MARLBOROUGH, Mass.,
Aug. 19, 2019 /PRNewswire/
-- Boston Scientific Corporation (NYSE:BSX) announced the U.S.
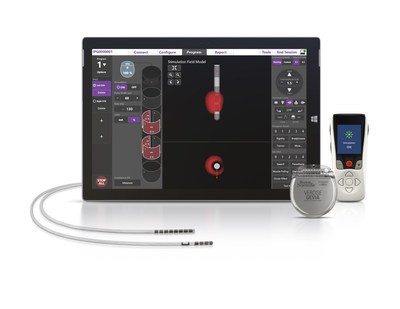
Food and Drug Administration (FDA) approval of its ImageReady™ MRI
labeling for the Vercise Gevia™ Deep Brain
Stimulation (DBS) System to be used in a full-body magnetic
resonance imaging (MRI) environment.1 This system,
with the Vercise Cartesia™ Directional Lead, is designed
to treat the symptoms of Parkinson's Disease (PD) by delivering
precisely targeted electrical stimulation in the brain to provide
optimal symptom relief and better control of unwanted side
effects.
An estimated ten million people worldwide are affected by
Parkinson's disease, causing symptoms such as shaking or tremors,
muscle stiffness, and slowness of movement.2 DBS therapy
helps patients with Parkinson's disease control their symptoms and
improve their quality of life. This FDA approval allows patients to
undergo a full-body MRI1 while benefiting from the
latest advances in DBS therapy including directional stimulation
and a longer-lasting rechargeable battery.
"When evaluating which DBS system is best for each of my
patients, I always consider the immediate and long term needs my
patient might have so that we can effectively address a patient's
therapeutic needs even as their disease progresses," said Dr.
Robert Gross, MBNA Bowman Endowed
Chair in Neurosurgery & Professor Department of Neurosurgery at
Emory University. "Customizable
therapy, battery life, the size of the device, and access to
MRI are factors patients should talk to their doctor about when
they are considering deep brain stimulation."
Clinical evidence from the INTREPID study demonstrated that
patients treated with the Vercise System sustained a 48%
improvement in motor function as measured by the Unified
Parkinson's Disease Rating Scale (UPDRS) III scores over two
years3, which is favorable to previous published reports
(25%, Follett, 2010).4
"Boston Scientific continually strives to deliver new solutions
that advance the field of neuromodulation and most importantly,
result in better outcomes for our patients around the world," said
Maulik Nanavaty, senior vice
president and president, Neuromodulation, Boston Scientific.
The Vercise Gevia with Cartesia Directional Lead was approved by
the U.S. FDA in January 2019,
following the first-generation Vercise DBS System approval in
December 2017. The ImageReady™ MRI
Vercise Gevia System has also been commercially available in the
European market since 2017.
About Boston Scientific
Boston Scientific transforms
lives through innovative medical solutions that improve the health
of patients around the world. As a global medical technology leader
for more than 35 years, we advance science for life by providing a
broad range of high performance solutions that address unmet
patient needs and reduce the cost of healthcare. For more
information, visit www.bostonscientific.com and connect on
Twitter and Facebook.
1 1.5 Tesla MRI conditional when all conditions of
use are met.
2
http://parkinson.org/Understanding-Parkinsons/Causes-and-Statistics/Statistics
3Vitek, J., et. al. A Two-Year Follow-Up of a
Prospective, Double Blinded, Multi-Center Randomized Controlled
Trial Evaluating Deep Brain Stimulation with a New Multiple Source,
Constant-Current Rechargeable System for Treatment of Parkinson's
Disease (INTREPID). INS 2019, Sydney,
Australia.
4Follett et al., N EnglJ Med. 2010 Jun 3;362(22):2077-91. Pallidal versus
subthalamic deep-brain stimulation for Parkinson's disease.
Cautionary Statement Regarding Forward-Looking
Statements
This press release contains forward-looking
statements within the meaning of Section 27A of the Securities Act
of 1933 and Section 21E of the Securities Exchange Act of 1934.
Forward-looking statements may be identified by words like
"anticipate," "expect," "project," "believe," "plan," "estimate,"
"intend" and similar words. These forward-looking statements are
based on our beliefs, assumptions and estimates using information
available to us at the time and are not intended to be guarantees
of future events or performance. These forward-looking statements
include, among other things, statements regarding our product
launches and product performance and impact. If our underlying
assumptions turn out to be incorrect, or if certain risks or
uncertainties materialize, actual results could vary materially
from the expectations and projections expressed or implied by our
forward-looking statements. These factors, in some cases, have
affected and in the future (together with other factors) could
affect our ability to implement our business strategy and may cause
actual results to differ materially from those contemplated by the
statements expressed in this press release. As a result, readers
are cautioned not to place undue reliance on any of our
forward-looking statements.
Factors that may cause such differences include, among other
things: future economic, competitive, reimbursement and regulatory
conditions; new product introductions; demographic trends; the
closing and integration of acquisitions; intellectual property;
litigation; financial market conditions; and future business
decisions made by us and our competitors. All of these factors are
difficult or impossible to predict accurately and many of them are
beyond our control. For a further list and description of these and
other important risks and uncertainties that may affect our future
operations, see Part I, Item 1A – Risk Factors in our most
recent Annual Report on Form 10-K filed with the Securities and
Exchange Commission, which we may update in Part II, Item 1A –
Risk Factors in Quarterly Reports on Form 10-Q we have filed
or will file hereafter. We disclaim any intention or obligation to
publicly update or revise any forward-looking statements to reflect
any change in our expectations or in events, conditions or
circumstances on which those expectations may be based, or that may
affect the likelihood that actual results will differ from those
contained in the forward-looking statements. This cautionary
statement is applicable to all forward-looking statements contained
in this document.
CONTACTS:
Rochelle Silsbee
Media Relations
(818) 812-7320 (office)
Rochelle.Silsbee@bsci.com
Susie Lisa, CFA
Investor Relations
(508) 683-5565 (office)
BSXInvestorRelations@bsci.com
 View original content to download
multimedia:http://www.prnewswire.com/news-releases/boston-scientific-announces-fda-approval-of-imageready-mri-for-vercise-gevia-deep-brain-stimulation-system-300903892.html
View original content to download
multimedia:http://www.prnewswire.com/news-releases/boston-scientific-announces-fda-approval-of-imageready-mri-for-vercise-gevia-deep-brain-stimulation-system-300903892.html
SOURCE Boston Scientific Corporation