Knight Therapeutics Announces the Canadian launch of PROBUPHINE™ for Opioid Use Disorder
October 29 2018 - 8:00AM

Knight Therapeutics Inc. (TSX:GUD) ("Knight"), a leading Canadian
specialty pharmaceutical company, announced today the commercial
availability of PROBUPHINE™ (buprenorphine hydrochloride subdermal
implant), for the management of opioid dependence in patients
clinically stabilized on no more than 8 mg of sublingual
buprenorphine in combination with counseling and psychosocial
support1. Knight has the exclusive right to distribute PROBUPHINE
in Canada under a license agreement with Titan Pharmaceuticals,
Inc. (NASDAQ:TTNP).
“Probuphine offers new hope for those struggling
with the issues of compliance, burden and stigmatization that often
accompanies opioid dependence,” said Jonathan Ross Goodman, Chief
Executive Officer of Knight. “This is a devastating public health
crisis and we are proud to be at the forefront of helping improve
the lives of those in need.”
"The approval of Probuphine by Health Canada
earlier this year was a milestone and we look forward to supporting
our partner Knight Therapeutics in this next phase," said Titan
President and CEO Sunil Bhonsle. "The commercial launch of
Probuphine provides the Canadian medical community with a novel,
long-term treatment alternative that can provide benefits to many
patients suffering from this disease."
About PROBUPHINE™
PROBUPHINE™ is the only subdermal implant
designed to deliver buprenorphine continuously for 6 months
following a single treatment. PROBUPHINE™ was developed using
ProNeura™, a continuous drug delivery system developed by Titan
Pharmaceuticals Inc. and consists of a small, solid implant made
from a mixture of ethylene-vinyl acetate (EVA) and buprenorphine.
Four implants are inserted subdermally in the inner side of the
patient’s upper arm by a healthcare professional through an
in-office procedure and removed in a similar manner at the end of
the treatment period. PROBUPHINE™ must be inserted and
removed by a healthcare professional who has successfully completed
a live training program, the PROBUPHINE Education Program.
Knight Therapeutics Inc. obtained Health Canada approval for
PROBUPHINE™ in April 2018, the first buprenorphine implant for the
long-term maintenance treatment of opioid addiction.
About Knight Therapeutics
Inc.
Knight Therapeutics Inc., headquartered in
Montreal, Canada, is a specialty pharmaceutical company focused on
acquiring or in-licensing innovative pharmaceutical products for
the Canadian and select international markets. Knight Therapeutics
Inc.'s shares trade on TSX under the symbol GUD. For more
information about Knight Therapeutics Inc., please visit the
company's web site at www.gudknight.com or www.sedar.com.
Forward-Looking Statement
This document contains forward-looking
statements for Knight Therapeutics Inc. and its subsidiaries. These
forward-looking statements, by their nature, necessarily involve
risks and uncertainties that could cause actual results to differ
materially from those contemplated by the forward-looking
statements.
Knight Therapeutics Inc. considers the
assumptions on which these forward-looking statements are based to
be reasonable at the time they were prepared but cautions the
reader that these assumptions regarding future events, many of
which are beyond the control of Knight Therapeutics Inc. and its
subsidiaries, may ultimately prove to be incorrect. Factors and
risks, which could cause actual results to differ materially from
current expectations are discussed in Knight Therapeutics Inc.'s
Annual Report and in Knight Therapeutics Inc.'s Annual Information
Form for the year ended December 31, 2017. Knight Therapeutics Inc.
disclaims any intention or obligation to update or revise any
forward-looking statements whether as a result of new information
or future events, except as required by law.
For further information, please contact:
Knight Therapeutics Inc.Samira SakhiaPresident
and Chief Financial OfficerTel: 514-678-8930Fax: 514-481-4116
Email: info@gudknight.com Website: www.gudknight.com
For product and/or live training inquiries,
please contact:
Tel: 1 844 483 5636Email:
medinfo@gudknight.com
References
- PROBUPHINE™ Product Monograph, Buprenorphine hydrochloride
implant, Subdermal implant, 80 mg, Partial Opioid Agonist, April
18, 2018.
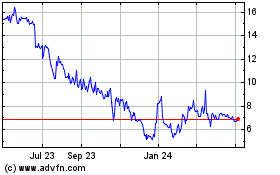
Titan Pharmaceuticals (NASDAQ:TTNP)
Historical Stock Chart
From Mar 2024 to Apr 2024
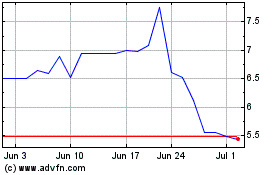
Titan Pharmaceuticals (NASDAQ:TTNP)
Historical Stock Chart
From Apr 2023 to Apr 2024