NeuroMetrix Names Jonathan Breck Harmel as National Director of Sales for its Emerging Prescription Neurotherapeutics Business
September 01 2022 - 9:00AM

NeuroMetrix, Inc. (Nasdaq: NURO) today announced that it has
appointed Jonathan "Breck" Harmel as National Director of Sales,
Neuromodulation. Mr. Harmel will be responsible for leading the
sales organization for the Company's prescription wearable
neurotherapeutics business in the U.S. market. The Company recently
received FDA De Novo authorization to market its Quell® device as
an aid for reducing the symptoms of fibromyalgia in adults with
high pain sensitivity. The Company received FDA Breakthrough
Designation for the use of Quell to treat fibromyalgia in July
2021.
Mr. Harmel brings 15 years of experience in high
growth medical technology and device sales, including building and
managing successful sales teams on a national scale. Mr. Harmel was
most recently the U.S. Director of Market Development at Neurolief,
which developed and is commercializing a wearable neuromodulation
device for migraine. Prior to Neurolief, Mr. Harmel was Regional
Director of Business Development for Neuronetics, which markets a
transcranial magnetic stimulation (TMS) device for the treatment of
major depression. Before that, he held sales leadership roles at
Virgin Pulse, CDx Diagnostics and Avantis Medical Systems.
"Breck's background in medical technology sales,
and in particular his leadership experience in the neuromodulation
sector, is highly relevant to our effort to launch and build a
prescription neurotherapeutics business powered by Quell
technology," said Shai N. Gozani M.D., Ph.D., NeuroMetrix Chief
Executive Officer. "His immediate priority will be to direct the
launch of Quell Fibromyalgia in the fourth quarter of this year. We
believe physicians treating patients with fibromyalgia will be
interested in Quell's clinical benefits and safety profile. Our
initial commercialization efforts will focus on rheumatologists and
pain medicine physicians. We are pleased to have Breck on board and
leading this effort."
About Quell Technology
Quell is an advanced, non-invasive, neuromodulation
technology that is covered by 23 U.S. utility patents. It is the
only wearable neuromodulator that is enabled by a proprietary
microchip that provides precise, high-power nerve stimulation in a
form factor the size of a credit card. Quell utilizes position and
motion sensing to automatically adjust stimulation for an optimal
user experience both day and night. The device supports Bluetooth®
low energy (BLE) to communicate with mobile apps for multiple
smartphone and smartwatch platforms. Quell is indicated for
treatment of fibromyalgia symptoms and chronic lower extremity
pain.
About NeuroMetrix
NeuroMetrix is an innovation-driven company with a
mission to improve individual and population health through
innovative medical devices and technology solutions for
neurological disorders and pain syndromes. The Company has three
commercial products. DPNCheck® is a diagnostic device that provides
rapid, point-of-care detection of peripheral neuropathies. ADVANCE®
is a diagnostic device that provides automated, in-office nerve
conduction studies for the evaluation of focal neuropathies. Quell®
is a wearable neuromodulation technology. For more information,
visit www.NeuroMetrix.com.
Source: NeuroMetrix, Inc.Thomas T. HigginsSVP and
Chief Financial
Officer781-314-2761neurometrix.ir@neurometrix.com
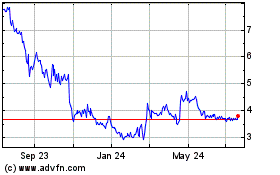
NeuroMetrix (NASDAQ:NURO)
Historical Stock Chart
From Mar 2024 to Apr 2024
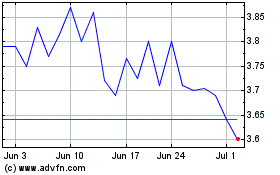
NeuroMetrix (NASDAQ:NURO)
Historical Stock Chart
From Apr 2023 to Apr 2024