MyMD’s lead drug candidate, MYMD-1, is being
developed to target aging and related diseases along with
autoimmune diseases including rheumatoid arthritis
MyMD Pharmaceuticals, Inc. (Nasdaq: MYMD) (“MyMD” or “the
Company”), a clinical stage pharmaceutical company committed to
extending healthy lifespan, announced today that Chris Chapman,
M.D., President, Director and Chief Medical Officer of MyMD, will
be honored by the Arthritis Foundation as its Medical Honoree of
2021, to be presented at the annual Commitment to a Cure Gala being
held October 28, 2021, in Washington, D.C.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20211026005789/en/
Chris Chapman, M.D., President, Director
and Chief Medical Officer of MyMD Pharmaceuticals, Inc. (Photo:
Business Wire)
“I am deeply honored by this recognition from the Arthritis
Foundation and am proud to be associated with this highly regarded
organization, consistently ranked as one of the very top charities
in the U.S.,” said Dr. Chapman. “Arthritis is America’s leading
cause of disability, and the Foundation’s dedication to the
prevention, control and cure of this disease has a tremendous
impact for millions of Americans every year. MyMD joins the
Arthritis Foundation in its mission to conquer arthritis with
life-changing science, namely through our clinical stage
immunometabolic regulator MYMD-1 which targets the root causes of
inflammation and has demonstrated efficacy in treating autoimmune
diseases in animal models.”
Greta Barnes, executive director for the Arthritis Foundation’s
Metro D.C. market, commented, “This honor is so fitting for Dr.
Chapman, a highly accomplished medical leader at the helm of drug
discovery for autoimmune diseases including arthritis. MyMD’s drug
candidate MYMD-1 could represent a transformation in therapeutic
solutions for individuals suffering from the pain, inflammation and
joint destruction of this devastating disease. The Arthritis
Foundation gratefully salutes Dr. Chapman and MyMD Pharmaceuticals
for their commitment to our common cause.”
All proceeds from the Arthritis Foundation’s Commitment to a
Cure Gala support the Arthritis Foundation’s efforts to combat the
disease and help more than 54 million Americans.
MyMD’s lead drug candidate, MYMD-1, is being developed to target
aging, aging-related diseases, and autoimmune diseases including
rheumatoid arthritis. MYMD-1 addresses the root causes of
autoimmune diseases by targeting pro-inflammatory cytokines,
primarily tumor necrosis factor-alpha (TNF-α), that are active in
autoimmune diseases, aging and some types of depression. The rising
prevalence of rheumatoid arthritis, psoriatic arthritis, and
inflammatory bowel disease are driving demand for TNF inhibitors
like MYMD-1. Studies estimate that the number of people suffering
from rheumatoid arthritis may rise to over 78 million by 2040.1
MyMD plans to advance MYMD-1 in the clinic with two simultaneous
Phase 2 trials beginning in the current quarter, one for delaying
aging and prolonging healthy lifespan, and the second for
COVID-19-associated depression and cytokine elevation.
Dr. Chapman Bio
Dr. Chapman is a seasoned executive and global medical expert
with extensive experience in designing and clinical trial protocols
and conducting clinical trials. Prior to joining MyMD and since
1999, Dr. Chapman has operated Chapman Pharmaceutical Consulting,
Inc., an organization that provides support to pharmaceutical and
biotech companies in North America, Europe, Japan, India and Africa
on issues such as product safety, pharmacovigilance, medical
devices, clinical trials and regulatory issues. He is also the
founder of Chapman Pharmaceutical Health Foundation, a nonprofit
organization established to solicit public funds and support
healthcare needs such as AIDS, diabetes, hypertension, lupus,
sickle cell anemia, malaria and tuberculosis. Dr. Chapman’s career
includes executive leadership positions with Organon
Pharmaceuticals and Quintiles (now known as IQVIA), board service
for Rock Creek Pharmaceuticals, Inc. He also spent two years as a
critical care physician on staff at Doctor's Community Hospital in
Lanham, Maryland.
Dr. Chapman received his M.D. degree from Georgetown University
in Washington, D.C. in 1987. He completed his internship in
internal medicine, a residency in anesthesiology and a fellowship
in cardiovascular and obstetric anesthesiology at Georgetown. Prior
to his time at Georgetown, Dr. Chapman served in the United States
Air Force as a B-52 crew chief in the Vietnam era. Dr. Chapman has
served at the Ellsworth Air Force Base in South Dakota, Andersen
Air Force Base in Guam; U-Tapao Royal Thai Navy Base in Thailand;
and Kadena Air Base in Japan.
Dr. Chapman is a graduate of the Harvard Kennedy School,
Cambridge, Massachusetts, in financial management (2020).
About the Arthritis Foundation
The Arthritis Foundation is fighting for all people who live
with arthritis. As a Champion of Yes, it's the Arthritis
Foundation's mission to turn the obstacles arthritis causes into
opportunities. The Arthritis Foundation champions life-changing
solutions and medical advancements, and it also provides ways for
people to connect, break down barriers in health care and join the
fight for a cure — uniting hearts, minds and resources to change
the future of arthritis. To join the fight to cure arthritis, visit
arthritis.org.
