Revance Announces Ground-Breaking Data Presentations at Leading International Neurotoxin Conference
January 15 2019 - 8:05AM
Business Wire
- Revance’s long-acting neuromodulator RT002,
along with its unique proprietary peptide and formulation
technology, will be featured in 11 podium and poster presentations
at the TOXINS 2019 conference in Denmark -
Revance Therapeutics, Inc. (NASDAQ:RVNC), a biotechnology
company developing next-generation neuromodulators for use in
treating aesthetic and therapeutic conditions, today announced 11
clinical and non-clinical presentations for DaxibotulinumtoxinA for
Injection (RT002) and the company’s unique proprietary peptide and
formulation technology at TOXINS 2019, a leading international
conference on neurotoxins to be held Jan. 16 through 19 in
Copenhagen, Denmark.
"We will have a significant presence at the bi-annual TOXINS
2019, which attracts scientific and clinical experts from around
the world who are specifically focused on the basic science and
clinical applications of neurotoxins," said Dan Browne, president
and chief executive officer at Revance. “Our 11 presentations will
cover a variety of evidence-based clinical results for our lead
product candidate RT002 in treating both glabellar (frown) lines
and cervical dystonia, along with detailed examinations of our
unique proprietary peptide and formulation technology. We’re
excited to share data on the first new neuromodulator in more than
30 years that delivers high response rates and long-lasting results
without compromising safety.”
Revance’s scheduled presentations are:
Podium Presentations
- Pooled Results from Two Phase 3 Pivotal
Studies of DaxibotulinumtoxinA for the Treatment of Glabellar Lines
(Jan. 17, 4:30 pm CET, Clinical Workshop 1B). Presenter: Dr. Steven
G. Yoelin, Ophthalmologist at Steve Yoelin MD Medical
Associates.
- Outcomes of Week-24 Completers and
Subjects who had Follow-Up Beyond Week 24 after a Single Treatment
of DaxibotulinumtoxinA; Results of a Phase 2 Open-Label
Dose-Escalating Study in Isolated Cervical Dystonia (Jan. 18, 3:30
pm CET, Clinical Workshop 3A). Presenter: Dr. Atul Patel, Kansas
City Bone & Joint Clinic.
Poster Presentations (Jan. 17 and 18, noon to 2:30 pm
CET)
Aesthetics
- Pooled Results from Two Phase 3 Pivotal
Studies of DaxibotulinumtoxinA for the Treatment of Glabellar
Lines
- Results of a Large Open-Label Safety
Study of DaxibotulinumtoxinA in Glabellar Lines
- A Head-to-Head Dose-Ranging Study of
DaxibotulinumtoxinA in the Treatment of Glabellar Lines: Results of
the BELMONT Study
Therapeutics
- Immunogenicity of DaxibotulinumtoxinA
in Adults with Cervical Dystonia from a Phase 2 Dose-Escalation
Multicenter Study
- Outcomes of Week-24 Completers and
Subjects who had Follow-Up Beyond Week 24 after a Single Treatment
of DaxibotulinumtoxinA: Results of a Phase 2 Open-Label,
Dose-Escalating Study in Isolated Cervical Dystonia
- Methodology of a Phase 2 Randomized,
Double-Blind, Placebo-Controlled, Parallel group, Dose-Ranging
36-week Multicenter Trial to Evaluate the Efficacy and Safety of
DaxibotulinumtoxinA for the Treatment of Upper Limb Spasticity in
Adults after Stroke or Traumatic Brain Injury
Non-Clinical
- Revance's Novel Peptide Excipient
(RTP004) and its Role in Stabilizing DaxibotulinumtoxinA against
Aggregation
- Development of Cell-Based Potency Assay
for Release and Stability Testing of Drug Substance and Drug
Product
- Non-Clinical Overview of
DaxibotulinumtoxinA to Support Registration for Human Use
- Novel Peptide Excipient RTP004 Enhances
the Binding of Botulinum Neurotoxin, a Cell Binding Domain HC to
Rat Brain Synaptosomes
- Stabilizing Effect of RTP004 on
Non-Specific Surface Adsorption in Drug Product Manufacturing of
DaxibotulinumtoxinA
About Revance Therapeutics, Inc.
