Easy-to-use at-home test provides results in
ten minutes with a doctor’s prescription
Quidel Corporation (NASDAQ: QDEL) (“Quidel”), a leading
provider of rapid diagnostic testing solutions, cellular-based
virology assays and molecular diagnostic systems, announced today
that it has received an Emergency Use Authorization (EUA) from the
U.S. Food and Drug Administration (FDA), allowing the company to
market its new QuickVue® At-Home COVID-19 Test for the
qualitative detection of the nucleocapsid protein antigen from
SARS-CoV-2. This test is authorized for prescription home use with
self-collected (unobserved) anterior nares (NS) swab specimens
directly from individuals aged 14 years and older who are suspected
of COVID-19 by their healthcare provider within the first six days
of the onset of symptoms. This test is also authorized for
prescription home use with adult-collected anterior NS samples
directly from individuals aged 8 years or older who are suspected
of COVID-19 by their healthcare provider within the first six days
of the onset of symptoms.
Quidel has provided rapid COVID-19 testing through doctors and
health care providers throughout the pandemic, manufacturing and
shipping millions of Sofia® tests last year. Now, Quidel’s
QuickVue® At-Home COVID-19 Test will allow patients with a
prescription to easily perform the test themselves and get results
in 10 minutes. The QuickVue® At-Home COVID-19 Test shows excellent
performance, with positive results agreeing with PCR 84.8% of the
time, and negative results agreeing 99.1% of the time, delivering
confidence to patients running the test and facilitating informed
discussions with doctors.
“Quidel’s employees have truly risen to the challenge of the
SARS pandemic. Our mission is to provide greater access to
frequent, affordable and highly accurate diagnostic testing, and
ultimately to enable individuals to take charge of their own health
and help them make prudent decisions to protect themselves and
their loved ones,” said Douglas Bryant, president and CEO of Quidel
Corporation. “This is an important first step in a two-step process
that, once additional ongoing studies are completed, will further
expand access to frequent testing at home.”
The QuickVue® At-Home COVID-19 Test employs the same Quidel
lateral flow technology used for decades by healthcare
professionals and features the same SARS CoV-2 rapid antigen test
strip and reagent solution that received an EUA from the FDA for
use in professional settings in December 2020. Quidel’s contract
with the National Institutes of Health’s Rapid Acceleration of
Diagnostics initiative was expanded in late 2020 to include the
QuickVue At-Home COVID-19 test, which expedited its emergency use
authorization.
Quidel recently started the buildout of a new manufacturing
facility in Carlsbad, CA. The 128,000 square foot facility is
expected to be the company’s highest-volume production facility and
begin operations in the second half of 2021, initially with a
mission to produce more than 50 million QuickVue® rapid antigen
tests per month, or 600 million tests per year at full
capacity.
“The flexibility of our QuickVue® At-Home COVID-19 Test for
meeting the urgent testing needs of individuals at home will save
time and enable doctors and telemedicine providers to determine
appropriate treatments without exposing their staff and other
patients to heightened risk of infection,” Mr. Bryant noted. “We
hope to bring the benefits of this technology to more broadly serve
consumers, school systems, businesses and remote communities in the
near future.”
Quidel’s QuickVue® brand launched in 1986 with visually read
rapid diagnostics focusing on women’s health and respiratory
diseases. In 1999, QuickVue® Influenza A+B was the first visually
read rapid test approved by the FDA for professional use. QuickVue®
was also the first flu test cleared by the FDA for use in
CLIA-Waived point-of-care facilities like doctors’ offices, urgent
care clinics and pharmacies. Today, QuickVue® is a market leading
platform in the professional segment for visually diagnosing
Influenza, respiratory syncytial virus, Strep A and a variety of
other illnesses. Since the launch of the QuickVue® brand into the
professional segment, more than 150 million QuickVue® diagnostic
tests have been sold.
