Integra LifeSciences Features the Integra(TM) Endoscopic Gastroc Release System (EGR) Product Line at the Annual ACFAS Meeting
March 09 2011 - 6:00AM

Integra LifeSciences Holdings Corporation (Nasdaq:IART) announced
today that it is featuring the Integra(TM) Endoscopic Gastroc
Release System (EGR) product line at the Annual Meeting of the
American College of Foot and Ankle Surgeons (ACFAS) on March 9-12,
in Fort Lauderdale, Florida. Integra's EGR System is a
disposable device with a unique articulating blade that allows
selective cutting of soft tissues. It is intended for use in
the surgical treatment for posterior heel cord, or equinus,
contracture. Integra will provide a hands-on demonstration,
complete with anatomic gastroc model and endoscope, at booth #101,
for surgeons to trial the EGR System.
"We've had excellent surgeon feedback on the EGR System," said
Pete Ligotti, Vice President of Marketing for Integra Extremity
Reconstruction. "The selective cutting provided by the
retractable blade system and the exceptional visualization have
exceeded their expectations, and have helped to limit uncertainty
when performing the procedure. We're really pleased that we can
give surgeons an opportunity to try out the device at the ACFAS
meeting."
Equinus contracture is a condition that limits ankle motion,
frequently due to contraction or tightness of the calf
muscle. Surgical treatment involves recession of selected
tissues of the calf muscle (composed of the gastrocnemius and
soleus muscle complex). Traditionally performed as an open
procedure, the recession of the gastrocnemius aponeurosis can be
performed endoscopically through a small incision. This is a
minimally invasive, less traumatic procedure that produces a
smaller and less apparent scar.
Gastrocnemius recession is being used increasingly as a
component in the surgical treatment of posterior tibial tendon
dysfunction (PTTD), diabetic forefoot ulcers, symptomatic acquired
flatfoot, and hallux valgus. Integra estimates that these
pathologies affect tens of thousands of patients
annually.
The EGR System is sold by Integra's Extremity Reconstruction
sales organization, which focuses on lower extremity fixation,
upper extremity fixation, tendon protection, peripheral nerve
repair/protection and wound repair.
Integra LifeSciences, a world leader in medical devices, is
dedicated to limiting uncertainty for surgeons, so they can
concentrate on providing the best patient care. Integra offers
innovative solutions in orthopedic surgery, neurosurgery, spine
surgery, and reconstructive and general surgery. For more
information, please visit www.integralife.com
This news release contains forward-looking statements within the
meaning of the Private Securities Litigation Reform Act of
1995. Forward-looking statements include, but are not limited
to, statements concerning the future use of the Integra™ Endoscopic
Gastroc Release System. Such forward-looking statements
involve risks and uncertainties that could cause actual results to
differ materially from predicted or expected results. Among
other things, the willingness of physicians to use this product may
affect the prospects for its use in clinical procedures. In
addition, the economic, competitive, governmental, technological
and other factors identified under the heading "Risk Factors"
included in Item IA of Integra's Annual Report on Form 10-K for the
year ended December 31, 2010 and information contained in
subsequent filings with the Securities and Exchange Commission
could affect actual results.
CONTACT: Integra LifeSciences Holdings Corporation
Gianna Sabella
Director, Corporate Communications
(609) 936-2389
gianna.sabella@integralife.com
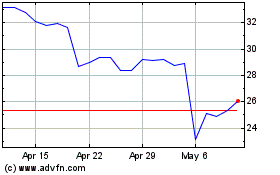
Integra LifeSciences (NASDAQ:IART)
Historical Stock Chart
From May 2024 to Jun 2024
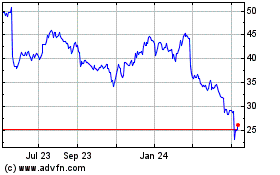
Integra LifeSciences (NASDAQ:IART)
Historical Stock Chart
From Jun 2023 to Jun 2024