MARLBOROUGH, Mass.,
July 20, 2018 /PRNewswire/
-- Boston Scientific Corporation (NYSE: BSX) today announced
it has signed an agreement to acquire Claret Medical, Inc., a
privately-held company that has developed and commercialized the
Sentinel® Cerebral Embolic Protection System. The device
is used to protect the brain during certain interventional
procedures, predominately in patients undergoing transcatheter
aortic valve replacement (TAVR). The transaction price consists of
$220 million in up-front cash,
as well as a potential reimbursement-based milestone payment of up
to $50 million.
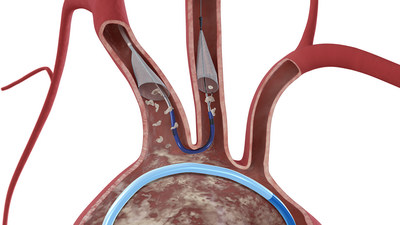
The Sentinel System – which received CE Mark in 2014 and FDA
clearance in 2017 – is the only device cleared to protect patients
against the risk of stroke during TAVR, a minimally-invasive
procedure to replace the aortic valve in patients with severe
aortic stenosis. Embolic debris such as calcium or tissue can break
loose during the procedure, travel through the bloodstream towards
the brain and potentially cause neurological and neurocognitive
damage. Recent studies have estimated approximately four percent of
patients experience a clinically-apparent stroke within 30 days of
a TAVR procedure.1,2,3,4,5,6
"Through the development and commercialization of the Sentinel
System, Claret Medical has successfully introduced a new layer of
safety and peace of mind for physicians and their patients
undergoing TAVR procedures," said Kevin
Ballinger, president, Interventional Cardiology, Boston
Scientific. "This acquisition will expand our commercial portfolio
to include an important adjunctive offering aimed at improving TAVR
patient outcomes. We also see potential for future use in other
left heart and endovascular procedures such as mitral valve repair
and replacement, left atrial appendage closure and pulmonary vein
isolation ablation procedures for atrial fibrillation."
In the pivotal SENTINEL trial, the landmark study that led to
FDA clearance and commercial introduction of this first-time
therapy in the U.S., the Sentinel System reduced the incidence of
strokes by 63 percent within the first 72 hours of the
procedure.7 In clinical studies, the Sentinel
System captured debris flowing towards the brain in 99 percent of
TAVR cases, regardless of the type of replacement valve that was
used.8
"We have been pleased by the consistent, high-quality clinical
evidence generated by the committed thought leaders at leading
academic centers, as well as the rapid physician adoption of the
device – utilized on average in 60 percent of TAVR procedures
performed across 100 centers – within just 12 months of
carefully-controlled U.S. commercialization," said Azin Parhizgar, PhD, president and chief
executive officer, Claret Medical. "Nearly 10,000 patients have
been treated worldwide with the Sentinel System, and we are
confident that the leadership of Boston Scientific will enable
increased momentum and improved patient access to this valuable
technology."
Claret Medical is based in Santa Rosa,
CA, and has approximately 120 employees. The acquisition is
projected to close during the third quarter of 2018, subject to
customary closing conditions.
The transaction is expected to be immaterial to adjusted
earnings per share in 2018, accretive in 2019 and increasingly
accretive thereafter. On a GAAP basis, the transaction is expected
to be less accretive, or more dilutive as the case may be, due to
amortization expense and acquisition-related net charges.
1 Leon, et al., N Engl J Med. 2010;363:1597-1607.
2 Webb, et al., J Am Coll Cardiol Intv.
2015;8:1797-1806.
3 Smith, et al., N Engl J Med. 2011;364:2187-98.
4 Leon, et al., N Engl J Med 2016;374:1609-20.
5 Popma, et al., J Am Coll Cardiol 2014;63:1972-81.
6 Adams, et al., N Engl
J Med 2014;370:1790-98.
7 Kapadia S, et al. JACC. Jan
2017; 69(4): 367-377.