The QuickVue At-Home COVID-19 Test is intended for self-use
and/or, as applicable for an adult lay user testing another person
aged 8 years or older in a non-laboratory setting. The QuickVue
At-Home COVID-19 Test is only for use under the Food and Drug
Administration’s Emergency Use Authorization. The QuickVue® At-Home
Covid-19 Test has not been FDA cleared or approved. The test has
been authorized only for the detection of proteins from SARS-CoV-2,
not for any other viruses or pathogens, and is only authorized for
the duration of the declaration that circumstances exist justifying
the authorization of emergency use of in vitro diagnostic tests for
detection and/or diagnosis of COVID-19 under Section 564(b)(1) of
the Federal Food, Drug and Cosmetic Act, 21 U.S.C. §
360bbb-3(b)(1), unless the declaration is terminated or
authorization is revoked sooner.
Visit www.quickvueathome.com for more information. For media
inquiries, contact media@quickvueathome.com.
Background Information:
Throughout this pandemic, Quidel has been at the forefront of
diagnostic innovation. From the company’s Lyra® PCR tests, to its
Sofia® rapid antigen tests for COVID-19, to its Sofia® “ABC”
combination test for the Influenza A+B and coronavirus, to its
QuickVue® SARS Antigen test authorized for emergency use by the FDA
for professional use and now available for at home prescription
use, Quidel has significantly advanced the science and the scale of
its operations.
Quidel was the first company to receive EUA from the FDA for a
rapid point-of-care antigen test for COVID-19 and remains at the
forefront of the battle against the coronavirus pandemic.
Sofia® 2 is Quidel’s best-selling cartridge-based,
instrument-read rapid diagnostic system for infectious disease
testing, which utilizes fluorescent chemistry design, an intuitive
graphical user interface, and optics system to provide a highly
accurate, objective and automated result in 15 minutes. The Sofia®
2 system also comes connected to Virena®, Quidel’s data management
system, which provides aggregated, de-identified testing data to
public health authorities in near real-time. In 2020, the company
increased the installed base of Sofia® diagnostic instruments to
more than 65,000 point-of-care locations.
In 2020, Quidel quickly increased its production of Sofia® rapid
antigen tests from approximately 500,000 units per week in March to
over 2 million tests per week in December. This year, Quidel is
building additional production lines with a goal to reach a
run-rate of over 240 million Sofia® tests per year by the third
quarter of 2021.
Quidel’s QuickVue® and Sofia® SARS Antigen test kits are made in
the greater San Diego metropolitan area of California.
This project is supported by the NIH Rapid Acceleration of
Diagnostics - Advanced Technology Platforms (RADx-ATP) program and
has been funded in whole or in part with Federal funds from the
Office of the Director, National Institutes of Health, Department
of Health and Human Services, under Contract No.
75N92020C00013.
About Quidel Corporation
Quidel Corporation (Nasdaq: QDEL) is a leading manufacturer of
diagnostic solutions at the point of care, delivering a continuum
of rapid testing technologies that further improve the quality of
health care throughout the globe. An innovator for over 40 years in
the medical device industry, Quidel pioneered the first FDA-cleared
point-of-care test for influenza in 1999 and was the first to
market a rapid SARS-CoV-2 antigen test in the U.S. Under trusted
brand names Sofia®, Solana®, Lyra®, Triage® and QuickVue®, Quidel’s
comprehensive product portfolio includes tests for a wide range of
infectious diseases, cardiac and autoimmune biomarkers, as well as
a host of products to detect COVID-19. With products made in
America, Quidel’s mission is to provide patients with immediate and
frequent access to highly accurate, affordable testing for the good
of our families, our communities and the world. For more
information about Quidel, visit quidel.com.
View our story told by our people at www.quidel.com/ourstory
Forward-Looking Statements
This press release contains forward-looking statements within
the meaning of the federal securities laws that involve material
risks, assumptions and uncertainties. Many possible events or
factors could affect our future results and performance, such that
our actual results and performance may differ materially from those
that may be described or implied in the forward-looking statements.
As such, no forward-looking statement can be guaranteed.
