Lilly Antibody Bebtelovimab Gets FDA EUA for Certain Mild-to-Moderate Covid-19 Patients
February 11 2022 - 4:59PM
Dow Jones News
By Josh Beckerman
Eli Lilly & Co.'s bebtelovimab, a monoclonal antibody
discovered by AbCellera Biologics Inc., received Emergency Use
Authorization from the U.S. Food and Drug Administration for
certain Covid-19 patients.
The EUA is for mild-to-moderate Covid-19 in adults and children
12-and-up weighing at least 88 pounds with a positive COVID-19
test, and who are at high risk for progression to severe Covid-19,
and for whom alternative treatment options approved or authorized
by the FDA aren't accessible or clinically appropriate, the FDA
said.
The agency "is carefully monitoring circulating viral variants
and their sensitivity to authorized monoclonal antibodies,
including bebtelovimab."
Lilly said the authorized dose of bebtelovimab is 175 mg given
as an intravenous injection over at least 30 seconds.
Last month, the FDA restricted Lilly's bamlanivimab and
etesevimab combination, as well as Regeneron Pharmaceuticals Inc.'s
REGEN-COV, citing lack of effectiveness against the omicron
variant. AbCellera discovered bamlanivimab.
Lilly said Thursday that it agreed to supply 600,000 doses of
bebtelovimab for at least $720 million in an agreement with the
U.S. government, subject to receiving the EUA.
"Early in 2021, prior to the identification of the Omicron
variant, Lilly scientists were already working to develop
bebtelovimab as a broadly neutralizing antibody that could be used
to fight a highly mutated variant, should one emerge," the company
said Friday.
Write to Josh Beckerman at josh.beckerman@wsj.com
(END) Dow Jones Newswires
February 11, 2022 16:44 ET (21:44 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
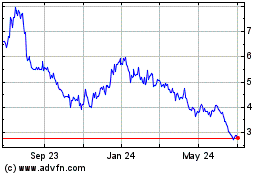
AbCellera Biologics (NASDAQ:ABCL)
Historical Stock Chart
From Mar 2024 to Apr 2024
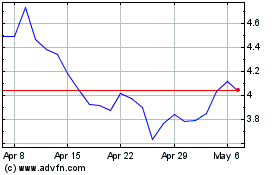
AbCellera Biologics (NASDAQ:ABCL)
Historical Stock Chart
From Apr 2023 to Apr 2024