives of patients diagnosed with difficult-to-treat epileptic
conditions, especially those suffering from rare diseases with
limited treatment options,” commented Mr. Fischer. “I look forward
to joining the other Board members in providing guidance to the
Marinus executive team as the company continues to advance their
oral and IV formulations of ganaxolone through clinical trials and
to commercialization.”
In addition to the commercialization of Topamax
(topiramate) for epilepsy and migraines, Mr. Fischer’s operating
responsibilities at Johnson & Johnson and its subsidiaries
encompassed the commercialization of products in the neurologic,
analgesic, anti-infective, cardiovascular, psychiatric and women’s
health areas. He is a member of the Boards of Directors of Agile
Therapeutics, Inc. and BioSig Technologies, Inc., and an advisor to
MedHab, LLC. Mr. Fischer earned a bachelor’s degree in general
studies from Ohio University and served as a captain in the U.S.
Air Force.
Mr. Fischer will fill the board seat previously
held by Anand Mehra, M.D. of Sofinnova Ventures, who has served as
a member of the Company’s Board of Directors since 2007 and has
stepped down from his role as a director of Marinus Pharmaceuticals
in order to devote his full time and efforts to other
commitments.
“I would like to thank Dr. Mehra for his nearly
decade of service and many valuable contributions during his tenure
as a board member,” commented Mr. Cashman. “We wish him
continued personal and career success in his future endeavors.”
About Marinus
Pharmaceuticals
Marinus Pharmaceuticals, Inc. is a
biopharmaceutical company dedicated to the development of
ganaxolone, which offers a new mechanism of action, demonstrated
efficacy and safety and convenient dosing, to improve the lives of
patients suffering from epilepsy and neuropsychiatric disorders.
Ganaxolone is a CNS-selective GABAA modulator that acts on a
well-characterized target in the brain known to have both
anti-seizure and anti-anxiety effects. Ganaxolone is being
developed in three different dose forms (IV, capsule and liquid)
intended to maximize therapeutic reach to adult and pediatric
patient populations in both acute and chronic care settings.
Ganaxolone IV is in a Phase 1 clinical trial to treat status
epilepticus. Ganaxolone IV is complemented by its oral dose forms,
providing the potential for IV-to-oral continuation therapy for
patients transitioning from acute care to outpatient settings.
Ganaxolone capsule and liquid is being studied in orphan pediatric
indications with comorbidities in seizures and behavior disorders –
PCDH19, CDKL5, and Fragile X Syndrome. For more information visit
www.marinuspharma.com.
Forward-Looking Statements
To the extent that statements contained in this
press release are not descriptions of historical facts regarding
Marinus, they are forward-looking statements reflecting the current
beliefs and expectations of management made pursuant to the safe
harbor provisions of the Private Securities Litigation Reform Act
of 1995. Words such as “may”, “will”, “expect”, “anticipate”,
“estimate”, “intend”, “believe”, and similar expressions (as well
as other words or expressions referencing future events, conditions
or circumstances) are intended to identify forward-looking
statements. Examples of forward looking statements contained
in this press release include, among others, statements regarding
our interpretation of preclinical studies, development plans for
our product candidate, including the development of dose forms, the
clinical trial testing schedule and milestones, the ability to
complete enrollment in our clinical trials, interpretation of
scientific basis for ganaxolone use, timing for availability and
release of data, the safety, potential efficacy and therapeutic
potential of our product candidate and our expectation regarding
the sufficiency of our working capital. Forward-looking statements
in this release involve substantial risks and uncertainties that
could cause our clinical development programs, future results,
performance or achievements to differ significantly from those
expressed or implied by the forward-looking statements. Such
risks and uncertainties include, among others, the uncertainties
inherent in the conduct of future clinical trials, the timing of
the clinical trials, enrollment in clinical trials, availability of
data from ongoing clinical trials, expectations for regulatory
approvals, and other matters, including the development of
formulations of ganaxolone, that could affect the availability or
commercial potential of our drug candidates. Marinus
undertakes no obligation to update or revise any forward-looking
statements. For a further description of the risks and
uncertainties that could cause actual results to differ from those
expressed in these forward-looking statements, as well as risks
relating to the business of the Company in general, see filings
Marinus has made with the Securities and Exchange Commission.
CONTACT:
Company:
Lisa M. Caperelli
Senior Director, Investor Relations & Corporate Communications
Marinus Pharmaceuticals, Inc.
484-801-4674
lcaperelli@marinuspharma.com
Media Contact:
Tiberend Strategic Advisors, Inc.
Amy S. Wheeler
646-362-5750
awheeler@tiberend.com
Marinus Pharmaceuticals (NASDAQ:MRNS)
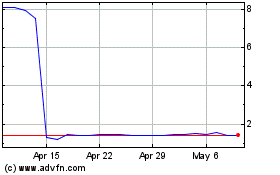
Historical Stock Chart
From Apr 2024 to May 2024
Marinus Pharmaceuticals (NASDAQ:MRNS)
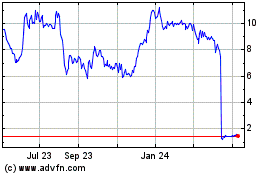
Historical Stock Chart
From May 2023 to May 2024