About MYMD-1
MYMD-1’s primary purpose is to slow the aging process and extend
healthy lifespan, and it is also showing promise as a potential
treatment for post- COVID-19 complications and as an anti-fibrotic
and anti-prolific therapeutic. MYMD-1 has shown effectiveness in
regulating the immune system by performing as a selective inhibitor
of tumor necrosis factor-alpha (TNF-α), a driver of chronic
inflammation. Unlike other therapies, MYMD-1 has been shown to
selectively block TNF-α when it becomes overactivated in autoimmune
diseases and cytokine storms, but not block it from doing its
normal job of being a first responder to any routine type of
moderate infection. MYMD-1’s ease of oral dosing is another
differentiator compared to currently available TNF-α blockers, all
of which require delivery by injection or infusion. No approved TNF
inhibitor has ever been dosed orally. In addition, the drug is not
immunosuppressive and has not been shown to cause the serious side
effects common with traditional therapies that treat inflammation.
MyMD expects to launch two Phase 2 trials during the current
quarter for MYMD-1 – one for delaying aging and prolonging healthy
lifespan, and the other for post- COVID-19 depression and cytokine
elevation.
About MyMD Pharmaceuticals, Inc.
MyMD Pharmaceuticals, Inc. (Nasdaq: MYMD), a clinical stage
pharmaceutical company committed to extending healthy lifespan, is
focused on developing two novel therapeutic platforms that treat
the causes of disease rather than only addressing the symptoms.
MYMD-1 is a drug platform based on a clinical stage small molecule
that regulates the immune system to control TNF-α, which drives
chronic inflammation, and other pro-inflammatory cell signaling
cytokines. MYMD-1 is being developed to delay aging, increase
longevity, and treat autoimmune diseases and COVID-19- associated
depression. The Company’s second drug platform, Supera-CBD, is
being developed to treat chronic pain, addiction and epilepsy.
Supera-CBD is a novel synthetic derivative of cannabidiol (CBD) and
is being developed to address and improve upon the rapidly growing
CBD market, which includes both FDA approved drugs and CBD products
not currently regulated as drugs. For more information, visit
www.mymd.com.
Cautionary Statement Regarding Forward-Looking
Statements
This press release may contain forward-looking statements. These
forward-looking statements involve known and unknown risks,
uncertainties and other factors which may cause actual results,
performance or achievements to be materially different from any
expected future results, performance, or achievements.
Forward-looking statements speak only as of the date they are made
and none of MyMD nor its affiliates assume any duty to update
forward-looking statements. Words such as "anticipate," "believe,"
"could," "estimate," "expect," "may," "plan," "will," "would'' and
other similar expressions are intended to identify these
forward-looking statements. Important factors that could cause
actual results to differ materially from those indicated by such
forward-looking statements include, without limitation: the timing
of, and MyMD’s ability to, obtain and maintain regulatory approvals
for clinical trials of MyMD’s pharmaceutical candidates; the timing
and results of MyMD’s planned clinical trials for its
pharmaceutical candidates; the amount of funds MyMD requires for
its pharmaceutical candidates; increased levels of competition;
changes in political, economic or regulatory conditions generally
and in the markets in which MyMD operates; MyMD’s ability to retain
and attract senior management and other key employees; MyMD’s
ability to quickly and effectively respond to new technological
developments; MyMD’s ability to protect its trade secrets or other
proprietary rights, operate without infringing upon the proprietary
rights of others and prevent others from infringing on MyMD’s
proprietary rights; and the impact of the ongoing COVID-19 pandemic
on MyMD’s results of operations, business plan and the global
economy. A discussion of these and other factors with respect to
MyMD is set forth in the Quarterly Report on Form 10-Q for the
quarterly period ended June 30, 2021, filed by MyMD on August 16,
2021. Forward-looking statements speak only as of the date they are
made and MyMD disclaims any intention or obligation to revise any
forward-looking statements, whether as a result of new information,
future events or otherwise.
1 Data published by Research and Markets, March 2018.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20211026005789/en/
MyMD: Robert Schatz (646) 421-9523 rschatz@mymd.com
www.mymd.com
Arthritis Foundation: Greta Barnes (202) 765-0285
mbarnes@arthritis.org
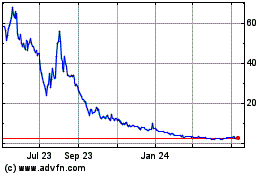
MyMD Pharmaceuticals (NASDAQ:MYMD)
Historical Stock Chart
From Mar 2024 to Apr 2024
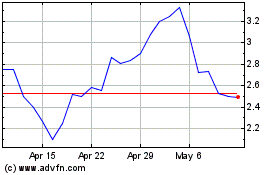
MyMD Pharmaceuticals (NASDAQ:MYMD)
Historical Stock Chart
From Apr 2023 to Apr 2024