8 Schmidt T, et al. JACC: Cardiovascular
Interventions. Jul 2018; 11(13): 1262-1273.
About Boston Scientific
Boston Scientific transforms lives through innovative medical
solutions that improve the health of patients around the world. As
a global medical technology leader for more than 35 years, we
advance science for life by providing a broad range of high
performance solutions that address unmet patient needs and reduce
the cost of healthcare. For more information,
visit www.bostonscientific.com and connect
on Twitter and Facebook.
Cautionary Statement Regarding Forward-Looking
Statements
This press release contains forward-looking statements within the
meaning of Section 27A of the Securities Act of 1933 and Section
21E of the Securities Exchange Act of 1934. Forward-looking
statements may be identified by words like "anticipate," "expect,"
"project," "believe," "plan," "estimate," "intend" and similar
words. These forward-looking statements are based on our beliefs,
assumptions and estimates using information available to us at the
time and are not intended to be guarantees of future events or
performance. These forward-looking statements include, among other
things, statements regarding the acquisition, the financial and
business impact of the transaction, product launches and product
performance and impact. If our underlying assumptions turn out to
be incorrect, or if certain risks or uncertainties materialize,
actual results could vary materially from the expectations and
projections expressed or implied by our forward-looking statements.
These factors, in some cases, have affected and in the future
(together with other factors) could affect our ability to implement
our business strategy and may cause actual results to differ
materially from those contemplated by the statements expressed in
this press release. As a result, readers are cautioned not to place
undue reliance on any of our forward-looking statements.
Factors that may cause such differences include, among other
things: future economic, competitive, reimbursement and regulatory
conditions; new product introductions; demographic trends; the
closing and integration of acquisitions; intellectual property;
litigation; financial market conditions; and future business
decisions made by us and our competitors. All of these factors are
difficult or impossible to predict accurately and many of them are
beyond our control. For a further list and description of these and
other important risks and uncertainties that may affect our future
operations, see Part I, Item 1A – Risk Factors in
our most recent Annual Report on Form 10-K filed with the
Securities and Exchange Commission, which we may update in Part II,
Item 1A – Risk Factors in Quarterly Reports on
Form 10-Q we have filed or will file hereafter. We disclaim any
intention or obligation to publicly update or revise any
forward-looking statements to reflect any change in our
expectations or in events, conditions or circumstances on which
those expectations may be based, or that may affect the likelihood
that actual results will differ from those contained in the
forward-looking statements. This cautionary statement is applicable
to all forward-looking statements contained in this document.
Use of Non-GAAP Financial Measures
To supplement our
consolidated financial statements presented on a GAAP basis, we
disclose certain non-GAAP financial measures, including adjusted
net income and adjusted net income (earnings) per share that
excludes certain charges and/or credits, such as amortization
expense and acquisition-related net charges (credits). These
non-GAAP financial measures are not in accordance with generally
accepted accounting principles in the
United States and should not be considered in isolation from
or as a replacement for the most directly comparable GAAP financial
measures. Further, other companies may calculate these non-GAAP
financial measures differently than we do, which may limit the
usefulness of those measures for comparative purposes. For further
information regarding our non-GAAP measures, see Part II, Item 7 -
Management's Discussion and Analysis of Financial Condition and
Results of Operations in our most recent Annual Report on Form
10-K, which we may update in Quarterly Reports on Form 10-Q we have
filed or will file hereafter.
CONTACTS:
Trish Backes
Media Relations
(651) 582-5887 (office)
trish.backes@bsci.com
Susie Lisa, CFA
Investor Relations
(508) 683-5565 (office)
BSXInvestorRelations@bsci.com
 View original content with
multimedia:http://www.prnewswire.com/news-releases/boston-scientific-announces-agreement-to-acquire-claret-medical-inc-300684162.html
View original content with
multimedia:http://www.prnewswire.com/news-releases/boston-scientific-announces-agreement-to-acquire-claret-medical-inc-300684162.html
SOURCE Boston Scientific Corporation