NEW HAVEN, Conn., Sept. 9, 2019 /PRNewswire/ -- Biohaven
Pharmaceutical Holding Company Ltd. (NYSE: BHVN) announced today
that it presented expanded data and post-hoc analyses from its long
term clinical study (BHV3000-201) demonstrating clinically
important benefits of rimegepant on migraine-specific disability
and quality of life in the late breaking oral and poster sessions
at the International Headache Conference in Dublin, Ireland.
Over 1,700 patients with migraine enrolled in this study.
Disability and quality of life outcome measures were assessed at
baseline and Weeks 12, 24, 36, and 52 using the Migraine Disability
Assessment (MIDAS) and the Migraine-Specific Quality of Life Scale
(MSQoL). Patients received rimegepant 75 mg as needed up to
once daily for the acute treatment of migraine. This dose has been
shown to produce statistically significant and clinically
meaningful benefits in three positive phase 3 pivotal trials. These
new analyses demonstrated improvements in migraine-related
disability that were clinically important and statistically
significant at all time points (p<.0001). Additionally,
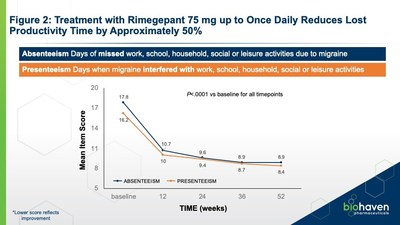
significant (p<.0001) improvements in absenteeism and
presenteeism items were seen throughout treatment follow-up.
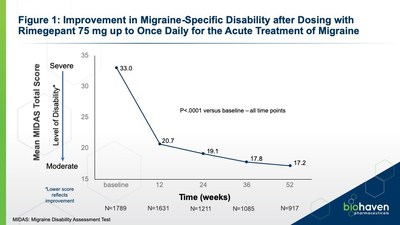
At baseline, the mean Total MIDAS Score was 33.8+0.8 (SE),
reflecting a severely disabled level of functioning for the study
population. Rimegepant demonstrated improvements in mean [95% C.I.]
change from baseline for Total MIDAS Score across weeks 12 to 52:
−12.5 [−13.93, −11.07] at Wk 12; −14.1 [−15.75, −12.51] at Wk 24;
−14.8 [−16.59, −13.01] at Wk 36; and −14.0 [−15.93, −12.13] at Wk
52. Improvements in MIDAS scores were clinically important and
statistically significant (P<.0001 versus baseline – all time
points). MIDAS is a validated 5-item instrument that queries
migraine-related absenteeism and lost productivity at work, school,
and home, as well as missed family, social, and leisure
activities.
Robert Croop, M.D., Biohaven's
Chief Development Officer – Neurology stated, "Acute treatment of
migraine with rimegepant over the one-year treatment period
decreased disability by approximately 41% as measured by the MIDAS
questionnaire reflecting improved patient outcomes in this study.
We believe that effective and well-tolerated oral acute therapies
for migraine will help patients return to their lives and improve
long-term outcomes."
The Migraine Specific Quality of Life (MsQOL, v2.1) is a
validated 14-item scale designed to measure how
migraine affects and/or limits daily functioning across three
domains: two domains assessing how migraine prevents activities
(Role-Restrictive [RR]; Role Preventative [RP]), and one domain
assessing the emotional impact of migraine (Emotional Role
[ER]). Mean MSQoL scores at baseline were 52.7 for RR, 67.9
for RP and 60.9 for ER, illustrating substantive negative impact of
migraine on patients. By Wk 12, rimegepant-treated subjects showed
clinically and statistically significant improvements: mean change
from baseline of +14.5, +11.5, and +15.4, for RR, RP, and ER
respectively, (p<0.0001). These benefits were sustained at Wk
24: +15.4,+12.8,+15.8 (p<0.0001), Wk 36: +17.4,+13.7+17.0,
(p<0.0001), and Wk 52: +17.6, +14.4, +17.2, (p<0.0001) for
RR, RP, and ER respectively.
Gil L'Italien, Ph.D., Head of Global Health Economics Outcomes
Research & Epidemiology at Biohaven stated, "These compelling
findings, showing sustained improvements in both migraine-specific
disability and quality of life, suggest an added long-term benefit
of rimegepant that derives from the effective acute treatment of
migraine. Both disability and quality of life improvements exceeded
the minimum clinically important difference by more than two-fold.
These benefits have important implications for reducing health care
costs, improving workplace productivity, and overall enhanced
patient wellbeing."
