Avidity Biosciences Lead Program Gets FDA Fast-Track Designation
October 18 2021 - 9:54AM
Dow Jones News
By Colin Kellaher
Avidity Biosciences Inc. on Monday said the U.S. Food and Drug
Administration granted fast-track designation to AOC 1001, its lead
program, for the genetic multisystem disorder myotonic dystrophy
type 1.
The biopharmaceutical company said there are currently no
approved treatments for the progressive and often fatal disease,
which primarily affects skeletal and cardiac muscle.
The FDA's fast-track program is designed to facilitate the
development and expedite the review of treatments for serious or
potentially life-threatening illnesses with high unmet medical
needs.
The FDA previously cleared Avidity to proceed with a Phase 1/2
study of AOC 1001 in adults with myotonic dystrophy type 1.
Shares of Avidity, which closed Friday at $21.52, rose 4.6% in
premarket trading Monday.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
October 18, 2021 09:39 ET (13:39 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
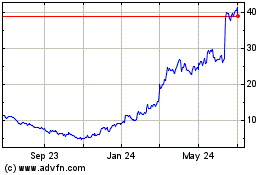
Avidity Biosciences (NASDAQ:RNA)
Historical Stock Chart
From Mar 2024 to Apr 2024
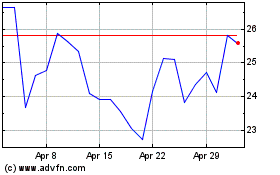
Avidity Biosciences (NASDAQ:RNA)
Historical Stock Chart
From Apr 2023 to Apr 2024