MEDIA ADVISORY: Ultragenyx Canada Announces Health Canada Approval of a Second Indication for Crysvita™ (Burosumab Injection) for the Treatment of Tumour Induced Osteomalacia in Adults
September 07 2021 - 8:00AM

Ultragenyx Pharmaceutical Inc., a biopharmaceutical company focused
on the development and commercialization of novel products for rare
and ultra-rare diseases, today announced that Crysvita™ (Burosumab
Injection) has been approved by Health Canada for the treatment of
Tumour Induced Osteomalacia (TIO) in adults. Crysvita is also
approved for the treatment of X-Linked Hypophosphatemia.
TIO is a rare disease that is characterized by the development
of tumours that cause weakened and softened bones. The tumours
associated with TIO release a protein known as fibroblast growth
factor 23 (FGF23) that lowers phosphate levels.
INDICATION FOR TUMOUR INDUCED OSTEOMALACIA
(TIO)
CRYSVITA (Burosumab Injection) is indicated for the treatment of
fibroblast growth factor 23 (FGF23)–related hypophosphatemia in
tumour-induced osteomalacia (TIO) associated with tumours that
cannot be curatively resected or localized in adult patients.
Treatment should be initiated and monitored by a health
professional experienced in the management of patients with
metabolic bone diseases.
Safety and efficacy in pediatric and geriatric populations have
not been established.
For Important Safety Information, please consult the Crysvita
Product Monograph here. The Product Monograph is also available by
calling 1-833-388-5872.
About Ultragenyx Pharmaceutical Inc.Ultragenyx
is a biopharmaceutical company committed to bringing novel
therapies to patients for the treatment of serious rare and
ultra-rare genetic diseases. The company has built a diverse
portfolio of approved medicines and treatment candidates aimed at
addressing diseases with high unmet medical need and clear biology,
for which there are typically no approved therapies treating the
underlying disease.
The company is led by a management team experienced in the
development and commercialization of rare disease therapeutics.
Ultragenyx’s strategy is predicated upon time- and cost-efficient
drug development, with the goal of delivering safe and effective
therapies to patients with the utmost urgency.
For more information on Ultragenyx, please visit the company's
website at: www.ultragenyx.com.
Forward-Looking Statements Except for the
historical information contained herein, the matters set forth in
this press release, including statements related to Ultragenyx's
expectations and projections regarding its future operating results
and financial performance, anticipated cost or expense reductions,
the timing, progress and plans for its clinical programs and
clinical studies, future regulatory interactions, and the
components and timing of regulatory submissions are forward-looking
statements within the meaning of the "safe harbor" provisions of
the Private Securities Litigation Reform Act of 1995. Such
forward-looking statements involve substantial risks and
uncertainties that could cause our clinical development programs,
collaboration with third parties, future results, performance or
achievements to differ significantly from those expressed or
implied by the forward-looking statements. Such risks and
uncertainties include, among others, the effects from the COVID-19
pandemic on the company’s commercialization activities, business
and operating results, risks related to reliance on third party
partners to conduct certain activities on the company’s behalf,
smaller than anticipated market opportunities for the company’s
products and product candidates, manufacturing risks, competition
from other therapies or products, and other matters that could
affect sufficiency of existing cash, cash equivalents and
short-term investments to fund operations, the company’s future
operating results and financial performance, and the availability
or commercial potential of Ultragenyx’s products and drug
candidates. Ultragenyx undertakes no obligation to update or revise
any forward-looking statements. For a further description of the
risks and uncertainties that could cause actual results to differ
from those expressed in these forward-looking statements, as well
as risks relating to the business of Ultragenyx in general, see
Ultragenyx's Quarterly Report on Form 10-Q filed with the
Securities and Exchange Commission on August 3, 2021, and its
subsequent periodic reports filed with the Securities and Exchange
Commission.
ContactsUltragenyx Pharmaceutical
Inc.MediaCarolyn Wanginfo@ultragenyx.com
Ultragenyx CanadaMonty Keast647-888-8615info@ultragenyx.com
A quote from Dr. Aliya Khan, Professor of Clinical Medicine,
Divisions of Endocrinology and Metabolism and Geriatrics; Director
of Fellowship in Metabolic Bone Disease and Director of the Calcium
Disorders Clinic at McMaster University is available
at: https://www.globenewswire.com/NewsRoom/AttachmentNg/417b422c-d325-4c9d-94bb-26488778a692
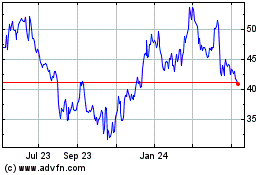
Ultragenyx Pharmaceutical (NASDAQ:RARE)
Historical Stock Chart
From Mar 2024 to Apr 2024
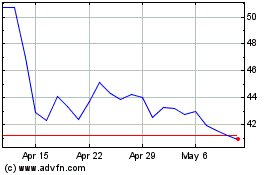
Ultragenyx Pharmaceutical (NASDAQ:RARE)
Historical Stock Chart
From Apr 2023 to Apr 2024