About Rimegepant and Study BHV3000-201
Rimegepant is Biohaven's orally-dosed calcitonin gene-related
peptide (CGRP) receptor antagonist, which the Company is developing
as a treatment for migraine. The Company submitted New Drug
Applications ("NDAs") with the U.S. Food and Drug Administration
(the "FDA") in the second quarter of this year and has disclosed
filing acceptance. Rimegepant represents a novel mechanism that
targets the underlying pathophysiology of migraine without causing
vasoconstriction. The efficacy and safety profile of rimegepant for
the acute treatment of migraine has now been established across
four randomized controlled trials to date: the three completed
pivotal Phase 3 trials, and a Phase 2b trial. Additional details on the Phase 3
trials are available in NEJM and The Lancet publications. The
company has submitted New Drug Applications with the U.S. Food and
Drug Administration which are currently under review.
BHV-3000-201 is a multicenter, long-term, open-label safety
study of rimegepant 75 mg oral tablet, comprising over 1,700
enrolled subjects. Eligible subjects included adults with ≥1 year
history of migraine. Subjects could treat migraine attacks of any
pain intensity with rimegepant 75 mg up to once daily over the
treatment period. Disability was assessed at baseline and
Weeks 12, 24, 36, and 52 using the Migraine Disability Assessment
(MIDAS) questionnaire. The Migraine-Specific Quality of Life Scale
(MSQoL) was also administered at the same time points to
measure how migraine affects and/or limits daily
functioning.
About Biohaven
Biohaven is a clinical-stage biopharmaceutical company with a
portfolio of innovative, late-stage product candidates targeting
neurological diseases, including rare disorders. Biohaven has
combined internal development and research with intellectual
property licensed from companies and institutions including
Bristol-Myers Squibb Company, AstraZeneca AB, Yale University, Catalent, Rutgers, and ALS Biopharma LLC. Currently,
Biohaven's lead development programs include multiple compounds
across its CGRP receptor antagonist, glutamate modulation, and
myeloperoxidase inhibitor platforms. Biohaven's common shares are
listed on the New York Stock Exchange and traded under the ticker
symbol BHVN. More information about Biohaven is available at
www.biohavenpharma.com.
Forward-Looking Statements
This news release includes forward-looking statements within the
meaning of the Private Securities Litigation Reform Act of 1995.
These forward-looking statements involve substantial risks and
uncertainties, including statements that are based on the current
expectations and assumptions of the Company's management. All
statements, other than statements of historical facts, included in
this press release regarding the Company's business and product
candidate plans and objectives are forward-looking statements.
Forward-looking statements include those related to: the
effectiveness and safety of rimegepant, the expected timing,
commencement and outcomes of the Company's planned and ongoing
clinical trials, the timing of planned interactions and filings
with the FDA, the timing and outcome of expected regulatory
filings, the potential commercialization of the Company's product
candidates and the potential for the Company's product candidates
to be first in class or best in class therapies. The use of certain
words, including "believe", "continue", "may", "on track",
"expects" and "will" and similar expressions, are intended to
identify forward-looking statements. Various important factors
could cause actual results or events to differ materially from
those that may be expressed or implied by our forward-looking
statements. Additional important factors to be considered in
connection with forward-looking statements are described in the
"Risk Factors" section of the Company's Annual Report on Form 10-K
filed with the Securities and Exchange Commission on February 28, 2019 and the Company's Quarterly
Report on Form 10-Q for the quarter filed with the Securities and
Exchange Commission on August 9,
2019. The forward-looking statements are made as of this
date and the Company does not undertake any obligation to update
any forward-looking statements, whether as a result of new
information, future events or otherwise, except as required by
law.
For further information, contact Dr. Vlad Coric, Biohaven's Chief Executive Officer
at Vlad.Coric@biohavenpharma.com
 View original content to download
multimedia:http://www.prnewswire.com/news-releases/biohaven-presents-data-demonstrating-reduction-in-migraine-related-disability-and-improvement-in-patient-reported-outcomes-after-oral-treatment-with-rimegepant-at-the-international-headache-conference-late-breaking-session-300913806.html
View original content to download
multimedia:http://www.prnewswire.com/news-releases/biohaven-presents-data-demonstrating-reduction-in-migraine-related-disability-and-improvement-in-patient-reported-outcomes-after-oral-treatment-with-rimegepant-at-the-international-headache-conference-late-breaking-session-300913806.html
SOURCE Biohaven Pharmaceutical Holding Company Ltd.