- Now available in the U.S., Pataday® Once Daily Relief is the
first and only eye drop to provide all day allergy itch relief
without a prescription1,2
- Approximately 66 million Americans who suffer from eye
allergies will now have OTC access to long-lasting,
prescription-strength relief that works in minutes1,2, 4-7
- National retail availability and robust media campaign to
begin March 2 in time for spring allergy season
Regulatory News:
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20200217005067/en/
Pataday Once Daily Relief® Packaging
Image (Photo: Business Wire)
Alcon (SIX/NYSE: ALC), the global leader in eye care dedicated
to helping people see brilliantly, today announced that Pataday
Once Daily Relief® (olopatadine hydrochloride ophthalmic solution
0.2%) and Pataday Twice Daily Relief® (olopatadine hydrochloride
ophthalmic solution 0.1%) have been approved by the Food and Drug
Administration (FDA) for sale over the counter (OTC) in the U.S.1,2
The Pataday brand features the No. 1 doctor-prescribed eye allergy
itch relief ingredient, and includes the first once daily allergy
itch relief drop to be available without a prescription for the
approximately 66 million Americans who suffer from eye
allergies.1,2, 4-7
“At Alcon, we are focused on delivering new eye care products
and solutions that meet the needs of patients and consumers,
including those who suffer from allergies,” said Sergio Duplan,
Region President, North America at Alcon. “We are proud to launch
prescription-strength Pataday as a convenient, more easily
accessible, over-the-counter option to relieve the itchy eyes that
almost 1-in-5 Americans experience due to ocular allergies.”
The rights to the Rx-to-OTC switch were secured by Alcon as part
of its separation from Novartis in April 2019. Alcon will market
both Patanol Solution (olopatadine 0.1%) and Pataday Solution
(olopatadine 0.2%) under the Pataday brand name as Pataday Twice
Daily Relief and Pataday Once Daily Relief, respectively. Both
strengths provide long-lasting, itchy eye relief that works in
minutes.1,2
“While eye allergies impact millions of Americans, only a small
percentage of those use over-the-counter allergy eye drops likely
due to a lack of awareness of effective options that treat the
problem at the source,” said Michael Cooper, OD, Solinsky Eye Care,
Hartford, CT. “For years, olopatadine has been my ‘go-to’ eye drop
for patients struggling with itchy allergy eyes and I’m thrilled
they will now be able to get the same relief over the counter
whenever they need it. My hope is that more allergy sufferers will
discover the benefits of Pataday and add it to their medicine
cabinet this spring allergy season and beyond.”
Pataday Once Daily Relief (olopatadine 0.2%) and Pataday Twice
Daily Relief (olopatadine 0.1%) are indicated for the temporary
relief of itchy eyes due to pollen, ragweed, grass, animal hair and
dander for ages 2 and older1,2. Pataday Twice Daily Relief
(olopatadine 0.1%) is also indicated for the temporary relief of
red eyes2. Since 2008, over 40 million prescriptions have been
written for olopatadine, the active ingredient in Pataday.3
Approximately 66 million Americans (1 in 5) suffer from ocular
allergies, but only 7 million use an over-the-counter allergy eye
drop to relieve their symptoms.4-7 By making Pataday (0.1% and
0.2%) available over the counter, more Americans will have access
to long-lasting, prescription-strength relief from the itching
associated with eye allergies.1,2
U.S. commercial availability of Pataday Once Daily Relief and
Pataday Twice Daily Relief will begin March 2 in time for the start
of spring allergy season, and will be available in all major drug,
food and mass market retailers – more than 40,000 stores – as well
as through online retailers. The launch is supported by a
multimillion-dollar media plan including a national TV advertising
campaign and strong online presence to reach eye allergy
sufferers.
Visit www.AboutPataday.com to learn more.
