Finch Therapeutics Provides Corporate Updates and Reports Second Quarter 2022 Financial Results
August 11 2022 - 7:00AM

Finch Therapeutics Group, Inc. (“Finch” or “Finch Therapeutics”)
(Nasdaq: FNCH), a clinical-stage microbiome therapeutics company
leveraging its Human-First Discovery® platform to develop a novel
class of orally administered biological drugs, today provided
corporate updates and reported financial results for the second
quarter ended June 30, 2022.
“We are excited by the advances we made this
quarter related to our lead program in recurrent C. difficile
infection, including significant progress on a number of activities
that we expect will position us to proceed with enrollment in our
PRISM4 Phase 3 trial of CP101 later this year. We are also pleased
to be presenting microbiome data from our PRISM-EXT Phase 2
open-label trial of CP101 in recurrent C. difficile at ACG 2022,”
said Mark Smith, PhD, Chief Executive Officer of Finch
Therapeutics.
Dr. Smith continued, “Additionally, we are
preparing to submit the IND for FIN-211 in children with autism and
significant GI symptoms, with the submission anticipated later this
year. We look forward to continuing to advance our mission to
harness the microbiome to transform outcomes for people impacted by
conditions linked to microbiome disruption.”
Recent Program Highlights
CP101 for Prevention of Recurrent
C. difficile Infection (CDI):
- Continued Progress with Enrollment-Enabling Activities
for the PRISM4 Phase 3 Trial of CP101: Finch has made
significant progress preparing to proceed with enrollment in
PRISM4, including progress on several manufacturing activities and
working with our clinical sites in preparation for PRISM4.
Additionally, Finch recently submitted for the FDA’s review and
agreement the validation package for one of its release tests and a
PRISM4 protocol amendment that reflects previously announced
changes to the PRISM4 protocol. Pending feedback from the FDA on
this recent submission and the completion of certain remaining
quality and manufacturing activities, Finch expects to proceed with
enrollment in PRISM4.
- Clinical Data from PRISM-EXT and PRISM3 Phase 2 Trials
Presented at Digestive Disease Week (DDW) 2022: In an oral
presentation, Finch presented topline efficacy and safety data from
the PRISM-EXT Phase 2 open-label trial of CP101 in recurrent CDI.
As previously announced, 80.3% and 78.8% of participants who
received CP101 following standard-of-care (SOC) antibiotics in
PRISM-EXT were without CDI recurrence through 8 weeks and 24 weeks
post-treatment, respectively (n=132). In a poster presentation,
Finch presented a combined, post-hoc analysis of data from the
PRISM3 Phase 2 placebo-controlled trial and the PRISM-EXT trial.
Across PRISM3 and PRISM-EXT, 85.0% of participants who received
CP101 following SOC antibiotics were without CDI recurrence through
8 weeks (n=214). No drug-related serious adverse events were
reported in any participants that received CP101 in PRISM3 or
PRISM-EXT. Drug-related treatment emergent adverse events were mild
(Grade 1) or moderate (Grade 2), and primarily gastrointestinal in
nature. The topline data presented at DDW 2022 are available under
the ‘Publications’ section of the Finch website.
- Translational Results from the PRISM-EXT Phase 2 Trial
Accepted for Presentation at ACG 2022: Today, Finch
announced that microbiome diversity and engraftment data from the
PRISM-EXT trial were accepted for presentation at the American
College of Gastroenterology (ACG) 2022 Annual Scientific Meeting
taking place October 21-26, 2022.
- Anticipated Upcoming Milestones: Finch expects
to proceed with enrollment in PRISM4 in H2 2022 and anticipates
that topline data from PRISM4 will be available in H1 2024.
FIN-211 for Autism Spectrum Disorder (ASD)
with Significant Gastrointestinal (GI) Symptoms:
- Continued Progress Preparing for the FIN-211 IND
Submission and AUSPIRE Phase 1b Trial: Finch is preparing
for the submission of the FIN-211 investigational new drug (IND)
application and the AUSPIRE Phase 1b trial of FIN-211 in children
with ASD and constipation. The AUSPIRE trial is designed to enroll
approximately 36 children who will be randomized to receive either
FIN-211 or placebo daily for eight weeks. The primary endpoint of
the trial will be safety and tolerability, with secondary endpoints
including behavioral and GI symptom assessment. Exploratory
endpoints will include additional behavioral and GI endpoints, as
well as pharmacokinetic and pharmacodynamic assessments.
- Anticipated Upcoming Milestones: Finch
anticipates submitting the IND for FIN-211 in Q4 2022. Finch plans
to provide further guidance on the expected timing of the AUSPIRE
trial in the future.
Second Quarter 2022 Financial
Results
- Finch reported a net loss of $22.7 million for the second
quarter of 2022, compared to a net loss of $15.2 million for the
same period in 2021. The net loss was driven by an increase in
operating expenses of $3.1 million compared to the second quarter
of 2021, in addition to a decrease in revenue of $2.5 million,
primarily due to the August 2021 amendment to the Takeda Agreement,
pursuant to which Finch transitioned primary responsibilities for
TAK-524 to Takeda in the third quarter of 2021, resulting in a
decrease in collaboration revenue in the current quarter.
