Trevena Presents Phase 1 Data on TRV250 for Acute Migraine at American College of Neuropsychopharmacology 2019 Annual Meeting...
December 12 2019 - 7:00AM

Trevena, Inc. (Nasdaq: TRVN), a biopharmaceutical
company focused on the development and commercialization of novel
medicines for patients with central nervous system (CNS) disorders,
today announced a poster presentation at the 58th Annual Meeting of
the American College of Neuropsychopharmacology (ACNP), in Orlando,
FL. The poster presentation featured results from a Phase 1
first-in-human study that evaluated the safety, tolerability, and
pharmacokinetics (PK) of TRV250, the Company’s novel G
protein-selective delta-receptor agonist being developed for the
acute treatment of migraine.
TRV250 was well-tolerated up to 30 mg, with a PK profile
appropriate for an acute migraine therapy. There were no serious
adverse events and no clinically significant changes in EEG, ECG,
or other safety parameters.
“We are excited to be pursuing TRV250 as a potential new
therapeutic option for migraine sufferers, many of whom struggle to
achieve relief with currently available options,” said Mark A.
Demitrack, M.D., Senior Vice President and Chief Medical Officer of
Trevena, Inc. “The positive data from this Phase 1 study informed
our recently initiated proof-of-concept study, with topline data
anticipated in the second half of next year.”
Poster Presentation Details
- Poster presentation (#142): “A Phase 1 Healthy Volunteer Study
of the Safety, Tolerability and Pharmacokinetics of TRV250, a G
Protein-Selective Delta Receptor Agonist”, Wednesday, Dec. 11th,
5:00-7:00 p.m. EDT
About TRV250
TRV250 is a G protein biased ligand targeting the delta
receptor, with potential to be a first-in-class, non-narcotic
mechanism for the treatment of migraine. TRV250 also may have
utility in a range of other central nervous system indications. As
a selective delta receptor modulator, TRV250 is not expected to
have the abuse and addiction liability of medications targeting the
mu opioid receptor. TRV250 is an investigational product and has
not been approved by FDA for distribution in the US.
About Trevena
Trevena, Inc. is a biopharmaceutical company focused on the
development and commercialization of novel medicines for patients
with CNS disorders. The Company has four novel and differentiated
investigational drug candidates, including IV oliceridine, for the
management of moderate to severe acute pain in hospitals, TRV250
for the acute treatment of migraine, and TRV734 for maintenance
treatment of opioid use disorder. The Company has also identified
TRV045, a novel S1P receptor modulator that may offer a new,
non-opioid approach to managing chronic pain.
Cautionary note on forward looking
statements
Any statements in this press release about future expectations,
plans and prospects for the Company, including statements about the
Company’s strategy, future operations, clinical development of its
therapeutic candidates, plans for potential future product
candidates and other statements containing the words “anticipate,”
“believe,” “estimate,” “expect,” “intend,” “may,” “plan,”
“predict,” “project,” “suggest,” “target,” “potential,” “will,”
“would,” “could,” “should,” “continue,” and similar expressions,
constitute forward-looking statements within the meaning of The
Private Securities Litigation Reform Act of 1995.
Actual results may differ materially from those indicated by
such forward-looking statements as a result of various important
factors, including: the status, timing, costs, results and
interpretation of the Company’s clinical trials; the uncertainties
inherent in conducting clinical trials; expectations for regulatory
approvals; availability of funding sufficient for the Company’s
foreseeable and unforeseeable operating expenses and capital
expenditure requirements; uncertainties related to the Company’s
intellectual property; other matters that could affect the
availability or commercial potential of the Company’s therapeutic
candidates; and other factors discussed in the Risk Factors set
forth in the Company’s Annual Report on Form 10-K and Quarterly
Reports on Form 10-Q filed with the Securities and Exchange
Commission (SEC) and in other filings the Company makes with the
SEC from time-to-time.
In addition, the forward-looking statements included in this
press release represent the Company’s views only as of the date
hereof. The Company anticipates that subsequent events and
developments may cause the Company’s views to change. However,
while the Company may elect to update these forward-looking
statements at some point in the future, it specifically disclaims
any obligation to do so, except as may be required by law.
This press release is not sanctioned by ACNP.
For more information, please contact: Bob
Yoder, SVP and Chief Business Officer Trevena, Inc. Phone:
610-354-8840
Trevena (NASDAQ:TRVN)
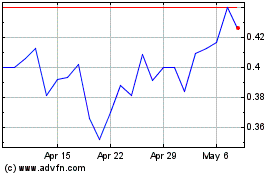
Historical Stock Chart
From Mar 2024 to Apr 2024
Trevena (NASDAQ:TRVN)
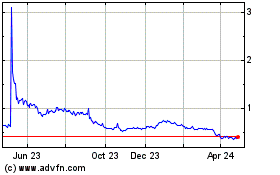
Historical Stock Chart
From Apr 2023 to Apr 2024