NEW HAVEN, Conn., Sept. 10, 2019 /PRNewswire/ -- Biohaven
Pharmaceutical Holding Company Ltd. (NYSE: BHVN), a biotechnology
company focused on advancing innovative therapies for neurological
and neuropsychiatric diseases, today announced completion of
enrollment in its Phase 2/3, double-blind, randomized,
placebo-controlled, dose-ranging trial of intranasally administered
vazegepant (formerly BHV-3500) for the acute treatment of migraine.
Vazegepant is a novel, structurally unique, third-generation
calcitonin gene-related peptide (CGRP) receptor antagonist and the
second product candidate in development for the acute treatment of
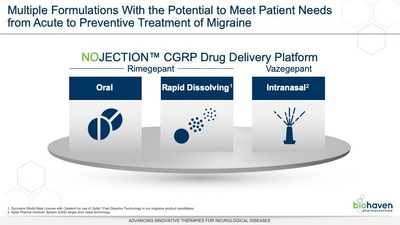
migraine from Biohaven's NOJECTION™ Migraine Platform.
The Phase 2/3 trial is designed to measure efficacy on
regulatory endpoints for acute treatment of migraine – pain freedom
and freedom from the most bothersome migraine-associated symptom at
2 hours post-dose – for vazegepant across three doses (5 mg, 10 mg
and 20 mg) versus placebo. The trial will also study other clinical
measures considered critically important to people living with
migraine, such as pain relief and the return to normal
functioning.
Elyse Stock, M.D., CMO of
Biohaven, commented, "Biohaven is proudly committed to innovating
new treatment options for people living with the debilitating
condition of migraine. Our Phase 2/3 trial of vazegepant represents
the first late-stage study of an intranasally delivered CGRP
receptor antagonist, propelling us a step closer to providing
patients with multiple, easy-to-use formulations to treat
migraine." Dr. Stock added, "Vazegepant is complementary to our
lead migraine asset, rimegepant, which has met the primary efficacy
endpoints in three completed Phase 3 clinical trials and is
currently under review with the FDA."
Intranasal vazegepant utilizes the Aptar Pharma Unidose System
(UDS), which is designed to enable systemic delivery of drugs
without the need for injection or administration by a healthcare
professional. This device is FDA-approved to deliver multiple drug
products marketed in the U.S., and is used by thousands of people
every day.
Robert Croop, M.D., Biohaven's
Chief Development Officer – Neurology, added, "We are excited to
complete enrollment with vazegepant, our third-generation CGRP
receptor antagonist, in this pivotal Phase 2/3 trial with the goal
of ultimately providing patients with important new treatment
options that can easily be self-administered whenever and wherever
a migraine strikes."
Vazegepant is the second of Biohaven's CGRP receptor-targeting
compounds to enter clinical trials. Biohaven's multiple CGRP
receptor antagonist product candidates, including rimegepant, and
expanded array of formulations including intranasal delivery and
Catalent's Zydis® oral fast-dissolve
tablet, are designed to meet patients' needs across the spectrum
from acute to preventive treatment of migraine.
About Migraine
Over 36 million Americans suffer from migraine. Migraine attacks
can differ in intensity and frequency, with many being highly
disabling. More than 90 percent of migraine sufferers are unable to
work or function normally during an attack. In the Global Burden of
Disease Study, updated in 2015, migraine was ranked as the seventh
highest cause worldwide of years lost due to disability. CGRP
receptor antagonists represent a novel class of drug candidates for
the treatment of migraine and are the first new class specific to
the acute treatment of migraine in over 25 years. This unique and
specific mode of action potentially offers an alternative to
current agents, particularly for patients who have
contraindications to the use of triptans, such as those with
underlying cardiovascular diseases, or who either do not respond or
have inadequate or inconsistent response to triptans or are
intolerant to them.
