Mirum Pharmaceuticals to Report Third Quarter 2021 Financial Results and Host Conference Call on November 15, 2021
November 08 2021 - 4:05PM
Business Wire
Mirum Pharmaceuticals, Inc. (Nasdaq: MIRM) today announced that
the company will report financial results for the quarter ended
September 30, 2021 on Monday, November 15, 2021. That same day,
Mirum will host a conference call and audio webcast at 1:30 p.m.
PT/4:30 p.m. ET to provide a business update.
Conference Call Details:
U.S. toll-free: 844.200.6205 International: 646.904.5544
Passcode: 588077
You may also access the call via webcast by visiting the
Investors & Media section on Mirum’s corporate website. A
replay of this webcast will be available for 30 days.
About Mirum Pharmaceuticals, Inc.
Mirum Pharmaceuticals, Inc. is a biopharmaceutical company
dedicated to transforming the treatment of rare liver diseases.
Mirum’s approved medication is LIVMARLI™ (maralixibat) oral
solution which is approved in the U.S. for the treatment of
cholestatic pruritus in patients with Alagille syndrome one year of
age and older.
Mirum’s late-stage pipeline includes two investigational
treatments for debilitating liver diseases affecting children and
adults. Maralixibat (LIVMARLI), an oral ileal bile acid transporter
(IBAT) inhibitor, is currently being evaluated in clinical trials
for pediatric liver diseases and includes the MARCH Phase 3 study
for progressive familial intrahepatic cholestasis (PFIC) and the
EMBARK Phase 2b study for patients with biliary atresia. In
addition, Mirum has an expanded access program open in Canada,
Australia, the UK and several countries in Europe for eligible
patients with Alagille syndrome.
Mirum has submitted a Marketing Authorization Application to the
European Medicines Agency for maralixibat for the treatment of
cholestatic liver disease in patients with Alagille syndrome.
Mirum’s second investigational treatment, volixibat, also an
oral IBAT inhibitor, is being evaluated in two registrational
studies including the OHANA Phase 2b study for pregnant women with
intrahepatic cholestasis of pregnancy and the VISTAS Phase 2b study
for adults with primary sclerosing cholangitis. Mirum is planning
to launch a Phase 2b study in primary biliary cholangitis later
this year.
To augment its pipeline in cholestatic liver disease, Mirum has
acquired the exclusive option to develop and commercialize gene
therapy programs VTX-803 and VTX-802 for PFIC3 and PFIC2,
respectively, from Vivet Therapeutics SAS, following preclinical
evaluation and investigational new drug-enabling studies.
Follow Mirum on Twitter, Facebook, LinkedIn and Instagram.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20211108006017/en/
Investors:
Ian Clements, Ph.D. ir@mirumpharma.com
Sam Martin Argot Partners ir@mirumpharma.com 212.600.1902
Media:
Erin Murphy media@mirumpharma.com
Mirum Pharmaceuticals (NASDAQ:MIRM)
Historical Stock Chart
From Mar 2024 to Apr 2024
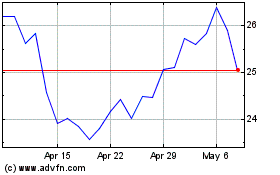
Mirum Pharmaceuticals (NASDAQ:MIRM)
Historical Stock Chart
From Apr 2023 to Apr 2024