Additionally, the prior period benefitted from other income due to
the gain on extinguishment of the PPP Loan of $1.8 million, while
in the current quarter the Company incurred other expense of $0.1
million.
- Research and development (R&D) expenses were relatively
flat, with $13.9 million of expenses for the second quarter of
2022, compared to $14.0 million for the same period in 2021. This
is due to an increase in platform related expenses including
personnel and manufacturing costs, as Finch continues to build out
its manufacturing platform. This increase was offset by a decrease
in costs related to Finch’s inflammatory bowel disease program due
to the transition of primary responsibilities for TAK-524 from
Finch to Takeda in the third quarter of 2021, in addition to a
decrease in costs associated with Finch’s previously announced
decision to suspend its chronic hepatitis B virus program.
- General and administrative (G&A) expenses were $8.2 million
for the second quarter of 2022, compared to $5.9 million for the
same period in 2021. The increase in G&A expense
quarter-over-quarter was primarily due to an increase in
professional fees, in addition to an increase in facility-related
costs and stock-based compensation expense. This increase was
partially offset by a decrease in consulting related expenses.
- Finch’s cash and cash equivalents as of June 30, 2022 was
$104.7 million, compared to $133.5 million as of December 31, 2021.
Finch believes its cash and cash equivalents on hand as of June 30,
2022, together with income under an executed sublease for a portion
of one of its office and lab facilities, will fund its operations
into Q1 2024. As the Takeda-partnered TAK-524 program nears the
next decision point in development, Takeda has informed Finch that
they are conducting a review of the plans, timeline, and budget for
the TAK-524 program as part of their portfolio review process. As a
result, the associated near-term milestones have been removed from
Finch’s cash runway analysis until Takeda has completed its
review.
About Finch Therapeutics
Finch Therapeutics is a clinical-stage microbiome
therapeutics company leveraging its Human-First Discovery® platform
to develop a novel class of orally administered biological drugs.
With the capabilities to develop both complete and targeted
microbiome therapeutics, Finch is advancing a rich pipeline of
candidates designed to address a wide range of unmet medical needs.
Finch’s lead candidate, CP101, is in late-stage clinical
development for the prevention of recurrent C. difficile infection
and has received Breakthrough Therapy and Fast Track designations
from the U.S. Food and Drug Administration. Finch is also
developing FIN-211 for children with autism spectrum disorder and
significant gastrointestinal symptoms. Finch has a partnership with
Takeda focused on the development of targeted microbiome
therapeutics for inflammatory bowel disease. Finch routinely posts
information that may be important to its investors on its website
at www.finchtherapeutics.com. Finch encourages investors to consult
the “Investors & News” section of its website regularly.
Human-First Discovery® is a registered trademark
of Finch Therapeutics Group, Inc.
Forward-Looking Statements
This press release includes “forward-looking
statements.” Words such as “will,” "anticipates," "believes,"
"expects," "intends," “plans,” “potential,” "projects,” “would” and
"future" or similar expressions are intended to identify
forward-looking statements. These forward-looking statements
include, but are not limited to, statements regarding: Finch’s
ability to advance its mission; Finch’s ability to complete
activities that it expects will position it to proceed with
enrollment in PRISM4 later this year; Finch’s plans to advance
FIN-211 into the clinic; the timeline for topline data from PRISM4
and plans to present translational data from PRISM-EXT; the
timeline for submission of the IND for FIN-211; the design and
anticipated enrollment in AUSPIRE; and Finch's anticipated runway.
Because such statements are subject to risks and uncertainties,
actual results may differ materially from those expressed or
implied by such forward-looking statements. These risks and
uncertainties include, among others: the risk that correspondence
from the FDA may require Finch to collect additional data or
information beyond what it currently expects, as well as unexpected
regulatory actions or delays, including requests for additional
safety and/or efficacy data or analysis of data, or government
regulation generally; uncertainties relating to regulatory
applications and related filing and approval timelines; Finch’s
limited operating history and historical losses; Finch’s ability to
raise additional funding to complete the development and any
commercialization of its product candidates; Finch’s dependence on
the success of its lead product candidate, CP101; the possibility
that Finch may be delayed in initiating, enrolling or completing
any clinical trials; results of clinical trials may not be
indicative of final or future results from later stage or larger
clinical trials (or in broader patient populations once the product
is approved for use by regulatory agencies) or may not be favorable
or may not support further development; Finch’s product candidates,
including CP101 and FIN-211 may not generate the benefits to
patients that are anticipated; results of clinical trials may not
be sufficient to satisfy regulatory authorities to approve Finch’s
product candidates in their targeted or other indications (or such
authorities may request additional trials or additional
information); Finch’s ability to comply with regulatory
requirements; ongoing regulatory obligations and continued
regulatory review may result in significant additional expense to
Finch and Finch may be subject to penalties for failure to comply;
competition from third parties that are developing products for
similar uses; Finch’s ability to maintain patent and other
intellectual property protection and the possibility that Finch’s
intellectual property rights may be infringed, invalid or
unenforceable or will be threatened by third parties; Finch’s
ability to qualify and scale its manufacturing capabilities in
anticipation of commencement of multiple global clinical trials;
Finch’s lack of experience in selling, marketing and distributing
its product candidates; Finch’s dependence on third parties in
connection with manufacturing, clinical trials and preclinical
studies; and risks relating to the impact and duration of the
COVID-19 pandemic on Finch’s business. These and other risks are
described more fully in Finch’s filings with the Securities and
Exchange Commission (“SEC”), including the section titled “Risk
Factors” in Finch’s Annual Report on Form 10-K for the year ended
December 31, 2021 filed with the SEC on March 31, 2022, as
supplemented by Finch’s Quarterly Report on Form 10-Q filed with
the SEC on May 16, 2022, as well as discussions of potential risks,
uncertainties, and other important factors in Finch’s other filings
with the SEC. All forward-looking statements contained in this
press release speak only as of the date on which they were made.
