Former FDA Official Engaged By cbdMD
February 04 2021 - 9:00AM
Business Wire
Dr. Sibyl Swift to Advise on Company’s
Regulatory Affairs
cbdMD, Inc. (NYSE American: YCBD, YCBDpA) (the “Company”), one
of the leading, and most highly trusted and recognized cannabidiol
(CBD) brands, announced today that it has retained former US Food
and Drug Administration (FDA) official Dr. Sibyl Swift as a
regulatory consultant to oversee the company’s regulatory
initiatives and prepare its products for further
certifications.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20210204005695/en/
Dr. Sibyl Swift (Photo: Business
Wire)
From 2014 to 2020, Dr. Swift served in various capacities at the
FDA, most notably as the Associate Director for Research and
Strategy in the FDA’s Office of Dietary Supplement Programs (ODSP),
where she worked closely on the agency’s science, research,
compliance, enforcement, and policy initiatives. In 2020, she
served as the Senior Vice President for Scientific & Regulatory
Affairs for the Natural Products Association (NPA), where she
addressed regulatory policies related to NDI notifications, FDA
guidance, and claims substantiation.
“We are grateful to Dr. Daniel Fabricant and the NPA for
allowing Dr. Swift to join our team. Dr. Swift’s experience and
science-based approach demonstrates to our customers, the FDA and
global regulatory authorities that cbdMD is committed to having our
products meet the highest regulatory standards for quality and
safety,” said Chairman and co-CEO, Marty Sumichrast.
About cbdMD, Inc.
cbdMD, Inc. is one of the leading, most highly trusted, and most
recognized cannabidiol (CBD) brands, whose current products include
CBD tinctures, CBD capsules, CBD gummies, CBD topicals, CBD
botanical skincare products, CBD bath bombs, and CBD pet products.
cbdMD is a member of the NPA, a proud partner of Bellator MMA and
Life Time, Inc., and has one of the largest rosters of professional
sports athletes who are part of “Team cbdMD.” To learn more about
cbdMD and our comprehensive line of over 100 SKUs of U.S. produced,
THC-free1 CBD products, please visit www.cbdMD.com, follow cbdMD on
Instagram and Facebook, or visit one of the 6,000 retail outlets
that carry cbdMD products.
Forward-Looking Statements
This press release contains certain forward-looking statements
that are based upon current expectations and involve certain risks
and uncertainties within the meaning of the U.S. Private Securities
Litigation Reform Act of 1995. Such forward-looking statements can
be identified by the use of words such as ''should,'' ''may,''
''intends,'' ''anticipates,'' ''believes,'' ''estimates,''
''projects,'' ''forecasts,'' ''expects,'' ''plans,'' and
''proposes.'' These forward-looking statements are not guarantees
of future performance and are subject to risks, uncertainties, and
other factors, some of which are beyond our control and difficult
to predict and could cause actual results to differ materially from
those expressed or forecasted in the forward-looking statements..
You are urged to carefully review and consider any cautionary
statements and other disclosures, including the statements made
under the heading "Risk Factors" in cbdMD, Inc.'s Annual Report on
Form 10-K for the fiscal year ended September 30, 2020 as filed
with the Securities and Exchange Commission (the "SEC") on December
22, 2020 and our other filings with the SEC. cbdMD, Inc. does not
undertake any duty to update any forward-looking statements except
as may be required by law. The information which appears on our
websites and our social media platforms, including, but not limited
to, Instagram and Facebook, is not part of this press release.
1 THC-free is defined as below the level of detection using
validated scientific analytical tools.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210204005695/en/
PR:
cbdMD, Inc. Lauren Greene Communications Specialist
Lauren.Greene@cbdMD.com (843) 743-9999
Investors:
cbdMD, Inc. John Weston Director of Investor Relations
John.Weston@cbdMD.com (704) 249-9515
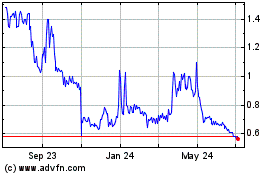
cbdMD (AMEX:YCBD)
Historical Stock Chart
From Mar 2024 to Apr 2024
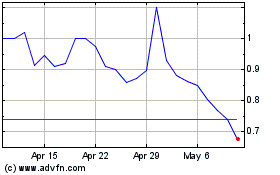
cbdMD (AMEX:YCBD)
Historical Stock Chart
From Apr 2023 to Apr 2024