PARIS, July 31, 2018 /PRNewswire/ -- Sanofi (NYSE:
SNY; EURONEXT: SAN)
|
Q2
2018
|
Change
|
Change
at CER
|
H1
2018
|
Change
|
Change
at CER
|
|
IFRS net sales
reported
|
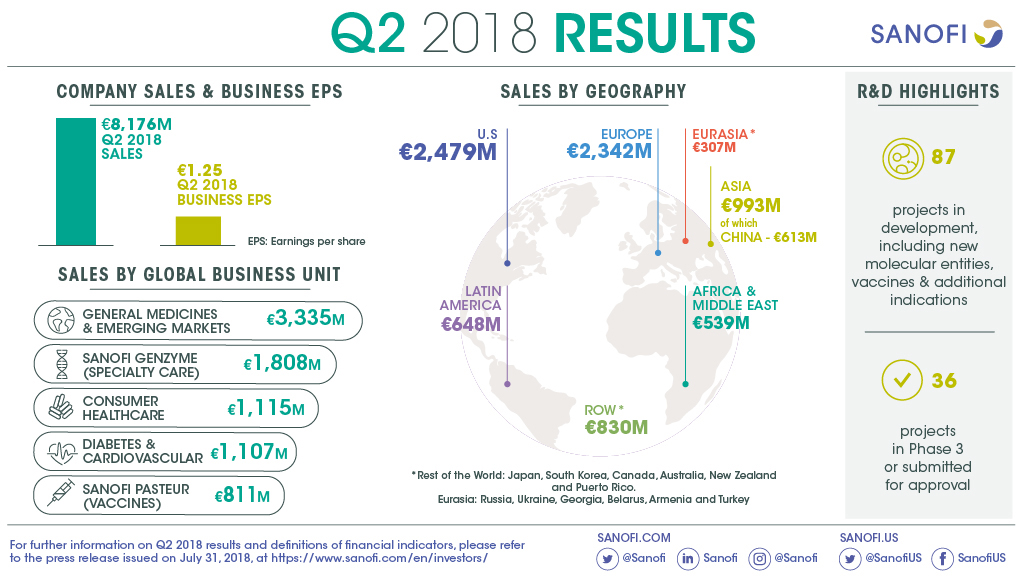
€8,176m
|
-5.7%
|
+0.1%
|
€16,074m
|
-7.2%
|
-0.1%
|
|
IFRS net income
reported
|
€762m
|
-26.2%
|
-
|
€1,778m
|
-73.6%(2)
|
-
|
|
IFRS EPS
reported
|
€0.61
|
-25.6%
|
-
|
€1.42
|
-73.4%(2)
|
-
|
|
Business net
income(1)
|
€1,558m
|
-7.9%
|
+0.4%
|
€3,156m
|
-9.4%
|
+0.4%
|
|
Business
EPS(1)
|
€1.25
|
-6.7%
|
+1.5%
|
€2.53
|
-8.3%
|
+1.4%
|
Experience the interactive Multichannel News Release here:
https://www.multivu.com/players/English/8375551-sanofi-earnings-results-q2-2018/
Sanofi Chief Executive Officer, Olivier Brandicourt, commented:
"In
the second quarter, we achieved significant milestones in building
our new Rare Blood Disorder franchise and the successful continued
execution of the global roll-out of Dupixent®. As
the impact from the U.S. losses of exclusivity peaked in the second
quarter, the growth of our diversified businesses largely
compensated for these headwinds. We look forward to entering
a new growth phase led by our increasing focus on Specialty Care
and our leadership positions in Emerging Markets and
Vaccines."
Second-quarter 2018 sales stable(3) with strong
contributions from Specialty Care and Emerging Markets
- Net sales were €8,176 million, down 5.7% on a reported basis,
up 0.1%(3) at CER and down 2.5% at CER/CS
(4).
- Sanofi Genzyme sales up 14.1% at CER/CS (4) (33.1%
at CER) driven by Dupixent® and consolidation of
Bioverativ.
- Vaccines sales down 15.7% reflecting high basis for comparison
and expected Pentaxim® supply constraint in China.
- CHC sales increased 4.1% supported by growth in Europe and Emerging
Markets(5).
- DCV(6) GBU sales down 15.6%; global Diabetes
franchise sales declined 11.9%, confirming expected trend for
year.
