- Desktop Health and Keystone are collaborating to make a broad
range of Keystone Industries 3D printing dental resins available on
Desktop Health’s new Einstein printer
- Partnership combines expertise in dental and photopolymer 3D
printing to bring high-speed and high-precision solutions to market
for dental applications spanning nightguards, splints, indirect
bonding trays, denture try-ins, and more
- KeySplint Soft, a market-leading resin developed by Keystone
for nightguards, dental splints, and bleaching trays is now
qualified on the Einstein printer
- Additional Keystone materials slated for qualification on the
Einstein platform include KeySplint Hard, KeyOrtho IBT, KeyDenture
Try-In, and KeyMask
- Desktop Health’s Einstein is a high-precision 3D printer
designed for dental professionals that leverages proprietary
NanoFit 385 and Hyperprint technology to fabricate a variety of
dental applications with ease and repeatability at high speeds
Desktop Health, a healthcare business within Desktop Metal, Inc.
(NYSE: DM) committed to developing 3D printing solutions for
personalized medicine, and Keystone Industries, a trusted dental
manufacturer of biocompatible, cutting-edge, patented photopolymer
resins, today announced a partnership to make a broad range of
Keystone 3D printing dental resins available on Desktop Health’s
Einstein printer, beginning with KeySplint Soft.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20220908005245/en/
KeySplint Soft is an FDA-510(k) cleared 3D printing resin for
the fabrication of flexible nightguards, dental splints, and
bleaching trays. This DLP-optimized resin combines the strength
needed to protect teeth with added flexibility for improved patient
comfort, all with a transparent and aesthetic appearance.
Through this partnership, Desktop Health and Keystone will
combine established resins with innovative print platforms to
deliver the highest productivity, precision and performance 3D
printing solutions to the dental market. The companies will
continue their collaboration to validate additional KeyPrint resins
for a range of dental applications on the Einstein platform over
the next several months, including KeySplint Hard, KeyOrtho IBT,
KeyDenture Try-In, and KeyMask. To learn more about KeySplint Soft
and the entire KeyPrint line of 3D printing resins, visit
https://keyprint.keystoneindustries.com/.
“We are excited to work with Keystone Industries to make
market-leading and trusted third-party resins available on our
breakthrough Einstein 3D printer for dental clinicians and labs,”
said Ric Fulop, Founder and CEO of Desktop Metal. “We believe this
partnership will help accelerate the adoption of digital solutions
in the dental industry by bringing new levels of precision and
productivity to advanced materials for a range of
applications.”
“Keystone Industries is committed to validating our KeyPrint
line of dental resins in the most innovative and widely-adopted 3D
printers for the dental market,” noted Ira Rosenau, president of
Keystone’s dental division. “Desktop Health and its Einstein 3D
printer fits well within the scope of our premium partnerships, and
we were able to quickly validate the Einstein as KeyPrint
Compatible, including with our leading KeySplint Soft material for
flexible night guards and splints. We are looking forward to
working with the creative Desktop Health team to offer dental
professionals an excellent combination of innovative resins with a
reliable, high-performance platform like the Einstein.”
Both the Einstein printer and KeySplint Soft resin are available
for purchase now. For more information, visit
https://health.desktopmetal.com/contact/.
Einstein Series Leveraging Digital Light Processing (DLP)
technology, the Einstein printer, designed specifically for dental
professionals, hits on all the key features essential to superior
3D printing: accuracy, speed, and versatility. Proprietary NanoFit
385 technology ensures that every application results in an
accurate fit and natural-looking finish with stunning clarity.
Equipped with Hyperprint™ technology to harness the power of heat
and closed-loop processing, the Einstein printer allows dental
professionals to fabricate a variety of dental applications, from
splints, guards, and models to dentures, with ease and
repeatability, and at up to 50 percent faster speeds than its
predecessor.
