ALX Oncology's Evorpacept Gets FDA Designation for Leukemia
June 29 2022 - 8:00AM
Dow Jones News
By Dean Seal
ALX Oncology Holdings Inc. said the Food and Drug Administration
granted orphan-drug designation to its drug evorpacept as a
treatment for acute myeloid leukemia.
Sophia Randolph, the clinical-stage immuno-oncology company's
chief medical officer, said Wednesday that the designation reflects
the regulator's recognition that evorpacept has shown potential for
improving the clinical outcomes of patients with advanced
cancers.
Earlier this year, the company said that the FDA had given
orphan-drug designation to evorpacept for the treatment of patients
with gastric cancer and gastroesophageal junction cancer.
Orphan-drug designation is a special status given to drugs that
show promise for potentially treating rare, or orphan, diseases
that have fewer than 200,000 cases a year in the U.S.
Shares of ALX Oncology rose 24% to $10 in premarket trading.
Write to Dean Seal at dean.seal@wsj.com
(END) Dow Jones Newswires
June 29, 2022 07:45 ET (11:45 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
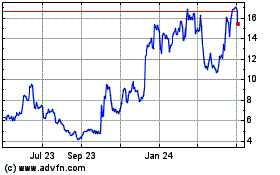
ALX Oncology (NASDAQ:ALXO)
Historical Stock Chart
From Mar 2024 to Apr 2024
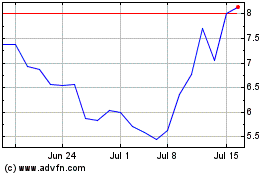
ALX Oncology (NASDAQ:ALXO)
Historical Stock Chart
From Apr 2023 to Apr 2024