Report of Foreign Issuer (6-k)
March 21 2018 - 7:01AM
Edgar (US Regulatory)
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
Report of Foreign Private Issuer Pursuant
to Rule 13a-16 or 15d-16
Under the Securities Exchange Act of 1934
For the Month of March 2018
001-37353
(Commission File Number)
BIONDVAX PHARMACEUTICALS LTD.
(Exact name of Registrant as specified in
its charter)
14 Einstein St.
Ness Ziona
Israel 7414002
(Address of principal executive offices)
Indicate by check mark whether the registrant
files or will file annual reports under cover Form 20-F or Form 40-F.
Form 20-F ☒ Form 40-F ☐
Indicate by check mark if the registrant
is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ____
Indicate by check mark if the registrant
is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ____
Indicate by check mark whether by furnishing
the information contained in this Form, the registrant is also thereby furnishing the information to the Commission pursuant to
Rule 12g3-2(b) under the Securities Exchange Act of 1934.
Yes ☐ No ☒
If “Yes” is marked, indicate
below the file number assigned to the registrant in connection with Rule 12g3-2(b): 82-N/A
In connection with
BiondVax Pharmaceuticals Ltd.’s (hereinafter, the “Registrant”, the “Company”, or “BiondVax”)
previous Current Report on form 6-K, dated January 24, 2018, announcing the signing of a letter of intent with a European contract
research organization, or CRO, for the purpose of conducting a phase 3 clinical trial in Europe planned by the Company to begin
prior to the 2018/19 flu season, and in connection with the Company’s previous Report on form 6-K dated March 14, 2018, regarding
the appointment of the CRO, the Company hereby announces that on March 15, 2018, the Company and the CRO executed a master service
agreement and work order.
According to the master
service agreement, the Company undertakes to pay remuneration as well as to reimburse the CRO for costs incurred as a result of
the master service agreement and work orders. The master service agreement shall be in effect as of March 8, 2018 for a period
of five years or later, if a work order remains in effect, and until such work order’s completion. The first work order which
governs the conduct of the Company’s clinical trial in Europe is scheduled for a total period of 32.5 months. The Company
has a right to terminate the master service agreement or the work order by giving a 45 days’ notice or in the event of a
material breach.
SIGNATURES
Pursuant to the requirements
of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned,
thereunto duly authorized.
|
|
BiondVax Pharmaceuticals Ltd.
|
|
|
|
|
|
Date: March 21, 2018
|
By:
|
/s/ Ron Babecoff
|
|
|
|
Ron Babecoff
|
|
|
|
Chief Executive Officer
|
BiondVax Pharmaceuticals (NASDAQ:BVXV)
Historical Stock Chart
From Mar 2024 to Apr 2024
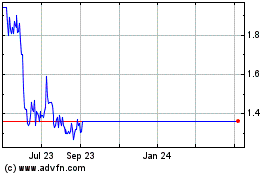
BiondVax Pharmaceuticals (NASDAQ:BVXV)
Historical Stock Chart
From Apr 2023 to Apr 2024