Novo Nordisk Files FDA New Drug Application for Obesity Drug
December 04 2020 - 8:37AM
Dow Jones News
By Dominic Chopping
Danish pharmaceutical company Novo Nordisk A/S said Friday that
it has submitted a new drug application to the U.S. Food and Drug
Administration for its semaglutide weight-loss drug.
"Obesity is associated with a wide range of serious
complications, yet many healthcare providers still do not have
sufficient medical options available to help people with this
chronic disease," said Novo Nordisk Chief Scientific Officer Mads
Krogsgaard Thomsen.
Semaglutide is a glucagon-like peptide-1 drug, or GLP-1, that
helps enhance the production of insulin and regulate appetite.
Novo Nordisk said it expects the review of its application to
complete in six months.
Write to Dominic Chopping at dominic.chopping@wsj.com
(END) Dow Jones Newswires
December 04, 2020 08:22 ET (13:22 GMT)
Copyright (c) 2020 Dow Jones & Company, Inc.
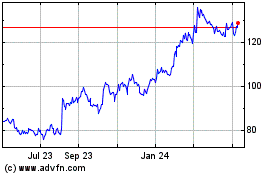
Novo Nordisk (NYSE:NVO)
Historical Stock Chart
From Mar 2024 to Apr 2024
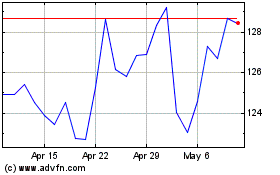
Novo Nordisk (NYSE:NVO)
Historical Stock Chart
From Apr 2023 to Apr 2024