Revance Therapeutics is an emerging Silicon Valley biotechnology
leader developing neuromodulators for the treatment of aesthetic
and therapeutic conditions. Revance uses a unique proprietary
peptide excipient technology to create novel, differentiated
therapies. The company’s lead compound, DaxibotulinumtoxinA for
Injection (RT002), is in clinical development for a broad range of
aesthetic and therapeutic indications, including glabellar lines,
cervical dystonia, plantar fasciitis, upper limb spasticity and
chronic migraine. RT002 has the potential to be the first
long-acting neuromodulator. The company is advancing a robust
pipeline of injectable and topical formulations of
daxibotulinumtoxinA. More information on Revance may be found at
www.revance.com.
“Revance Therapeutics” and the Revance logo are registered
trademarks of Revance Therapeutics, Inc.
Forward-Looking Statements
This press release contains forward-looking statements,
including statements related to Revance Therapeutics' 2018
financial outlook, expected cash burn and other financial
performance, the process and timing of, and ability to complete,
current and anticipated future clinical development of our
investigational drug product candidates, the initiation, design,
timing and results of our clinical studies, including the SAKURA 3
study of RT002, ASPEN Phase 3 program, JUNIPER Phase 2 and other
clinical programs relating to RT002 and related results and
reporting of such results; results of our non-clinical programs;
statements about our business strategy, timeline and other goals
and market for our anticipated products, plans and prospects;
including our pre-commercialization plans and timing of our
potential BLA filing for RT002 to treat glabellar (frown) lines;
and statements about our ability to obtain regulatory approval with
respect to our drug; and potential benefits of our drug product
candidates and our excipient peptide and other technologies.
Forward-looking statements are subject to risks and
uncertainties that could cause actual results to differ materially
from our expectations. These risks and uncertainties include, but
are not limited to: the outcome, cost, and timing of our product
development activities and clinical trials; the uncertain clinical
development process, including the risk that clinical trials may
not have an effective design or generate positive results; our
ability to obtain and maintain regulatory approval of our drug
product candidates; our ability to obtain funding for our
operations; our plans to research, develop, and commercialize our
drug product candidates; our ability to achieve market acceptance
of our drug product candidates; unanticipated costs or delays in
research, development, and commercialization efforts; the
applicability of clinical study results to actual outcomes; the
size and growth potential of the markets for our drug product
candidates; our ability to successfully commercialize our drug
product candidates and the timing of commercialization activities;
the rate and degree of market acceptance of our drug product
candidates; our ability to develop sales and marketing
capabilities; the accuracy of our estimates regarding expenses,
future revenues, capital requirements and needs for financing; our
ability to continue obtaining and maintaining intellectual property
protection for our drug product candidates; and other risks.
Detailed information regarding factors that may cause actual
results to differ materially from the results expressed or implied
by statements in this press release may be found in Revance's
periodic filings with the Securities and Exchange
Commission (the "SEC"), including factors described in the
section entitled "Risk Factors" of our quarterly report on Form
10-Q filed November 2, 2018. These forward-looking statements
speak only as of the date hereof. Revance disclaims any obligation
to update these forward-looking statements.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20190115005234/en/
INVESTORSRevance Therapeutics, Inc.:Jeanie Herbert(714)
325-3584jherbert@revance.comorBurns
McClellan, Inc.:John Grimaldi(212)
213-0006jgrimaldi@burnsmc.comorMEDIAGeneral Media:TOGORUN:Mariann
Caprino(917) 242-1087m.caprino@togorun.comorTrade Media:Nadine
Tosk(504) 453-8344nadinepr@gmail.com
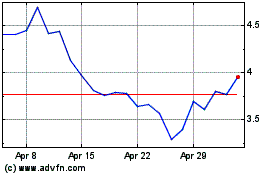
Revance Therapeutics (NASDAQ:RVNC)
Historical Stock Chart
From Mar 2024 to Apr 2024
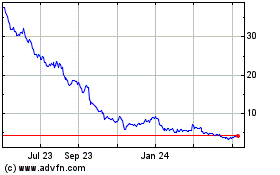
Revance Therapeutics (NASDAQ:RVNC)
Historical Stock Chart
From Apr 2023 to Apr 2024