Tilray® Imports GMP-Certified CBD into the U.S. for Two Clinical Trials Led by NYU School of Medicine
August 08 2019 - 8:00AM
Business Wire
NYU School of Medicine will lead trials testing CBD treatment
for patients with Alcohol Use Disorder and Post-Traumatic Stress
Disorder with Alcohol Use Disorder
Tilray, Inc. (NASDAQ:TLRY), a global pioneer in cannabis
research, cultivation, production, and distribution, today
announced it has received approval from the U.S. government and
successfully imported an initial shipment of medical cannabinoids
into the U.S. to support two clinical trials led by NYU School of
Medicine.
The two studies will test the efficacy of cannabidiol, or CBD,
to treat patients suffering from Alcohol Use Disorder (AUD) and
patients suffering from AUD comorbid with Post-Traumatic Stress
Disorder (PTSD). Tilray has provided a Good Manufacturing Practices
(GMP)-certified CBD formulation in capsule form for the
studies.
Michael Bogenschutz, M.D., Professor in the Department of
Psychiatry at NYU School of Medicine, will lead one trial focused
on testing CBD in patients with AUD, and Charles R. Marmar, M.D.,
the Lucius Littauer Professor and Chair of the Department of
Psychiatry at NYU School of Medicine, will lead another trial
testing CBD in patients with AUD comorbid with PTSD. Both trials
are expected to begin recruitment in the summer of 2019 and will
take place at NYU School of Medicine.
The first NYU led clinical trial is a double-blind, randomized
study designed to assess CBD treatment in patients with AUD. NYU
will evaluate the safety and tolerability of a daily CBD treatment
regimen in an AUD population and assess the impact of CBD on
neuropsychological and neurophysiological domains implicated in
AUD.
The second clinical trial is a double-blind, randomized study
designed to assess the safety, tolerability, efficacy, and effect
size of CBD in reducing alcohol use and PTSD symptoms for those
suffering from AUD comorbid with PTSD.
Quotes:
- “We’re proud to support these two clinical trials led by NYU
School of Medicine,” says Philippe Lucas, Tilray’s VP of Global
Patient Research and Access. “Tilray is committed to supporting
research that can provide further evidence for the safety and
efficacy of medical cannabis and, ultimately, data to support the
treatment of more patients in need around the world.”
- “NYU School of Medicine enjoys an outstanding reputation in
advancing the study of CBD for its use in treating several
illnesses, most notably various forms of epilepsy,” says Dr. Marmar
of NYU School of Medicine. “We are now coupling that experience
with our past and ongoing efforts to better understand and treat
PTSD and AUD. We are confident that our studies will lead to
benefits. Our returning veterans, in particular, who suffer from
PTSD resulting from their military service, deserve our best
efforts.”
- “Preclinical work and limited human experience suggest that CBD
may have anti-addictive effects that may be useful in the treatment
of alcohol use disorder and other substance use disorders,” says
Dr. Bogenschutz of NYU School of Medicine. “Our study is the first
to investigate the effects of CBD in the treatment of alcohol use
disorder, and will provide important new information on its
mechanisms of action and clinical benefits.”
About Tilray® Tilray is a global pioneer in the research,
cultivation, production and distribution of cannabis and
cannabinoids currently serving tens of thousands of patients and
consumers in 13 countries spanning five continents.
Cautionary note regarding forward-looking statements:
This press release contains “forward-looking statements” within
the meaning of the U.S. Private Securities Litigation Reform Act of
1995 and “forward-looking information” within the meaning of
Canadian securities laws, or collectively, forward-looking
statements. Forward-looking statements or
forward-looking-information in this press release may be identified
by the use of words such as, “may”, “would”, “could”, “will”,
“likely”, “expect”, “anticipate”, “believe, “intend”, “plan”,
“forecast”, “project”, “estimate”, “outlook” and other similar
expressions. Forward-looking statements are not a guarantee of
future performance, future action or any particular result to
clinical trials and are based upon a number of estimates and
assumptions of management in light of management’s experience and
perception of trends, current conditions and expected developments,
as well as other factors that management believes to be relevant
and reasonable in the circumstances, including assumptions in
respect of current and future market conditions, the current and
future regulatory environment and future approvals and permits.
Actual results, performance or achievement could differ materially
from that expressed in, or implied by, any forward-looking
statements in this press release, and, accordingly, you should not
place undue reliance on any such forward-looking statements and
they are not guarantees of future results. Please see the heading
“Risk Factors” in Tilray’s Quarterly Report on Form 10-Q, which was
filed with the Securities and Exchange Commission and Canadian
securities regulators on May 15, 2019, assumptions, uncertainties
and other factors that may cause actual future results or
anticipated events to differ materially from those expressed or
implied in any forward-looking statements. Tilray does not
undertake and specifically declines any obligation to update any
forward-looking statements that are included herein, except in
accordance with applicable securities laws.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20190808005081/en/
Media: Chrissy Roebuck, +1-833-206-8161,
news@tilray.com Investors: Katie Turner, +1-646-277-1228,
Katie.turner@icrinc.com
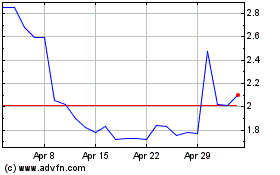
Tilray Brands (NASDAQ:TLRY)
Historical Stock Chart
From Mar 2024 to Apr 2024
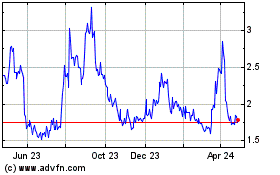
Tilray Brands (NASDAQ:TLRY)
Historical Stock Chart
From Apr 2023 to Apr 2024