- Emerging Markets sales(5) increased 5.2% with
double-digit growth in China.
2018 business EPS guidance range slightly narrowed
- Second-quarter 2018 business EPS(1) up 1.5% at CER
to €1.25.
- Second-quarter 2018 IFRS EPS was €0.61 (-25.6%).
- Business EPS(1) in 2018 now expected to grow 3% to
5% at CER(7) barring unforeseen major adverse
events.
- Currency impact on 2018 business EPS is estimated to be around
-6% applying the average July exchange rates.
Key achievements in sustaining innovation in R&D
- Sanofi completed the acquisition of Ablynx in May,
internalizing the innovative Nanobody® platform.
- Positive CHMP recommendation for CabliviTM for
aTTP(8).
- Phase 1/2a data on BIVV001, an extended factor VIII therapy,
demonstrated half-life of 37 hours.
- A phase 2/3 study is being initiated on venglustat, an oral
glucosylceramide synthase (GCS) inhibitor, in
ADPKD(9).
- Positive phase 3 trial evaluating Dupixent® to treat
moderate-to-severe atopic dermatitis in adolescents.
- Priority review granted in the U.S. to cemiplimab for the
treatment of CSCC(10).
- ZynquistaTM (sotagliflozin) accepted for review by
the FDA in type 1 diabetes.
- Praluent® ODYSSEY OUTCOMES results submitted to the
FDA and EMA in Q2.
(1) In order to facilitate an understanding of operational
performance, Sanofi comments on the business net income statement.
Business net income is a non-GAAP financial measure (see Appendix 8
for definitions). The consolidated income statement for Q2 2018 is
provided in Appendix 3 and a reconciliation of reported IFRS net
income to business net income is set forth in Appendix 4; (2)
Excluding Animal Health gain on disposal, first-half IFRS net
income was down 22.6% and first-half IFRS EPS was down 22.4%;(3)
Changes in net sales are expressed at constant exchange rates (CER)
unless otherwise indicated (see Appendix 10); (4) Constant
Structure: Adjusted for Bioverativ acquisition; (5) See definition
page 8; (6) DCV: Diabetes and Cardiovascular; (7) 2017 business EPS
was €5.52; (8) Acquired thrombotic thrombocytopenic purpura; (9)
Autosomal Dominant Polycystic Kidney Disease; (10) Cutaneous
Squamous Cell Carcinoma
R&D update
Consult Appendix 6 for full overview of Sanofi's R&D
pipeline
Regulatory update
Regulatory updates since April 27,
2018 include the following:
- In June, the European Medicines Agency's (EMA) Committee for
Medicinal Products for Human Use (CHMP) recommended approval of
CabliviTM (caplacizumab) in the European Union
for the treatment of adults experiencing an episode of acquired
thrombotic thrombocytopenic purpura (aTTP), a rare blood-clotting
disorder. CabliviTM was developed by Ablynx, a Sanofi
Company.
- In May, ZynquistaTM (sotagliflozin) was
accepted for review by the U.S. Food and Drug Administration (FDA)
in type 1 diabetes.
- In April, the FDA accepted for priority review the Biologics
License Application (BLA) for cemiplimab (collaboration with
Regeneron) for the treatment of patients with metastatic cutaneous
squamous cell carcinoma (CSCC) or patients with locally advanced
CSCC who are not candidates for surgery.
- The results of the ODYSSEY OUTCOMES study, which showed
Praluent® (collaboration with Regeneron)
significantly reduced the risk of major adverse cardiovascular
events in patients who had suffered a recent acute coronary
syndrome, were submitted to the FDA and EMA in the second
quarter.
At the end of April 2018, the
R&D pipeline contained 87 projects including 40 new molecular
entities in clinical development. 36 projects are in phase 3 or
have been submitted to the regulatory authorities for approval.
Portfolio update
Phase 3:
- In June, positive non-inferiority results of the BRIGHT study
comparing Toujeo® to insulin degludec were
presented at the American Diabetes Association (ADA).
- In May, Sanofi and Regeneron announced that a pivotal phase 3
trial evaluating Dupixent® to treat
moderate-to-severe atopic dermatitis in adolescents (aged 12-17
years) met its primary and key secondary endpoints. The U.S.
regulatory submission for patients aged 12-17 years is planned for
third quarter 2018.