Except to the extent required by law, Finch undertakes no
obligation to update such statements to reflect events that occur
or circumstances that exist after the date on which they were
made.
Investor Contact:
Stephen Jasper Gilmartin Group (858) 525-2047
stephen@gilmartinir.com
Media Contact:
Jenna Urban Berry & Company Public Relations
(212) 253-8881 jurban@berrypr.com
Finch Therapeutics Group,
Inc.
Condensed Consolidated Statements of
Operations (Unaudited) (in thousands, except share
and per share data)
|
|
|
FOR THE THREE MONTHS ENDED
JUNE 30, |
|
|
FOR THE SIX MONTHS ENDED
JUNE 30, |
|
|
|
|
2022 |
|
|
2021 |
|
|
2022 |
|
|
2021 |
|
|
Revenue: |
|
|
|
|
|
|
|
|
|
|
|
|
|
Collaboration revenue |
|
$ |
361 |
|
|
$ |
2,830 |
|
|
$ |
715 |
|
|
$ |
6,383 |
|
|
Total revenue |
|
|
361 |
|
|
|
2,830 |
|
|
|
715 |
|
|
|
6,383 |
|
|
Operating expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
Research and development |
|
|
13,923 |
|
|
|
13,964 |
|
|
|
29,453 |
|
|
|
26,939 |
|
|
General and administrative |
|
|
8,164 |
|
|
|
5,882 |
|
|
|
17,568 |
|
|
|
10,433 |
|
|
Restructuring expense |
|
|
903 |
|
|
|
- |
|
|
|
903 |
|
|
|
- |
|
|
Total operating expenses |
|
|
22,990 |
|
|
|
19,846 |
|
|
|
47,924 |
|
|
|
37,372 |
|
|
Loss from operations |
|
|
(22,629 |
) |
|
|
(17,016 |
) |
|
|
(47,209 |
) |
|
|
(30,989 |
) |
|
Other (expense) income |
|
|
(71 |
) |
|
|
1,847 |
|
|
|
(58 |
) |
|
|
1,839 |
|
|
Net loss |
|
$ |
(22,700 |
) |
|
$ |
(15,169 |
) |
|
$ |
(47,267 |
) |
|
$ |
(29,150 |
) |
|
Net loss per share attributable to common stockholders—basic and
diluted |
|
$ |
(0.48 |
) |
|
$ |
(0.32 |
) |
|
$ |
(0.99 |
) |
|
$ |
(0.95 |
) |
|
Weighted-average common stock outstanding—basic and diluted |
|
|
47,576,349 |
|
|
|
47,379,887 |
|
|
|
47,552,780 |
|
|
|
30,798,698 |
|
Finch Therapeutics Group, Inc.
Condensed Consolidated Balance Sheet Data
(Unaudited) (in thousands)
|
|
|
JUNE 30, 2022 |
|
|
DECEMBER 31, 2021 |
|
|
Assets: |
|
|
|
|
|
|
|
Cash and cash equivalents |
|
$ |
104,673 |
|
|
$ |
133,481 |
|
|
Other assets |
|
|
123,628 |
|
|
|
91,888 |
|
|
Total assets |
|
$ |
228,301 |
|
|
$ |
225,369 |
|
|
Liabilities, redeemable convertible preferred stock and
stockholders' equity |
|
|
|
|
|
|
|
Liabilities |
|
|
69,209 |
|
|
|
23,145 |
|
|
Stockholders' equity |
|
|
159,092 |
|
|
|
202,224 |
|
|
Total liabilities, redeemable convertible preferred stock and
stockholders' equity |
|
$ |
228,301 |
|
|
$ |
225,369 |
|
Finch Therapeutics (NASDAQ:FNCH)
Historical Stock Chart
From Jun 2024 to Jul 2024
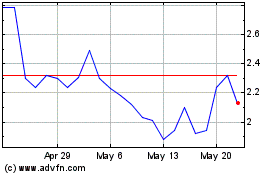
Finch Therapeutics (NASDAQ:FNCH)
Historical Stock Chart
From Jul 2023 to Jul 2024