Forward-looking
Statements
This press release contains “forward-looking statements” within
the meaning of the safe harbor provisions of the United States
Private Securities Litigation Reform Act of 1995. Forward-looking
statements can be identified by words such as: “anticipate,”
“intend,” “commitment,” “look forward,” “maintain,” “plan,” “goal,”
“seek,” “believe,” “project,” “estimate,” “expect,” “strategy,”
“future,” “likely,” “may,” “should,” “will” and similar references
to future periods.
Forward-looking statements are neither historical facts nor
assurances of future performance. Instead, they are based only on
our current beliefs, expectations and assumptions regarding the
future of our business, future plans and strategies, and other
future conditions. Because forward-looking statements relate to the
future, they are subject to inherent uncertainties and risks that
are difficult to predict. Some of these factors are discussed in
our filings with the United States Securities and Exchange
Commission, including our Form 20-F. Should one or more of these
uncertainties or risks materialize, or should underlying
assumptions prove incorrect, actual results may vary materially
from those anticipated. Therefore, you should not rely on any of
these forward-looking statements.
Forward-looking statements in this press release speak only as
of the date they are made, and we assume no obligation to update
forward-looking statements as a result of new information, future
events or otherwise.
About Ocular Allergies
Ocular allergies, or allergic conjunctivitis, is the most common
allergy affecting the eyes.8 It occurs when the eye becomes exposed
to seasonal airborne pollutants such as grass, tree pollen and mold
spores.8 In addition, environmental effects such as pollution along
with dust mites and animal dander in the home can also lead to
allergic conjunctivitis. 8 In response to this irritation, the
body’s immune system releases a chemical known as histamine that
can cause eyes to itch or become irritated.9 Itchy eye is the most
common symptom of eye allergies.9
About Alcon
Alcon helps people see brilliantly. As the global leader in eye
care with a heritage spanning more than seven decades, we offer the
broadest portfolio of products to enhance sight and improve
people’s lives. Our Surgical and Vision Care products touch the
lives of more than 260 million people in over 140 countries each
year living with conditions like cataracts, glaucoma, retinal
diseases and refractive errors. Our more than 20,000 associates are
enhancing the quality of life through innovative products,
partnerships with eye care professionals and programs that advance
access to quality eye care. Learn more at www.alcon.com.
References
- PATADAY® [package insert]. Fort Worth, TX: Alcon Inc; 2010
- PATANOL® [package insert]. Fort Worth, TX: Alcon Inc; 2002
- IQVIA. Data on file. 2019.
- Gomes PJ. Trends in prevalence and treatment of ocular allergy.
Curr Opin Allergy Clin Immunol. 2014; 14: 451-456.
- Singh, K, Axelrod S, Bielory L. The epidemiology of ocular and
nasal allergy in the United States, 1988-1994.
- US Population Census 2019. 6. Nielsen Household Panel Data Feb.
2019.
- Nielsen Latest 52 weeks 2/6/2019
- AAAI Common Eye Symptoms Could be an Allergy
- Ono SJ, Abelson MB. Allergic conjunctivitis: Update on
pathophysiology and prospects for future treatment. J Allergy Clin
Immunol. 2005;115(1):118-122
Connect with us on Facebook LinkedIn
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200217005067/en/
Investor Relations Christina
Cheng + 41 589 112 110 (Geneva) + 1 817 615 2789 (Fort Worth)
investor.relations@alcon.com Media
Relations Steven Smith + 41 589 112 111 (Geneva) + 1 817
551 8057 (Fort Worth) globalmedia.relations@alcon.com
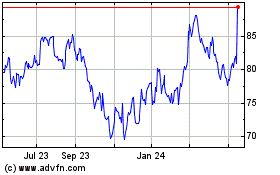
Alcon (NYSE:ALC)
Historical Stock Chart
From Aug 2024 to Sep 2024
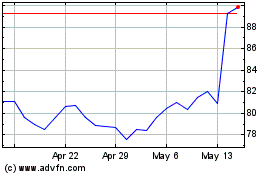
Alcon (NYSE:ALC)
Historical Stock Chart
From Sep 2023 to Sep 2024