About Biohaven
Biohaven is a clinical-stage biopharmaceutical company with a
portfolio of innovative, late-stage product candidates targeting
neurological diseases, including rare disorders. Biohaven has
combined internal development and research with intellectual
property licensed from companies and institutions including
Bristol-Myers Squibb Company, AstraZeneca AB, Yale University, Catalent, Rutgers, and ALS Biopharma LLC. Currently,
Biohaven's lead development programs include multiple compounds
across its CGRP receptor antagonist, glutamate modulation, and
myeloperoxidase inhibitor platforms. Biohaven's common shares are
listed on the New York Stock Exchange and traded under the ticker
symbol BHVN. More information about Biohaven is available at
www.biohavenpharma.com.
About Aptar Pharma
Aptar Pharma is part of AptarGroup, Inc. (NYSE: ATR), a
leading global supplier of a broad range of innovative dispensing,
sealing and active packaging solutions for the beauty, personal
care, home care, prescription drug, consumer health care,
injectables, food and beverage markets. Aptar uses insights,
design, engineering and science to create innovative packaging
technologies that build brand value for its customers, and, in
turn, make a meaningful difference in the lives, looks, health and
homes of people around the world. Aptar is headquartered in
Crystal Lake, Illinois and has
over 14,000 dedicated employees in 18 different countries. For more
information, visit www.aptar.com/pharma. Media Contact:
Carolyn Penot, Aptar Pharma, +33 1
39 17 20 38, carolyn.penot@aptar.com
Forward-Looking Statements
This news release includes forward-looking statements within the
meaning of the Private Securities Litigation Reform Act of 1995.
These forward-looking statements involve substantial risks and
uncertainties, including statements that are based on the current
expectations and assumptions of the Company's management. All
statements, other than statements of historical facts, included in
this press release, including statements about the potential
safety, efficacy and attractive mode of administration of
vazegepant as a treatment for migraine, as well as its potential
for ultra-rapid onset and sustained activity, the potential of the
Company's CGRP receptor antagonist drug candidates to provide an
improved, effective and safe treatment option for the acute and
preventive treatment of migraine and the Company's expected
timelines for receipt of data from clinical trials, are
forward-looking statements. The use of certain words, including
"believe," "potential" and "will" and similar expressions, is
intended to identify forward-looking statements. The Company may
not actually achieve the plans and objectives disclosed in the
forward-looking statements, and you should not place undue reliance
on the Company's forward-looking statements. Various important
factors could cause actual results or events to differ materially
from those that may be expressed or implied by our forward-looking
statements, including those described in the "Risk Factors" section
of the Company's Annual Report on Form 10-K filed with the
Securities and Exchange Commission on February 28, 2019, the Company's Quarterly Report
on Form 10-Q for the quarter filed with the Securities and Exchange
Commission on August 9, 2019, and
other filings Biohaven makes with the U.S. Securities and Exchange
Commission from time to time. The forward-looking statements are
made as of this date and the Company does not undertake any
obligation to update any forward-looking statements, whether as a
result of new information, future events or otherwise, except as
required by law.
For further information, contact Dr. Vlad Coric, Chief Executive Officer, Biohaven at
Vlad.Coric@biohavenpharma.com
 View original content to download
multimedia:http://www.prnewswire.com/news-releases/biohaven-advances-nojection-migraine-platform-with-completion-of-enrollment-of-pivotal-phase-23-trial-of-vazegepant-the-first-intranasally-administered-cgrp-receptor-antagonist-in-development-for-the-acute-treatment-of-migraine-300914911.html
View original content to download
multimedia:http://www.prnewswire.com/news-releases/biohaven-advances-nojection-migraine-platform-with-completion-of-enrollment-of-pivotal-phase-23-trial-of-vazegepant-the-first-intranasally-administered-cgrp-receptor-antagonist-in-development-for-the-acute-treatment-of-migraine-300914911.html
SOURCE Biohaven Pharmaceutical Holding Company Ltd.