Marinus Pharmaceuticals Unveils New Brand as the Company Prepares for Commercialization
February 23 2021 - 7:00AM
Business Wire
Updated logo and brand identity highlight
Marinus’ focus on commitment, innovation and community as a leader
in rare epilepsies
Marinus Pharmaceuticals, Inc. (Nasdaq: MRNS), a pharmaceutical
company dedicated to the development of innovative therapeutics to
treat rare seizure disorders, today unveiled its new brand identity
rooted in the company’s guiding principles of commitment,
innovation and community which are signified by three
interconnected rings. The logo represents a new era as Marinus
continues to advance as a leader in rare epilepsies and prepares
for commercialization.
With patients as its focus, Marinus Pharmaceuticals strives to
create an awareness for the rare epilepsies it aims to treat. As
such, each of the company’s patient communities are embodied in the
new logo’s interconnected green, purple, and blue rings--the colors
that represent CDKL5 deficiency disorder, tuberous sclerosis
complex, status epilepticus, and PCDH19-related epilepsy.
“Our current and future success will be grounded in our
commitment to patients, the continued innovation of ganaxolone, and
our deep partnerships within the community,” said Christy Shafer,
Chief Commercial Officer for Marinus Pharmaceuticals. “This new
logo not only shows the shared interconnectivity and continued
dedication to these foundations but also our evolution as a
late-stage pharmaceutical company.”
In accordance with the rebrand, Marinus is unveiling a variety
of updated communication channels including its website, Twitter,
LinkedIn, Facebook and YouTube.
To learn more about Marinus Pharmaceuticals and its focus on
commitment, innovation, and community, visit:
www.marinuspharma.com.
About Marinus Pharmaceuticals
Marinus Pharmaceuticals, Inc. is a pharmaceutical company
dedicated to the development of innovative therapeutics to treat
rare seizure disorders. Ganaxolone is a positive allosteric
modulator of GABAA receptors that acts on a well-characterized
target in the brain known to have anti-seizure, antidepressant and
anti-anxiety effects. Ganaxolone is being developed in IV and oral
dose formulations intended to maximize therapeutic reach to adult
and pediatric patient populations in both acute and chronic care
settings. Marinus recently completed the first ever Phase 3 pivotal
trial in children with CDKL5 deficiency disorder and is conducting
a Phase 2 trial in tuberous sclerosis complex, as well as a Phase 2
biomarker-driven proof-of-concept trial in PCDH19-related epilepsy.
The company is initiating a Phase 3 trial in status epilepticus.
For more information visit www.marinuspharma.com.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210223005361/en/
Sasha Damouni Ellis Vice President, Investor Relations &
Corporate Communications Marinus Pharmaceuticals, Inc. 484-253-6792
sdamouni@marinuspharma.com
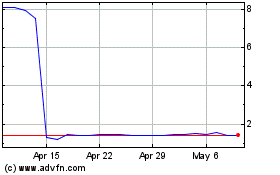
Marinus Pharmaceuticals (NASDAQ:MRNS)
Historical Stock Chart
From Mar 2024 to Apr 2024
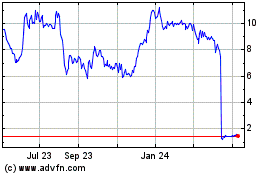
Marinus Pharmaceuticals (NASDAQ:MRNS)
Historical Stock Chart
From Apr 2023 to Apr 2024