About Desktop Health Desktop Health, a healthcare
business within Desktop Metal and based in Burlington,
Massachusetts, is leveraging additive manufacturing and advanced
materials to personalize patient care. Desktop Health is vertically
integrated to develop, manufacture and commercialize applications
across a range of healthcare specialties, including dental, medical
and biofabrication. For more on Desktop Health, visit
www.desktophealth.com.
About Desktop Metal Desktop Metal, Inc., based in
Burlington, Massachusetts, is accelerating the transformation of
manufacturing with an expansive portfolio of 3D printing solutions,
from rapid prototyping to mass production. Founded in 2015 by
leaders in advanced manufacturing, metallurgy, and robotics, the
company is addressing the unmet challenges of speed, cost, and
quality to make additive manufacturing an essential tool for
engineers and manufacturers around the world. Desktop Metal was
selected as one of the world’s 30 most promising Technology
Pioneers by the World Economic Forum, named to MIT Technology
Review’s list of 50 Smartest Companies, and the 2021 winner of Fast
Company’s Innovation by Design Award in materials and Fast
Company’s Next Big Things in Tech Award for sustainability. For
more information, visit www.desktopmetal.com.
About Keystone Industries Keystone’s group of dental
companies focuses largely on consumable digital, laboratory,
operatory, and preventative products. The company is a global
supplier, maintaining a diverse network of more than 800 U.S. and
international dental distribution partners in 70+ countries.
Keystone has developed industry-leading products such as its
innovative KeyPrint line of 3D printing resins, Diamond D®
high-impact denture base, and the Pro-Form® line of thermoplastics,
innovative and patent-protected laboratory offerings such as
Enamelite ceramic spray glazes, and the award-winning Gelato prophy
paste. Keystone has spent nearly three decades formulating and
producing biocompatible, cutting-edge, patented photopolymer resins
for dental and cosmetic nail applications. Keystone maintains FDA
licensing, ISO 13485 (medical device) and 22716 certifications,
along with GMP certification and international product
registrations.
Forward-looking Statements This press release contains
certain forward-looking statements within the meaning of the
federal securities laws. Forward-looking statements generally are
identified by the words “believe,” “project,” “expect,”
“anticipate,” “estimate,” “intend,” “strategy,” “future,”
“opportunity,” “plan,” “may,” “should,” “will,” “would,” “will be,”
“will continue,” “will likely result,” and similar expressions.
Forward-looking statements are predictions, projections and other
statements about future events that are based on current
expectations and assumptions and, as a result, are subject to
risks, uncertainties. Many factors could cause actual future events
to differ materially from the forward-looking statements in this
document, including but not limited to, the risks and uncertainties
set forth in Desktop Metal, Inc.'s filings with the U.S. Securities
and Exchange Commission. These filings identify and address other
important risks and uncertainties that could cause actual events
and results to differ materially from those contained in the
forward-looking statements. Forward-looking statements speak only
as of the date they are made. Readers are cautioned not to put
undue reliance on forward-looking statements, and Desktop Metal,
Inc. assumes no obligation and does not intend to update or revise
these forward-looking statements, whether as a result of new
information, future events, or otherwise.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220908005245/en/
Media Relations: Sarah Webster (724)516-2336
Sarahwebster@desktopmetal.com
Investor Relations: Jay Gentzkow (781) 730-2110
jaygentzkow@desktopmetal.com
Desktop Metal (NYSE:DM)
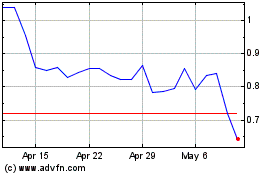
Historical Stock Chart
From Aug 2024 to Sep 2024
Desktop Metal (NYSE:DM)
Historical Stock Chart
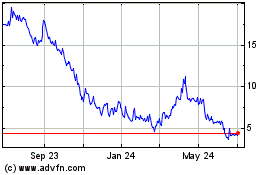
From Sep 2023 to Sep 2024