- Mavacamten (SAR439152/MYK461; partnership with MyoKardia), a
myosin inhibitor, moved into phase 3 in obstructive hypertrophic
cardiomyopathy (HCM). A phase 2 study in non-obstructive HCM also
started.
- A trial evaluating Cerdelga® in pediatric
type 1 Gaucher patients switching from ERT was initiated.
- A study evaluating Praluent® (collaboration
with Regeneron) in children with heterozygous familial
hypercholesterolemia (HeFH) was initiated.
- Sotagliflozin (partnership with Lexicon) moved into
phase 3 in patients with worsening heart failure in diabetes.
- A phase 2/3 study evaluating venglustat, an oral
glucosylceramide synthase (GCS) inhibitor, in patients at risk of
rapidly progressive Autosomal Dominant Polycystic Kidney Disease
(ADPKD) is in the process of being initiated.
- A cardiovascular outcome study, AMPLITUDE-O, evaluating
efpeglenatide was initiated.
Phase 2:
- ALX-0171, an anti RSV Nanobody®, entered into
Sanofi's portfolio through the acquisition of Ablynx.
- SAR440340, an anti-IL33
monoclonal antibody (partnership with Regeneron), is in the process
of being initiated in a phase 2 study in chronic obstructive
pulmonary disease (COPD).
- Phase 1/2a data on BIVV001, an extended factor VIII
therapy, was presented at the World Federation of Hemophilia (WFH)
and demonstrated a half-life of 37 hours.
- Decisions were taken to stop the development of SAR566658, a maytansin-loaded anti CA6
monoclonal antibody, in triple negative breast cancer and a
recombinant subunit vaccine against tuberculosis.
- ST-400, a gene editing technology (collaboration between
Sangamo and Ablynx), entered phase 2 in beta thalassemia.
- A phase 2 study evaluating dupilumab in grass
immunotherapy was initiated.
To access the full press release of the 2018 Q2 results,
please click here.
Forward-Looking Statements
This press release contains forward-looking statements as
defined in the Private Securities Litigation Reform Act of 1995, as
amended. Forward-looking statements are statements that are not
historical facts. These statements include projections and
estimates and their underlying assumptions, statements regarding
plans, objectives, intentions and expectations with respect to
future financial results, events, operations, services, product
development and potential, and statements regarding future
performance. Forward-looking statements are generally identified by
the words "expects", "anticipates", "believes", "intends",
"estimates", "plans" and similar expressions. Although Sanofi's
management believes that the expectations reflected in such
forward-looking statements are reasonable, investors are cautioned
that forward-looking information and statements are subject to
various risks and uncertainties, many of which are difficult to
predict and generally beyond the control of Sanofi, that could
cause actual results and developments to differ materially from
those expressed in, or implied or projected by, the forward-looking
information and statements. These risks and uncertainties include
among other things, the uncertainties inherent in research and
development, future clinical data and analysis, including post
marketing, decisions by regulatory authorities, such as the FDA or
the EMA, regarding whether and when to approve any drug, device or
biological application that may be filed for any such product
candidates as well as their decisions regarding labelling and other
matters that could affect the availability or commercial potential
of such product candidates, the absence of guarantee that the
product candidates if approved will be commercially successful, the
future approval and commercial success of therapeutic alternatives,
Sanofi's ability to benefit from external growth opportunities, to
complete related transactions, and/or obtain regulatory clearances,
risks associated with intellectual property and any related pending
or future litigation and the ultimate outcome of such
litigation, trends in exchange rates and prevailing interest
rates, volatile economic conditions, the impact of cost containment
initiatives and subsequent changes thereto, the average number of
shares outstanding as well as those discussed or identified in the
public filings with the SEC and the AMF made by Sanofi, including
those listed under "Risk Factors" and "Cautionary Statement
Regarding Forward-Looking Statements" in Sanofi's annual report on
Form 20-F for the year ended December 31,
2017. Other than as required by applicable law, Sanofi does
not undertake any obligation to update or revise any
forward-looking information or statements.




 View original
content:http://www.prnewswire.com/news-releases/q2-2018-performance-positions-sanofi-for-new-growth-phase-300689186.html
View original
content:http://www.prnewswire.com/news-releases/q2-2018-performance-positions-sanofi-for-new-growth-phase-300689186.html
SOURCE Sanofi