Differences in actual results and performance may arise as a result
of a number of factors including, without limitation: the impact
and duration of the COVID-19 global pandemic; competition from
other providers of diagnostic products; our ability to accurately
forecast demand for our products and products in development,
including in new market segments; our ability to develop new
technologies, products and markets and to commercialize new
products; our reliance on sales of our COVID-19 and influenza
diagnostic tests; our reliance on a limited number of key
distributors; quantity of our product in our distributors’
inventory or distribution channels; changes in the buying patterns
of our distributors; the financial soundness of our customers and
suppliers; lower than anticipated market penetration of our
products; third-party reimbursement policies and potential cost
constraints; our ability to meet demand for our products;
interruptions, delays or shortages in the supply of raw materials,
components and other products and services; failures in our
information technology and storage systems; our exposure to data
corruption, cyber-based attacks, security breaches and privacy
violations; international risks, including but not limited to,
economic, political and regulatory risks; continuing worldwide
political and social uncertainty; our development, acquisition and
protection of proprietary technology rights; intellectual property
risks, including but not limited to, infringement litigation; the
loss of Emergency Use Authorizations for our COVID-19 products and
failures or delays in receipt of reviews or regulatory approvals,
clearances or authorizations for new products or related to
currently-marketed products by the U.S. Food and Drug
Administration (the “FDA”) or other regulatory authorities or loss
of any previously received regulatory approvals, clearances or
authorizations or other adverse actions by regulatory authorities;
our contracts with government entities involve future funding,
compliance and possible sanctions risks; product defects; changes
in government policies and regulations and compliance risks related
thereto; our ability to manage our growth strategy and successfully
identify, acquire and integrate potential acquisition targets or
technologies and our ability to obtain financing; our acquisition
of Alere’s Triage® business presents certain risks to our business
and operations; the level of our deferred payment obligations; our
exposure to claims and litigation that could result in significant
expenses and could ultimately result in an unfavorable outcome for
us, including the ongoing litigation between us and Beckman
Coulter, Inc.; we may need to raise additional funds to finance our
future capital or operating needs; our debt, deferred and
contingent payment obligations; competition for and loss of
management and key personnel; business risks not covered by
insurance; changes in tax rates and exposure to additional tax
liabilities or assessments; and provisions in our charter documents
and Delaware law that might delay or impede stockholder actions
with respect to business combinations or similar transactions.
Forward-looking statements typically are identified by the use of
terms such as “may,” “will,” “should,” “might,” “expect,”
“anticipate,” “estimate,” “plan,” “intend,” “goal,” “project,”
“strategy,” “future,” and similar words, although some
forward-looking statements are expressed differently. The risks
described in reports and registration statements that we file with
the Securities and Exchange Commission from time to time, should be
carefully considered, including those discussed in Item 1A, “Risk
Factors” and elsewhere in our Annual Report on Form 10‑K for the
year ended December 31, 2020 and in our subsequent Quarterly
Reports on Form 10‑Q. You are cautioned not to place undue reliance
on these forward-looking statements, which reflect management’s
analysis only as of the date of this press release. Except as
required by law, we undertake no obligation to publicly release any
revision or update of these forward-looking statements, whether as
a result of new information, future events or otherwise.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210301005734/en/
Quidel Contact: Quidel Corporation Randy Steward Chief Financial
Officer (858) 552-7931
Investors Contact: Quidel Corporation Ruben Argueta (858)
646-8023 rargueta@quidel.com
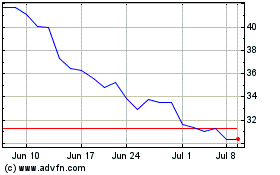
QuidelOrtho (NASDAQ:QDEL)
Historical Stock Chart
From Jun 2024 to Jul 2024
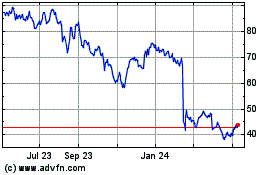
QuidelOrtho (NASDAQ:QDEL)
Historical Stock Chart
From Jul 2023 to Jul 2024