Vaxxinity’s COVID-19 Vaccine Candidate UB-612 Produces High Levels of Neutralizing Antibodies Against Omicron and Other Variants of Concern
February 11 2022 - 9:28AM

Vaxxinity, Inc. (Nasdaq: VAXX), a U.S. company pioneering the
development of a new class of immunotherapeutic vaccines, today
announced results from studies demonstrating the ability of UB-612,
its COVID-19 vaccine candidate, to elicit a broad immune response
against multiple variants of concern, and specifically more than
three-times higher titers of neutralizing antibodies against the
Omicron variant of SARS-CoV-2 than an approved mRNA vaccine with
boosters.
“The ability of these sera to neutralize Omicron at these high
levels is extraordinary and unprecedented considering that Omicron
has more than 30 potential immune evading mutations and deletions
across its spike protein, 15 of which are in the RBD region where
our UB-612 vaccine is directed. Such data show that UB-612 could
potentially be a unique vaccine candidate that is highly effective
at mobilizing the immune system against a broad range of both
current and future SARS-CoV-2 variants,” said Farshad Guirakhoo,
Ph.D., Chief Scientific Officer at Vaxxinity.
The studies were conducted by David Goldblatt, M.B. Ch.B.,
Ph.D., at University College London (UCL) and VisMederi, a
Coalition for Epidemic Preparedness Innovations (CEPI)-centralized
laboratory.
Sera from 92 participants from UB-612’s Phase 2 study including
8 placebos (randomized and tested blinded) and up to 36
participants in the Phase 1 study, half of the latter boosted with
a third dose of UB-612, were used in these studies to analyze
binding of IgG and neutralization against multiple Variants of
Concern and Variants of Interest, including Omicron.
Key results from the studies include:
- Three doses of UB-612 elicited neutralizing antibody titers of
GMT VNT50 of 335 against the Omicron variant of SARS-CoV-2, over
3-fold higher than reported after three doses of an approved mRNA
vaccine [1].
- The receptor-binding domain (RBD)- and spike (S)
protein-binding IgG antibodies after booster immunization with
UB-612 produced high cross-reactivity against multiple SARS-CoV-2
variants, including Alpha, Beta, Delta, Gamma and Omicron, similar
to or higher than those of approved vaccines and boosters [2].
- The RBD-binding antibodies against the Wuhan strain reported
for several authorized vaccines are predictive of UB-612 vaccine
efficacy against COVID-19 at approximately 95% after three doses
[2].
Vaxxinity plans to publish these data and present the findings
at World Vaccine Congress in April 2022. UB-612 has been evaluated
in Phase 1 and Phase 2 studies and is preparing to launch a global
pivotal booster study later this year.
Mei Mei Hu, CEO of Vaxxinity, said, “We are highly encouraged by
these data as they show that UB-612 produces levels of
neutralization comparable or exceeding current market leading
vaccines while achieving a striking breadth of coverage against
multiple variants of concern. Our hope is that we can provide a
safe alternative vaccine option that can address not only today’s
COVID but also tomorrow’s and the next day’s mutations.”
About the IgG Binding and Neutralization
Studies
UCL performed multiple RBD- and Spike protein-binding assays on
the UB-612 sera against multiple Variants of Concern/Variants of
Interest and compared these to historical data generated with sera
from subjects immunized with relevant approved comparator mRNA and
Adenovirus COVID-19 vaccines in the same assays.
VisMederi assessed neutralization of UB-612 in live VNT assays
against a Wuhan-like prototype strain (Italy INM1 strain), Omicron
and Delta. VisMederi is part of the CEPI laboratory network and has
developed validated live VNT assays referenced in regulatory
submissions and scientific publications.
About UB-612
UB-612 is the first multitope protein/peptide-based vaccine
candidate for SARS-CoV-2. The vaccine candidate is designed to
activate both B and T-cell arms of the immune system. Phase 1 and
Phase 2 trials of UB-612 have shown UB-612 to be well tolerated
with no significant safety findings to date, while observing UB-612
generated antibodies that can bind to the S1-RBD protein and
neutralize SARS-CoV-2, in addition to driving a T-lymphocyte
response.
About Vaxxinity
Vaxxinity, Inc. is a purpose-driven biotechnology company
committed to democratizing healthcare across the globe. The company
is pioneering a new class of synthetic, peptide-based
immunotherapeutic vaccines aimed at disrupting the existing
treatment paradigm for chronic disease, increasingly dominated by
monoclonal antibodies, which suffer from prohibitive costs and
cumbersome administration. The company’s proprietary technology
platform has enabled the innovation of novel pipeline candidates
designed to bring the efficiency of vaccines to the treatment of
chronic diseases, including Alzheimer’s, Parkinson’s, migraine, and
hypercholesterolemia. The technology is also implemented as part of
a COVID-19 vaccine program. Vaxxinity has optimized its pipeline to
achieve a potentially historic, global impact on human health.
For more information about Vaxxinity, Inc., visit
www.vaxxinity.com and follow us on social media @vaxxinity.
Forward-looking Statement
This press release includes forward-looking statements within
the meaning of the Private Securities Litigation Reform Act of
1995. The use of certain words, including "believe," "may,"
“continue,” “advancing,” and "will" and similar expressions, are
intended to identify forward-looking statements. These
forward-looking statements involve substantial risks and
uncertainties, including statements that are based on the current
expectations and assumptions of Vaxxinity’s management about the
development of a new class of immunotherapeutic vaccines and the
innovation and efficacy of Vaxxinity’s product candidates. Various
important factors could cause actual results or events to differ
materially from those that may be expressed or implied by our
forward-looking statements. Additional important factors to be
considered in connection with forward-looking statements are
described in the "Risk Factors" section of the Company's Quarterly
Report on Form 10-Q filed with the Securities and Exchange
Commission on December 23, 2021. The forward-looking statements are
made as of this date and Vaxxinity does not undertake any
obligation to update any forward-looking statements, whether as a
result of new information, future events or otherwise, except as
required by law.
Investor ContactClaudia
Styslingervaxxinity@argotpartners.com
Press ContactPhilip
Cowdellmedia@vaxxinity.com
[1] For further details on the method, please see Muik, A. et
al, Science. 2022. Data from UB-612 were generated at the same
laboratory as part of the CEPI centralized laboratory network.
There may be minor differences that could have influenced the
outcome of the study, including but not limited to sample size,
virus dose amount used, and the fact that samples were not tested
head-to-head.
[2] Comparison to historical data generated from comparator
vaccines. Goldblatt et al, Vaccine 2022.
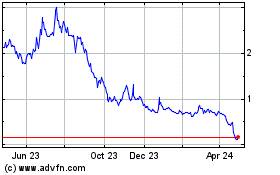
Vaxxinity (NASDAQ:VAXX)
Historical Stock Chart
From Mar 2024 to Apr 2024
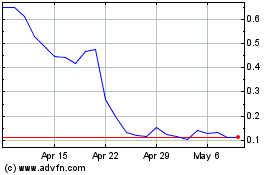
Vaxxinity (NASDAQ:VAXX)
Historical Stock Chart
From Apr 2023 to Apr 2024