CLEVELAND, Oct. 17, 2017 /PRNewswire/ -- ViewRay, Inc.
(Nasdaq: VRAY), maker of the world's first and only clinical
MRI-guided radiation therapy system, announced today outcomes
data presented during the recent 2017 Annual Meeting of the
American Society for Radiation Oncology (ASTRO). This data
highlighted compelling early results using the company's MRIdian
system for the treatment of inoperable, locally advanced pancreatic
cancer.
Unlike any other system on the market today, MRIdian allows
clinicians to see the tumor and nearby soft-tissue anatomy
throughout radiation treatments using real-time diagnostic
MR-visualization. As a result, MRIdian's on-table adaptive
radiation therapy allows doctors to respond to subtle anatomical
changes observed on a day-to-day basis and reshape the dose to
better match the current contours of the tumor. By more accurately
targeting the tumor, and reducing treatment radiation dose to
surrounding organs such as the duodenum, small bowel, stomach and
liver, a higher and potentially more effective radiation dose may
be delivered without increasing the risks of side effects and
complications for the patient.
As featured in the ASTRO Scientific Session poster titled "High
Dose Adaptive MRI Guided Radiation Therapy Improves Overall
Survival of Inoperable Pancreatic Cancer," key data points and
findings from the presentation include the following:
- The study detailed a retrospective review of 42
locally-advanced pancreatic cancer patients treated with MRIdian at
four institutions (University of California
Los Angeles, University of
Wisconsin, VU University Medical Center, Washington University).
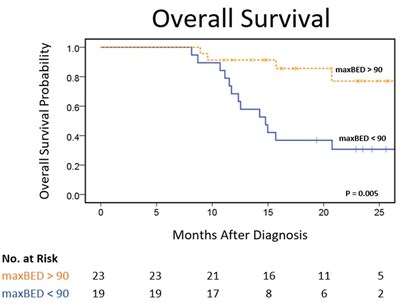
- The authors examined survival and toxicity rates for two unique
cohorts of patients. One sample received a higher biologically
effective dose (maxBED10 >90), primarily enabled by MRIdian
MR-guided on-table adaptive radiotherapy. The other sample received
a lower, more conventional biologically effective dose (maxBED10
<90), using non-adaptive therapy.
- The cohort receiving a higher dose demonstrated a near doubling
of median overall survival (Kaplan-Meier estimated median overall
survival of 27.8 months compared to 14.8 months).
- Patients treated with higher radiation doses reported no grade
3 or higher toxicities (0 percent). In comparison, those patients
receiving lower doses via non-adaptive treatments experienced 15.8
percent grade 3 or higher toxicities.
"High-definition MR now enables oncologists to detect the
slightest anatomical changes that occur from one day to the next
and in real-time while the patient is being treated. Coupled with
new adaptive radiation therapy software tools, we create new
customized plans in minutes, all while the patient is on the
treatment table," said Percy Lee,
M.D., senior author and Associate Professor and Vice Chair of
Education for the Department of Radiation Oncology at the David
Geffen School of Medicine at UCLA. "The
data presented at the 2017 ASTRO Annual Meeting suggests that
higher radiation doses with adaptive MR guided radiation therapy
may improve survival in pancreatic cancer while maintaining a very
favorable toxicity profile. These outcomes warrant further
investigations."
The early results highlighted above, along with other data
obtained using MRIdian will be tested in a multi-center,
prospective, single-arm clinical trial for inoperable, locally
advanced or borderline resectable pancreatic cancer. The trial will
be conducted by ViewRay's Clinical Cooperative Think Tank
(C2T2), a group of MRIdian medical
institutions focused on evidence gathering to support MR-guided
radiation therapy. For more information regarding this clinical
trial, please visit
http://go.viewray.com/pancreatic_cancer_trial_info.
Presentations discussing the multi-institutional poster in
ViewRay's ASTRO booth are available on the company's website
at http://www.viewray.com/ASTRO_2017.
About ViewRay
ViewRay®, Inc. (Nasdaq: VRAY), designs, manufactures and markets
the MRIdian® radiation therapy system. MRIdian is built upon a
proprietary high-definition MR imaging system designed from the
ground up to address the unique challenges and clinical workflow
for advanced radiation oncology. Unlike MR systems used in
diagnostic radiology, MRIdian's high-definition MR was purposely
built to deliver high-precision radiation without unnecessary beam
distortion, and consequently, help to mitigate skin toxicity and
other safety concerns that may otherwise arise when high magnetic
fields interact with radiation beams. ViewRay and MRIdian are
registered trademarks of ViewRay, Inc.
This press release contains forward-looking statements.
Statements in this press release that are not purely historical are
forward-looking statements. Such statements are subject to risks
and uncertainties that could cause future results to differ
materially from those referenced. Forward looking statements with
respect to predicted improved survival described in the poster
presentation and video, are based on current expectations from the
presented retroactive study and involve inherent risks and
uncertainties about the potential health and medical benefits of
MR-guided Radiation Therapy for the treatment of patients with
locally advanced pancreatic cancer. Further studies, including the
planned prospective multi-institutional study referenced in this
press release, as well as patient data covering longer periods of
time post-treatment, could lead to different or contrary results
from those expressed during ASTRO or in the poster presentation.
Given these uncertainties, the reader is advised not to place any
undue reliance on any forward-looking statements. Additional risk
factors include, among others, the ability to raise the additional
funding needed to continue to pursue ViewRay's business and product
development plans, the inherent uncertainties associated with
developing new products or technologies, competition in the
industry in which ViewRay operates and overall market conditions.
These forward-looking statements are made as of the date of this
press release, and ViewRay assumes no obligation to update the
forward-looking statements, or to update the reasons why actual
results could differ from those projected in the forward-looking
statements, except as required by law. Investors should consult all
of the information set forth herein and should also refer to the
risk factor disclosure set forth in the reports and other documents
ViewRay files with the SEC available at www.sec.gov.
View original content with
multimedia:http://www.prnewswire.com/news-releases/early-clinical-data-suggests-nearly-2x-prolonged-median-survival-for-inoperable-locally-advanced-pancreatic-cancer-with-mridian-mr-guided-radiation-therapy-300537789.html
SOURCE ViewRay, Inc.