Eton Pharmaceuticals Down Nearly 17% As FDA Doesn't Approve EM-100
July 12 2019 - 7:25PM
Dow Jones News
By Josh Beckerman
Eton Pharmaceuticals Inc. (ETON) shares fell nearly 17% to $6.91
after hours as the U.S Food and Drug Administration didn't approve
its EM-100 ophthalmic solution for allergic conjunctivitis.
The FDA application was made by Eton's partner, which wasn't
named in a statement Friday, but in February, Bausch Health Cos.
(BHC) said it acquired U.S. rights to EM-100.
Eton said no concerns were raised about the clinical data, and
believes the issues cited in an FDA Complete Response Letter "can
be addressed in the coming months."
Bausch Health didn't immediately reply to a request for comment
late Friday.
Write to Josh Beckerman at josh.beckerman@wsj.com
(END) Dow Jones Newswires
July 12, 2019 19:10 ET (23:10 GMT)
Copyright (c) 2019 Dow Jones & Company, Inc.
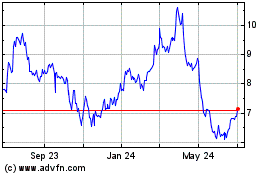
Bausch Health Companies (NYSE:BHC)
Historical Stock Chart
From Mar 2024 to Apr 2024
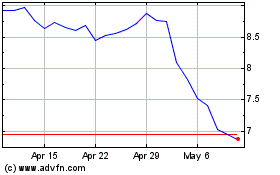
Bausch Health Companies (NYSE:BHC)
Historical Stock Chart
From Apr 2023 to Apr 2024