Rigel Pharmaceuticals Shares Leap Premarket on FDA OK of Rezlidhia
December 02 2022 - 7:14AM
Dow Jones News
By Colin Kellaher
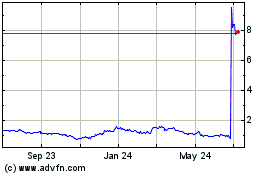
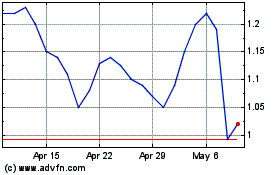
Shares of Rigel Pharmaceuticals Inc. jumped more than 40% in
premarket trading Friday after the biotechnology company said the
U.S. Food and Drug Administration approved Rezlidhia for certain
leukemia patients.
The South San Francisco, Calif., company said the approval
covers Rezlidhia for adults with relapsed or refractory acute
myeloid leukemia with a susceptible isocitrate dehydrogenase-1
mutation as detected by an FDA-approved test.
Rigel in August said it had inked an exclusive license agreement
with Forma Therapeutics Holdings Inc. to develop, manufacture and
commercialize the drug, which was under FDA review at the time with
a target action date of Feb. 15, 2023.
Danish pharmaceutical company Novo Nordisk AS acquired Forma in
October in a $1.1 billion deal.
Rigel said it will be responsible for the launch and
commercialization of Rezlidhia in the U.S., adding that it plans to
work with potential partners to further develop and commercialize
the drug outside the U.S.
Rigel shares, which closed Thursday at 68.4 cents, were recently
up nearly 43% to 97.7 cents in premarket trading.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
December 02, 2022 06:59 ET (11:59 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
Rigel Pharmaceuticals (NASDAQ:RIGL)
Historical Stock Chart
From Mar 2024 to Apr 2024
Rigel Pharmaceuticals (NASDAQ:RIGL)
Historical Stock Chart
From Apr 2023 to Apr 2024