NeuroMetrix to Introduce Quell® Fibromyalgia at the American College of Rheumatology 2022 Annual Meeting
November 08 2022 - 9:00AM

NeuroMetrix, Inc. (Nasdaq: NURO) will introduce Quell Fibromyalgia
at the American College of Rheumatology Annual Meeting, ACR
Convergence 2022, on November 12 – 14 in Philadelphia,
Pennsylvania. Quell Fibromyalgia is a wearable neuromodulation
technology that is the first and only FDA authorized medical device
to help reduce the symptoms of fibromyalgia. Conference attendees
are encouraged to visit the Company’s booth #1745 for a
demonstration and to review prescribing details for fibromyalgia
patients.
Fibromyalgia is a chronic condition characterized by generalized
pain, fatigue, poor sleep, memory and concentration impairments,
mood disorders and other disabling symptoms. The prevalence of
fibromyalgia is estimated to be 2 to 6 percent of the U.S. adult
population (5 to 15 million people).
Quell Fibromyalgia emerged from the NeuroMetrix pipeline of
prescription wearable neurotherapeutics, two of which received FDA
Breakthrough Device Designation: fibromyalgia and chemotherapy
induced peripheral neuropathy (CIPN).
"We are pleased to contribute to the unmet need for safe and
effective treatments for people with fibromyalgia," said Shai N.
Gozani, M.D., Ph.D., Chief Executive Officer, NeuroMetrix. "This
conference is an opportunity for us to exchange information with
rheumatologists who are managing patients with fibromyalgia."
Quell Fibromyalgia Indications
Quell Fibromyalgia is a transcutaneous electrical nerve
stimulation (TENS) device indicated as an aid for reducing the
symptoms of fibromyalgia in adults with high pain sensitivity. The
device may be used during sleep.
Limitations
The sale, distribution, and use of Quell Fibromyalgia is
restricted to prescription use in accordance with 21 CFR 801.109.
The product labeling should be reviewed for a complete list of
contraindications, precautions and warnings.
About Quell Technology
Quell is an advanced, non-invasive, neuromodulation technology
that is covered by 23 U.S. utility patents. It is the only wearable
neuromodulator that is enabled by a proprietary microchip to
provide precise, high-power nerve stimulation in a form factor the
size of a credit card. Quell utilizes position and motion sensing
to automatically adjust stimulation for an optimal user experience
both day and night. The device supports Bluetooth® low energy (BLE)
to communicate with mobile apps for multiple smartphone and
smartwatch platforms.
About NeuroMetrix
NeuroMetrix is an innovation-driven company with a mission to
improve individual and population health through novel medical
devices and technology solutions for neurological disorders and
pain syndromes. The Company has three commercial products.
DPNCheck® is a diagnostic device that provides rapid, point-of-care
detection of peripheral neuropathies. ADVANCE® is a diagnostic
device that provides automated, in-office nerve conduction studies
for the evaluation of focal neuropathies. Quell® Fibromyalgia is a
wearable neuromodulator that is the first and only FDA-authorized
medical device to help reduce the symptoms of fibromyalgia. For
more information, visit www.neurometrix.com.
Source: NeuroMetrix, Inc.
Thomas T. Higgins SVP and Chief Financial Officer 781-314-2761
neurometrix.ir@neurometrix.com
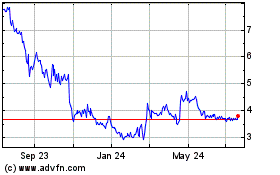
NeuroMetrix (NASDAQ:NURO)
Historical Stock Chart
From Mar 2024 to Apr 2024
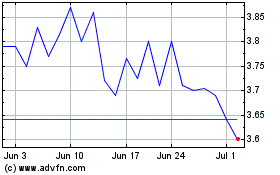
NeuroMetrix (NASDAQ:NURO)
Historical Stock Chart
From Apr 2023 to Apr 2024